
Chemistry in Context
8th Edition
ISBN: 9780073522975
Author: American Chemical Society
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 7Q
Consider Figure 4.23.
- a. How does the present concentration of CO2 in the atmosphere compare with its concentration 20,000 years ago? With its concentration 120,000 years ago?
- b. How does the present temperature of the atmosphere compare with the 1950–1980 mean temperature? With the temperature 20,000 years ago? How does each of these values compare with the average temperature 120,000 years ago?
- c. Do your answers to parts a and b indicate causation, correlation, or no relation? Explain.
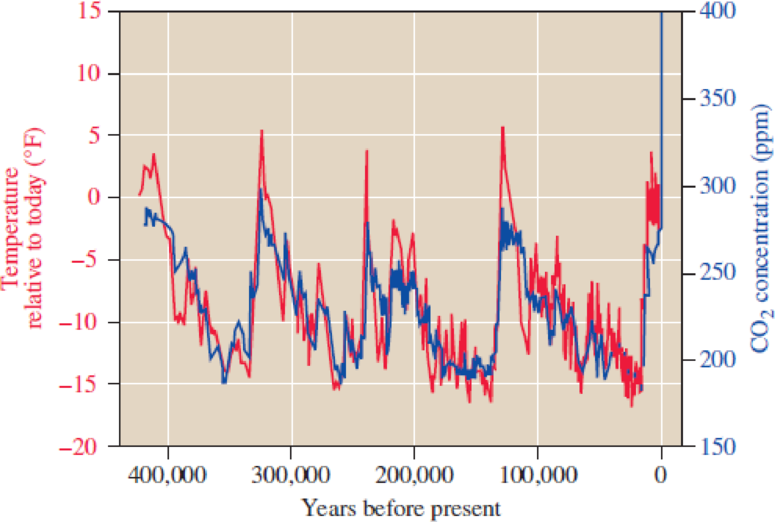
Figure 4.23
Carbon dioxide concentration (blue) and global temperatures (red) over the last 400,000 years from ice core data.
Source: Environmental Defense Fund
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Describe the mesomeric or resonance effect and differentiate between types +E or +M and -R or -M.
I need help with the following two problems, understanding them in a simple manner. Can you please draw them out for me with a detailed explanation so that I can better comprehend? I'm a visual person, so I definitely need that. Thank you very much!
Problem 54, could you please explain it in detail? Thank you! Step by step, I'm really confused, so please don't make it overly complex. My question is to visually draw it out and demonstrate it to me; I'm confused about that problem, please (not just in words) but demonstrate it to me in all due essence (visually) with descriptions.
Chapter 3 Solutions
Chemistry in Context
Ch. 3.1 - Prob. 3.3YTCh. 3.1 - You Decide Evaporating Coal Mines Writing in the...Ch. 3.2 - Prob. 3.6SCCh. 3.3 - Prob. 3.9YTCh. 3.4 - Prob. 3.10CTCh. 3.6 - Skill Building Isotopes of Nitrogen Nitrogen (N2)...Ch. 3.6 - You Decide Marshmallows and Pennies Avogadros...Ch. 3.7 - You Decide Checking Carbon from Cars A...Ch. 3.8 - Prob. 3.19YTCh. 3.8 - Prob. 3.20CT
Ch. 3.8 - Prob. 3.21CTCh. 3.9 - Prob. 3.22CTCh. 3.9 - Prob. 3.23YTCh. 3.9 - Prob. 3.24CTCh. 3.10 - Prob. 3.26CTCh. 3.10 - Prob. 3.27CTCh. 3.11 - Prob. 3.29CTCh. 3.11 - Prob. 3.30YTCh. 3.11 - Prob. 3.31YTCh. 3.11 - Prob. 3.32CTCh. 3.11 - You Decide Climate Change Insurance? Mitigation of...Ch. 3.11 - Prob. 3.34SCCh. 3.11 - Prob. 3.35CTCh. 3 - Prob. 3.1CTCh. 3 - The chapter concluded with a quote from John...Ch. 3 - Prob. 2QCh. 3 - Prob. 3QCh. 3 - Prob. 5QCh. 3 - a. It is estimated that 29 megajoules per square...Ch. 3 - Consider Figure 4.23. a. How does the present...Ch. 3 - Understanding Earths energy balance is essential...Ch. 3 - Explain each of these observations. a. A car...Ch. 3 - Prob. 10QCh. 3 - Draw the Lewis structure and name the molecular...Ch. 3 - Draw the Lewis structure and name the molecular...Ch. 3 - a. Draw the Lewis structure for methanol (wood...Ch. 3 - a. Draw the Lewis structure for ethene (ethylene),...Ch. 3 - Prob. 15QCh. 3 - Prob. 16QCh. 3 - Prob. 17QCh. 3 - Prob. 18QCh. 3 - Termites possess enzymes that allow them to break...Ch. 3 - Consider Figure 4.4. a. Which sector has the...Ch. 3 - Silver has an atomic number of 47. a. Give the...Ch. 3 - Silver only has two naturally occurring isotopes:...Ch. 3 - a. Calculate the average mass in grams of an...Ch. 3 - Prob. 24QCh. 3 - Prob. 25QCh. 3 - The total mass of carbon in living systems is...Ch. 3 - Prob. 27QCh. 3 - Prob. 28QCh. 3 - Prob. 29QCh. 3 - Prob. 30QCh. 3 - The Arctic has been called our canary in the coal...Ch. 3 - Do you think the comment made in the cartoon is...Ch. 3 - Prob. 33QCh. 3 - Prob. 34QCh. 3 - Over the last 20 years, about 120 billion tons of...Ch. 3 - Prob. 36QCh. 3 - Prob. 37QCh. 3 - Prob. 38QCh. 3 - Ethanol, C2H5OH, can be produced from sugars and...Ch. 3 - Prob. 40QCh. 3 - Prob. 41QCh. 3 - Prob. 42QCh. 3 - Prob. 43QCh. 3 - Prob. 44QCh. 3 - Prob. 45QCh. 3 - Prob. 46QCh. 3 - Prob. 47QCh. 3 - Prob. 48QCh. 3 - Prob. 49QCh. 3 - Prob. 50QCh. 3 - Former vice president Al Gore writes in his 2006...Ch. 3 - Prob. 52QCh. 3 - Prob. 53QCh. 3 - Data taken over time reveal an increase in CO2 in...Ch. 3 - In the energy industry, 1 standard cubic foot...Ch. 3 - Prob. 56QCh. 3 - A solar oven is a low-tech, low-cost device for...Ch. 3 - In 2005, the European Union adopted a...Ch. 3 - The world community responded differently to the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, and Biological Chemistry - 4th edition
What were the major microbiological interests of Martinus Beijerinck and Sergei Winogradsky? It can be said tha...
Brock Biology of Microorganisms (15th Edition)
To test your knowledge, discuss the following topics with a study partner or in writing ideally from memory. Th...
HUMAN ANATOMY
More than one choice may apply. Using the terms listed below, fill in the blank with the proper term. anterior ...
Essentials of Human Anatomy & Physiology (12th Edition)
Why is it unlikely that two neighboring water molecules would be arranged like this?
Campbell Biology (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- So, the first image is what I'm trying to understand regarding my approach. The second image illustrates my teacher's method, and the third image includes my notes on the concepts behind these types of problems.arrow_forwardHAND DRAWarrow_forwardDraw a mental model for calcium chloride mixed with sodium phosphatearrow_forward
- here is my question (problem number 20) please explain to me thanks!arrow_forwardThe bromination of anisole is an extremely fast reaction. Complete the resonance structures of the intermediate arenium cation for the reaction (Part 1), and then answer the question that follows (Part 2).arrow_forwardDrawing of 3-fluro-2methylphenolarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning  Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY