
EBK ORGANIC AND BIOLOGICAL CHEMISTRY
7th Edition
ISBN: 9780100547742
Author: STOKER
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 4.94EP
Which carbon atom is the hemiacetal carbon atom in each of the following structures?
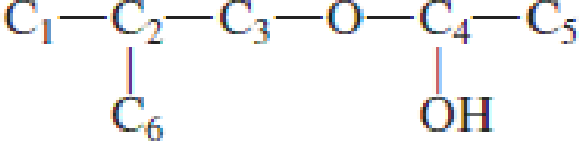
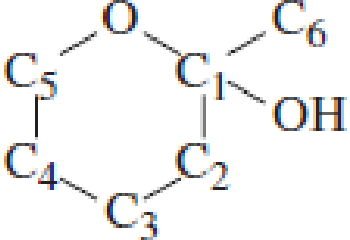
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
please help me with my homework
help
The temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the
pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes.
pressure (atm)
2
0
0
200
400
temperature (K)
X
Chapter 4 Solutions
EBK ORGANIC AND BIOLOGICAL CHEMISTRY
Ch. 4.1 - Prob. 1QQCh. 4.1 - Prob. 2QQCh. 4.2 - Prob. 1QQCh. 4.2 - Prob. 2QQCh. 4.2 - Prob. 3QQCh. 4.3 - Prob. 1QQCh. 4.3 - Prob. 2QQCh. 4.3 - Prob. 3QQCh. 4.4 - Prob. 1QQCh. 4.4 - Prob. 2QQ
Ch. 4.4 - Prob. 3QQCh. 4.4 - Prob. 4QQCh. 4.4 - Prob. 5QQCh. 4.5 - Prob. 1QQCh. 4.5 - Prob. 2QQCh. 4.5 - Prob. 3QQCh. 4.5 - Prob. 4QQCh. 4.5 - Prob. 5QQCh. 4.6 - Prob. 1QQCh. 4.6 - Prob. 2QQCh. 4.6 - Prob. 3QQCh. 4.7 - Prob. 1QQCh. 4.7 - Prob. 2QQCh. 4.8 - Prob. 1QQCh. 4.8 - Prob. 2QQCh. 4.9 - Prob. 1QQCh. 4.9 - Prob. 2QQCh. 4.10 - Prob. 1QQCh. 4.10 - Prob. 2QQCh. 4.10 - Prob. 3QQCh. 4.10 - Prob. 4QQCh. 4.11 - Prob. 1QQCh. 4.11 - Prob. 2QQCh. 4.11 - Prob. 3QQCh. 4.11 - Prob. 4QQCh. 4.11 - Prob. 5QQCh. 4.12 - Prob. 1QQCh. 4.12 - Prob. 2QQCh. 4 - Prob. 4.1EPCh. 4 - Prob. 4.2EPCh. 4 - Prob. 4.3EPCh. 4 - In terms of polarity, which carbonyl group atom...Ch. 4 - Prob. 4.5EPCh. 4 - Prob. 4.6EPCh. 4 - Prob. 4.7EPCh. 4 - Prob. 4.8EPCh. 4 - Prob. 4.9EPCh. 4 - Prob. 4.10EPCh. 4 - Prob. 4.11EPCh. 4 - Classify each of the following structures as an...Ch. 4 - Prob. 4.13EPCh. 4 - Prob. 4.14EPCh. 4 - Prob. 4.15EPCh. 4 - Prob. 4.16EPCh. 4 - Prob. 4.17EPCh. 4 - Prob. 4.18EPCh. 4 - Prob. 4.19EPCh. 4 - Prob. 4.20EPCh. 4 - Prob. 4.21EPCh. 4 - Prob. 4.22EPCh. 4 - Prob. 4.23EPCh. 4 - Prob. 4.24EPCh. 4 - Prob. 4.25EPCh. 4 - Prob. 4.26EPCh. 4 - Prob. 4.27EPCh. 4 - Prob. 4.28EPCh. 4 - Name the functional group(s) present in each of...Ch. 4 - Prob. 4.30EPCh. 4 - Prob. 4.31EPCh. 4 - Prob. 4.32EPCh. 4 - Prob. 4.33EPCh. 4 - Prob. 4.34EPCh. 4 - Prob. 4.35EPCh. 4 - Prob. 4.36EPCh. 4 - Draw a structural formula for each of the...Ch. 4 - Prob. 4.38EPCh. 4 - Prob. 4.39EPCh. 4 - Prob. 4.40EPCh. 4 - Draw a structural formula for each of the...Ch. 4 - Prob. 4.42EPCh. 4 - Prob. 4.43EPCh. 4 - Name the functional group(s) present in each of...Ch. 4 - Prob. 4.45EPCh. 4 - Prob. 4.46EPCh. 4 - Prob. 4.47EPCh. 4 - Prob. 4.48EPCh. 4 - Prob. 4.49EPCh. 4 - Give IUPAC names for all saturated...Ch. 4 - Prob. 4.51EPCh. 4 - Prob. 4.52EPCh. 4 - Prob. 4.53EPCh. 4 - Prob. 4.54EPCh. 4 - Prob. 4.55EPCh. 4 - Prob. 4.56EPCh. 4 - Prob. 4.57EPCh. 4 - Prob. 4.58EPCh. 4 - Prob. 4.59EPCh. 4 - Prob. 4.60EPCh. 4 - Prob. 4.61EPCh. 4 - Prob. 4.62EPCh. 4 - Prob. 4.63EPCh. 4 - Prob. 4.64EPCh. 4 - Which member in each of the following pairs of...Ch. 4 - Prob. 4.66EPCh. 4 - Which member in each of the following pairs of...Ch. 4 - Prob. 4.68EPCh. 4 - Prob. 4.69EPCh. 4 - How many hydrogen bonds can form between an...Ch. 4 - Prob. 4.71EPCh. 4 - Prob. 4.72EPCh. 4 - Draw the structure of the alcohol needed to...Ch. 4 - Prob. 4.74EPCh. 4 - Prob. 4.75EPCh. 4 - Prob. 4.76EPCh. 4 - Prob. 4.77EPCh. 4 - Prob. 4.78EPCh. 4 - Prob. 4.79EPCh. 4 - Prob. 4.80EPCh. 4 - Prob. 4.81EPCh. 4 - Which of the following compounds would react with...Ch. 4 - Prob. 4.83EPCh. 4 - Prob. 4.84EPCh. 4 - Which of the three compounds pentanal,...Ch. 4 - Prob. 4.86EPCh. 4 - Prob. 4.87EPCh. 4 - Prob. 4.88EPCh. 4 - Prob. 4.89EPCh. 4 - Prob. 4.90EPCh. 4 - Prob. 4.91EPCh. 4 - Indicate whether each of the following compounds...Ch. 4 - Which carbon atom is the hemiacetal carbon atom in...Ch. 4 - Which carbon atom is the hemiacetal carbon atom in...Ch. 4 - Prob. 4.95EPCh. 4 - Prob. 4.96EPCh. 4 - Prob. 4.97EPCh. 4 - Prob. 4.98EPCh. 4 - Prob. 4.99EPCh. 4 - Indicate whether each of the following compounds...Ch. 4 - Prob. 4.101EPCh. 4 - Prob. 4.102EPCh. 4 - Prob. 4.103EPCh. 4 - Prob. 4.104EPCh. 4 - Prob. 4.105EPCh. 4 - Prob. 4.106EPCh. 4 - Prob. 4.107EPCh. 4 - Name each of the compounds in Problem 15-106 in...Ch. 4 - Prob. 4.109EPCh. 4 - Prob. 4.110EPCh. 4 - Prob. 4.111EPCh. 4 - Prob. 4.112EPCh. 4 - Prob. 4.113EPCh. 4 - Prob. 4.114EPCh. 4 - Prob. 4.115EPCh. 4 - Prob. 4.116EPCh. 4 - Prob. 4.117EPCh. 4 - Prob. 4.118EP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- QUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forwarder your payment details | bar xb Home | bartleby x + aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1 O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 1 3- 0- 0 200 Explanation Check temperature (K) 400 X Q Search L G 2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Cearrow_forward
- 5.arrow_forward6.arrow_forward0/5 alekscgi/x/sl.exe/1o_u-IgNglkr7j8P3jH-IQs_pBaHhvlTCeeBZbufuBYTi0Hz7m7D3ZcSLEFovsXaorzoFtUs | AbtAURtkqzol 1HRAS286, O States of Matter Sketching a described thermodynamic change on a phase diagram The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 3 pressure (atm) + 0- 0 5+ 200 temperature (K) 400 Explanation Check X 0+ F3 F4 F5 F6 F7 S 2025 McGraw Hill LLC All Rights Reserved. Terms of Use Privacy Center Accessibility Q Search LUCR + F8 F9 F10 F11 F12 * % & ( 5 6 7 8 9 Y'S Dele Insert PrtSc + Backsarrow_forward
- 5.arrow_forward9arrow_forwardalekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IQs_pBanHhvlTCeeBZbufu BYTI0Hz7m7D3ZS18w-nDB10538ZsAtmorZoFusYj2Xu9b78gZo- O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 3- 200 temperature (K) Explanation Chick Q Sowncharrow_forward
- 0+ aleksog/x/lsl.exe/1ou-lgNgkr7j8P3H-IQs pBaHhviTCeeBZbufuBYTOHz7m7D3ZStEPTBSB3u9bsp3Da pl19qomOXLhvWbH9wmXW5zm O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 Gab The temperature on a sample of pure X held at 0.75 atm and -229. °C is increased until the sample sublimes. The temperature is then held constant and the pressure is decreased by 0.50 atm. On the phase diagram below draw a path that shows this set of changes. F3 pressure (atm) 0- 0 200 Explanation temperature (K) Check F4 F5 ☀+ Q Search Chill Will an 9 ENG F6 F7 F8 F9 8 Delete F10 F11 F12 Insert PrtSc 114 d Ararrow_forwardx + LEKS: Using a phase diagram a X n/alekscgi/x/lsl.exe/10_u-IgNsikr7j8P3jH-IQs_pBan HhvlTCeeBZbufu BYTI0Hz7m7D3ZcHYUt80XL-5alyVpw ○ States of Matter Using a phase diagram to find a phase transition temperature or pressure Use the phase diagram of Substance X below to find the melting point of X when the pressure above the solid is 1.1 atm. pressure (atm) 16 08- solid liquid- 0 200 400 gas 600 temperature (K) Note: your answer must be within 25 °C of the exact answer to be graded correct. × 5arrow_forwardS: Using a phase diagram leksogi/x/sl.exe/1ou-IgNs kr 7j8P3jH-IQs_pBan HhvTCeeBZbufuBYTI0Hz7m7D3ZdHYU+80XL-5alyVp O States of Matter Using a phase diagram to find a phase transition temperature or pressure se the phase diagram of Substance X below to find the boiling point of X when the pressure on the liquid is 1.6 atm. pressure (atm) 32- 16- solid liquid 0. gas 100 200 temperature (K) 300 Note: your answer must be within 12.5 °C of the exact answer to be graded correct. 10 Explanation Check § Q Search J 2025 McGraw Hill LLC. All Rights Researrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY