
LCPO CHEMISTRY W/MODIFIED MASTERING
8th Edition
ISBN: 9780135214756
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 8.67SP
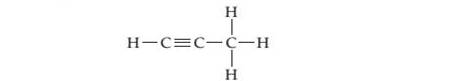
Describe the hybridization of each carbon atom in propyne
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Suppose the rate of evaporation in a hot, dry region is 1.76 meters per year, and the seawater there has a salinity of 35 ‰. Assuming a 93% yield, how much salt (NaCl) can be harvested each year from 1 km2 of solar evaporation ponds that use this seawater as a source?
help
Explain why only the lone pairs on the central atom are taken into consideration when predicting molecular shape
Chapter 8 Solutions
LCPO CHEMISTRY W/MODIFIED MASTERING
Ch. 8 - Prob. 8.1PCh. 8 - What is the number and geometric arrangement of...Ch. 8 - PRACTICE 8.3 Acetic acid, CH3CO2H , is the main...Ch. 8 - APPLY 8.4 Benzene, C6H6 , is a cyclic molecule in...Ch. 8 - PRACTICE 8.5 Identify the orbitals that overlap to...Ch. 8 - APPLY 8.6 Describe the bonding in propane, C3H8 ,...Ch. 8 - PRACTICE 8.7 Describe the hybridization of the...Ch. 8 - Describe the hybridization of each carbon atom in...Ch. 8 - Which orbitals overlap to form the sigma and pi...Ch. 8 - APPLY 8.10 Describe the hybridization of the...
Ch. 8 - Prob. 8.11PCh. 8 - Conceptual APPLY 8.12 Match the following...Ch. 8 - Prob. 8.13PCh. 8 - Prob. 8.14ACh. 8 - Prob. 8.15PCh. 8 - Prob. 8.16ACh. 8 - Prob. 8.17ACh. 8 - Prob. 8.18ACh. 8 - The B2 molecule has a MO diagram similar to that...Ch. 8 - Prob. 8.20ACh. 8 - PRACTICE 8.23 Draw two resonance structures for...Ch. 8 - APPLY 8.24 Draw two resonance structures for the...Ch. 8 - Prob. 8.23PCh. 8 - Prob. 8.24PCh. 8 - Caffeine is the most widely used stimulant...Ch. 8 - Prob. 8.26PCh. 8 - What is the geometry around the central atom in...Ch. 8 - What is the geometry around the central atom in...Ch. 8 - Three of the following molecular models have a...Ch. 8 - Identify each of the following sets of hybrid...Ch. 8 - The VSEPR model is a simple predictive tool that...Ch. 8 - The following ball-and-stick molecular model is a...Ch. 8 - The following ball-and-stick molecular model is a...Ch. 8 - Prob. 8.34CPCh. 8 - The dipole moment of methanol is =1.70D . Use...Ch. 8 - Methylarnine, CH3NH2 , is responsible for the odor...Ch. 8 - Prob. 8.37CPCh. 8 - Prob. 8.38SPCh. 8 - What shape do you expect for molecules that meet...Ch. 8 - How many charge clouds are there around the...Ch. 8 - Prob. 8.41SPCh. 8 - What shape do you expect for each of the following...Ch. 8 - What shape do you expect for each of the following...Ch. 8 - What shape do you expect for each of the following...Ch. 8 - Prob. 8.45SPCh. 8 - Prob. 8.46SPCh. 8 - What shape do you expect for each of the following...Ch. 8 - What bond angles do you expect for each of the...Ch. 8 - What bond angles do you expect for each of the...Ch. 8 - Acrylonitrile is used as the starting material for...Ch. 8 - Predict values for all bond angles in dimethyl...Ch. 8 - Oceanographers study the mixing of water masses by...Ch. 8 - A potential replacement for the chlorofluorocarbon...Ch. 8 - Explain why cyclohexane, a substance that contains...Ch. 8 - Like cyclohexane (Problem 8.54), benzene also...Ch. 8 - Use VSEPR theory to answer the following...Ch. 8 - Draw an electron-dot structure for each of the...Ch. 8 - What is the difference in spatial distribution...Ch. 8 - The average CC bond dissociation energy (D) is 350...Ch. 8 - What hybridization do you expect for atoms that...Ch. 8 - What spatial arrangement of charge clouds...Ch. 8 - What hybridization would you expect for the...Ch. 8 - What hybridization would you expect for the...Ch. 8 - Oxaloacetic acid is an intermediate involved in...Ch. 8 - The atoms in the amino acid glycine are connected...Ch. 8 - Describe the hybridization of the carbon atom in...Ch. 8 - Describe the hybridization of each carbon atom in...Ch. 8 - Bupropion, marketed as Wellbutr in, is a heavily...Ch. 8 - Efavirenz, marketed as Sustiva, is a medication...Ch. 8 - What is the hybridization of the B and N atoms in...Ch. 8 - Prob. 8.71SPCh. 8 - Aspirin has the following connections among atoms....Ch. 8 - The cation [HCNXeF]+ is entirely linear. Draw an...Ch. 8 - Acrylonitrile (C3H3N) is a molecule that is...Ch. 8 - The odor of cinnamon oil is due to cinnamaldehyde,...Ch. 8 - The following molecular model is a representation...Ch. 