
(a)
Interpretation: An efficient synthesis for the given transformations has to be proposed.
Concept Introduction:
Friedel-Crafts Alkylation: The Friedel-Crafts alkylation involves the electrophilic substitution of alkyl groups on
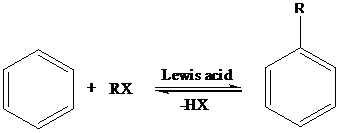
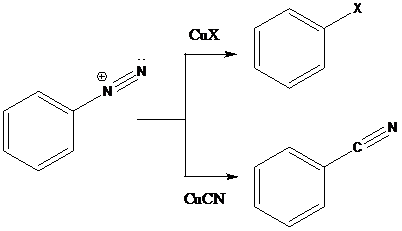
Sandmeyer reaction: Sandmeyer reactions use copper salts as the reagents. Here, aryldiazonium salt is converted into aryl halides or aryl cyanides by using copper halides or copper cyanides.
A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
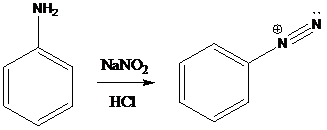
To find: Propose an efficient synthesis for the given transformation (a)
Decide the transformation
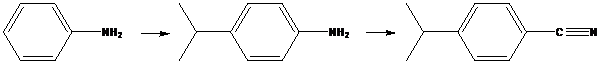
(b)
Interpretation: An efficient synthesis for the given transformations has to be proposed.
Concept Introduction:
Electrophilic aromatic substitution: Electrophilic aromatic substitution is a reaction in which the hydrogen atom of an aromatic ring is replaced as a result of an electrophilic attack on the aromatic ring. Here are three general steps to an electrophilic aromatic substitution.
- 1) Generation of an electrophile
- 2) Attack of the electrophile on the aromatic ring, creating a resonance-stabilized carbocation called an arenium ion. It loses aromaticity in this step, so the energy of activation is high. Furthermore, this is the rate-determining step of the reaction because of the disruption of aromaticity. The arenium ion is a hybrid resonance structure. There are three general resonance contributors of an arenium ion.
- 3) Deprotonation of the arenium ion by a weak base to regain aromaticity.
Sandmeyer reaction: Sandmeyer reactions use copper salts as the reagents. Here, aryldiazonium salt is converted into aryl halides or aryl cyanides by using copper halides or copper cyanides.

A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
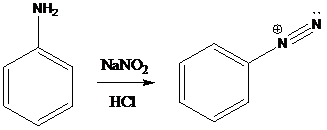
To find: Propose an efficient synthesis for the given transformation (b)
Apply a retrosynthetic analysis
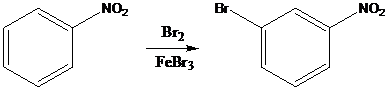
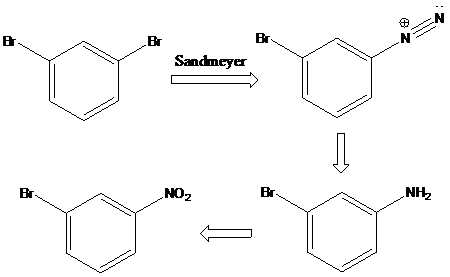
(c)
Interpretation: An efficient synthesis for the given transformations has to be proposed.
Concept Introduction:
Friedel–Crafts Acylation: The Friedel–Crafts acylation is the reaction of an arene with acyl chlorides or anhydrides using a strong Lewis acid catalyst. This reaction proceeds via electrophilic aromatic substitution to form monoacylated products.
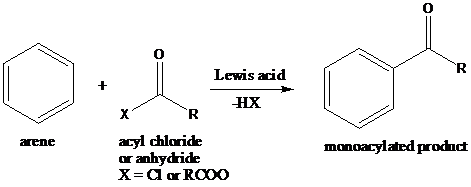
A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
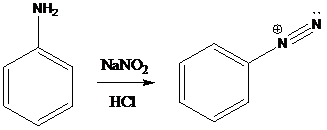
To find: Propose an efficient synthesis for the given transformation (c)
Apply a retrosynthetic analysis

(d)
Interpretation: An efficient synthesis for the given transformations has to be proposed.
Concept Introduction:
Friedel-Crafts Alkylation: The Friedel-Crafts alkylation involves the electrophilic substitution of alkyl groups on aromatic rings when arenes are treated with alkyl halides in presence of Lewis acids. This reaction is catalyzed by Lewis acids like anhydrous AlCl3, FeX3, ZnCl2, BF3 etc.
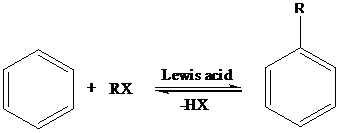
Sandmeyer reaction: Sandmeyer reactions use copper salts as the reagents. Here, aryldiazonium salt is converted into aryl halides or aryl cyanides by using copper halides or copper cyanides.

A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
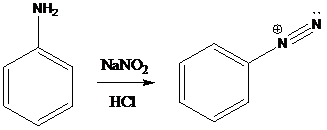
To find: Propose an efficient synthesis for the given transformation (d)
Apply a retrosynthetic analysis

(e)
Interpretation: An efficient synthesis for the given transformations has to be proposed.
Concept Introduction:
Electrophilic aromatic substitution: Electrophilic aromatic substitution is a reaction in which the hydrogen atom of an aromatic ring is replaced as a result of an electrophilic attack on the aromatic ring. Here are three general steps to an electrophilic aromatic substitution.
- 1) Generation of an electrophile
- 2) Attack of the electrophile on the aromatic ring, creating a resonance-stabilized carbocation called an arenium ion. It loses aromaticity in this step, so the energy of activation is high. Furthermore, this is the rate-determining step of the reaction because of the disruption of aromaticity. The arenium ion is a hybrid resonance structure. There are three general resonance contributors of an arenium ion.
- 3) Deprotonation of the arenium ion by a weak base to regain aromaticity.
Schiemann reaction: It is the reaction in which diazonium salt is converted into fluoro group using fluoroboric acid (HBF4).
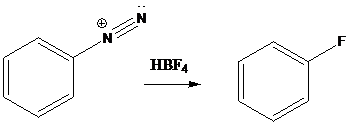
A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
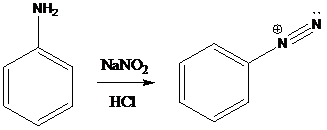
To find: Propose an efficient synthesis for the given transformation (e)
Apply a retrosynthetic analysis

(f)
Interpretation: An efficient synthesis for the given transformations has to be proposed.
Concept Introduction:
Sandmeyer reaction: Sandmeyer reactions use copper salts as the reagents. Here, aryldiazonium salt is converted into aryl halides or aryl cyanides by using copper halides or copper cyanides.

A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
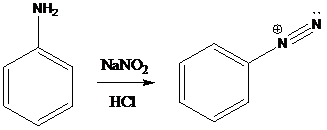
To find: Propose an efficient synthesis for the given transformation (f)
Perform elimination-addition process
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Organic Chemistry, Binder Ready Version
- Draw the mechanism for the formation of diol by starting with one pen and all in... basic conditions then acidic conditions then draw the mechanism for the formation of a carboxylic acid from your product.arrow_forwardDraw the mechanism for the oxidation of 3-bromo-cyclohexan-1-ol.arrow_forwardConvert the following Fischer projection to Haworth projections. show work and show the arrows please.arrow_forward
- Draw the mechanism for the substitution reaction converting an alcohol into an alkyl halide. If chirality is important to the reaction include it.arrow_forwardWrite, in words three different reactions we can use to make an alcohol.arrow_forwardDraw the reduction mechanism for the reduction of the aldehyde.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





