
Concept explainers
(a)
Interpretation:
The
Concept introduction:
VSEPR theory helps to draw the molecular shape as:
| Total number of valence electron pairs | Number of bond pairs | Number of lone pairs | Geometry and shape |
| 2 | 2 | 0 | linear |
| 3 | 3 | 0 | Trigonal planar |
| 2 | 1 | bent | |
| 4 | 4 | 0 | Tetrahedral |
| 3 | 1 | Pyramidal | |
| 2 | 2 | Bent |
Shape of molecules gives the bond angle.
(a)
Answer to Problem 49A
The
Explanation of Solution
Lewis structure of
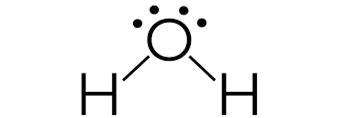
It shows, there are 2 bond pairs and 2 lone pairs around oxygen atom. Therefore, by VSEPR theory its shape is pyramidal and
(b)
Interpretation:
The
Concept introduction:
VSEPR theory helps to draw the molecular shape as:
| Total number of valence electron pairs | Number of bond pairs | Number of lone pairs | Geometry and shape |
| 2 | 2 | 0 | linear |
| 3 | 3 | 0 | Trigonal planar |
| 2 | 1 | bent | |
| 4 | 4 | 0 | Tetrahedral |
| 3 | 1 | Pyramidal | |
| 2 | 2 | Bent |
Shape of molecules gives the bond angle.
(b)
Answer to Problem 49A
The
Explanation of Solution
Lewis structure of
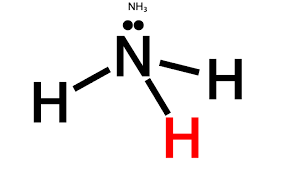
It shows, there are 3 bond pairs and 1 lone-pair around nitrogen atom. Therefore, by VSEPR theory its shape is pyramidal and bond angle is
(c)
Interpretation:
The
Concept introduction:
VSEPR theory helps to draw the molecular shape as:
| Total number of valence electron pairs | Number of bond pairs | Number of lone pairs | Geometry and shape |
| 2 | 2 | 0 | linear |
| 3 | 3 | 0 | Trigonal planar |
| 2 | 1 | bent | |
| 4 | 4 | 0 | Tetrahedral |
| 3 | 1 | Pyramidal | |
| 2 | 2 | Bent |
Shape of molecules gives the bond angle.
(c)
Answer to Problem 49A
The
Explanation of Solution
Lewis structure of
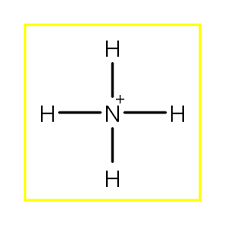
It shows, there are 4 bond pairs and no lone pairs around nitrogen atom. Therefore, by VSEPR theory its shape is tetrahedral and bond angle is normal tetrahedron angle.
(d)
Interpretation:
The
Concept introduction:
VSEPR theory helps to draw the molecular shape as:
| Total number of valence electron pairs | Number of bond pairs | Number of lone pairs | Geometry and shape |
| 2 | 2 | 0 | linear |
| 3 | 3 | 0 | Trigonal planar |
| 2 | 1 | bent | |
| 4 | 4 | 0 | Tetrahedral |
| 3 | 1 | Pyramidal | |
| 2 | 2 | Bent |
Shape of molecules gives the bond angle.
(d)
Answer to Problem 49A
The
Explanation of Solution
Lewis structure of
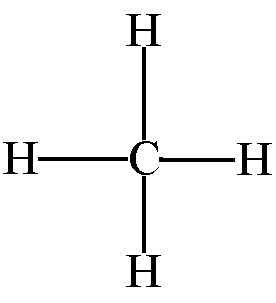
It shows, there are 4 bond pairs and no lone pairs around carbon atom. Therefore, by VSEPR theory its shape is tetrahedral and bond angle is normal tetrahedron angle.
Chapter 12 Solutions
World of Chemistry
- Indicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forwardIndicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forward
- Indicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forwardQuestion 3 (4 points), Draw a full arrow-pushing mechanism for the following reaction Please draw all structures clearly. Note that this intramolecular cyclization is analogous to the mechanism for halohydrin formation. COH Br + HBr Brarrow_forward
- Indicate the products obtained if 2,2-dimethylpropanal and acetaldehyde are mixed with sodium ethoxide in ethanol.arrow_forwardIndicate the products obtained if 2,2-dimethylpropanal and acetaldehyde are reacted with sodium ethoxide in ethanol.arrow_forward2,2-Dimethylpropanal and acetaldehyde are reacted with sodium ethoxide in ethanol. Indicate the products obtained.arrow_forward
- Add conditions above and below the arrow that turn the reactant below into the product below in a single transformationADS fint anditions 百 Abl res condinese NC ง Add on condtions 1.0 B H,N.arrow_forward3. Provide all the steps and reagents for this synthesis. OHarrow_forwardSteps and explanationarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