8 - Prob. 8.77SPCh. 8 - Which of the following substances would you expect...Ch. 8 - Which of the following substances would you expect...Ch. 8 - Why is the dipole moment of SO2 1.63 D hut that of...Ch. 8 - Prob. 8.81SPCh. 8 - The class of ions PtX42 , where X is a halogen,...Ch. 8 - Prob. 8.83SPCh. 8 - Prob. 8.84SPCh. 8 - Prob. 8.85SPCh. 8 - Prob. 8.86SPCh. 8 - Prob. 8.87SPCh. 8 - What are the most important kinds of...Ch. 8 - Of the substances Xe, CH3Cl , and HF which has:...Ch. 8 - Methanol (CH3OH;bp=65C) boils nearly 230 °C higher...Ch. 8 - Prob. 8.91SPCh. 8 - Prob. 8.92SPCh. 8 - Prob. 8.93SPCh. 8 - A liquid sample contains methylamine (CH3NH2)...Ch. 8 - Prob. 8.95SPCh. 8 - What is the difference in spatial distribution...Ch. 8 - Prob. 8.97SPCh. 8 - Use the MO energy diagram in Figure 8.22b to...Ch. 8 - Use the MO energy diagram in Figure 8.22 a to...Ch. 8 - The C2 molecule can be represented by an MO...Ch. 8 - Prob. 8.101SPCh. 8 - Prob. 8.102SPCh. 8 - Prob. 8.103SPCh. 8 - Draw a molecular orbital energy diagram for Li2 ....Ch. 8 - Calcium carbide, CaC2 , reacts with water to...Ch. 8 - At high temperatures, sulfur vapor is...Ch. 8 - Carbon monoxide is produced by incomplete...Ch. 8 - Make a sketch showing the location and geometry of...Ch. 8 - Make a sketch showing the location and geometry of...Ch. 8 - Prob. 8.110MPCh. 8 - Prob. 8.111MPCh. 8 - Prob. 8.112MPCh. 8 - Prob. 8.113MPCh. 8 - Just as individual bonds in a molecule are often...Ch. 8 - Cyclooctatetraenedian ion, C8H82 , is an organic...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (ME EX1) Prblm #9/10 Can you explain in detail (step by step) I'm so confused with these problems. For turmber 13 can u turn them into lewis dot structures so I can better understand because, and then as well explain the resonance structure part. Thanks for the help.arrow_forwardProblems 19 and 20: (ME EX1) Can you please explain the following in detail? I'm having trouble understanding them. Both problems are difficult for me to explain in detail, so please include the drawings and answers.arrow_forward(ME EX1) Prblm #4-11 Can you please help me and explain these I'm very confused in detail please. Prblm number 9 I don't understand at all (its soo confusing to me and redraw it so I can better depict it).arrow_forward
- ME EX1) Prblm #19-20 I'm so confused with these problems. Can you please help me solve them and explain them? Problems number 19-20, and thanks! step by step and in detail for me please helparrow_forwardCalculate the flux of oxygen between the ocean and the atmosphere, given that: Temp = 18°C Salinity = 35 ppt Density = 1025 kg/m3 Oxygen concentration measured in bulk water = 263.84 mmol/m3 Wind speed = 7.4 m/s Oxygen is observed to be about 10% initially supersaturatedarrow_forward( ME EX1) Prblm 27-28: Can you explain to me both prblms in detail and for prblm 28 what do you mean bi conjugated bi ponds and those structures I'm confused...arrow_forward
- A. Determine the number of electrons in a system of cyclic conjugation (zero if no cyclic conjugation). B. Specify whether the species is "a"-aromatic, "aa"-anti-aromatic, or "na"-non-aromatic (neither aromatic nor anti-aromatic). (Presume rings to be planar unless structure obviously prevents planarity. If there is more than one conjugated ring, count electrons in the largest.) 1. A.Electrons in a cyclic conjugated system. 18 B.The compound is (a, aa, or na) a 2. A.Electrons in a cyclic conjugated system. 10 B.The compound is (a, aa, or na) naarrow_forwardWater is boiling at 1 atm pressure in a stainless steel pan on an electric range. It is observed that 2 kg of liquid water evaporates in 30 min. Find the rate of heat transfer to the water (kW).arrow_forwardCould you please turn this into a complete Lewis dot structure formula for me so I can visualize it more clearly? and then do the explaining for the resonance structures that were given please.arrow_forward
- Could you please turn this into a complete Lewis dot structure formula for me so I can visualize it more clearly? and then do the explaining for the question.arrow_forwardplease solve. If the answer is "no error" and it asks me to type something, and i typed a-helix, its always wrong.arrow_forwardCan you please solve and explain this for me in a simple way? I cant seem to comprehend this problem.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY