
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
8th Edition
ISBN: 9780134015187
Author: John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 17.43AP
Give systematic names for the following
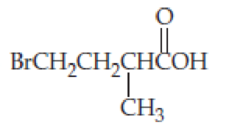
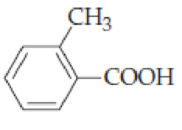
- (a) (CH3CH2)3CCOOH
- (b) CH3(CH2)5COOH
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
how would you make this plot in excel?
what is the product
What is the product?
Chapter 17 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
Ch. 17.1 - Identify the following molecules as a carboxylic...Ch. 17.1 - Prob. 17.2PCh. 17.1 - Prob. 17.3PCh. 17.1 - Prob. 17.4PCh. 17.1 - Prob. 17.5PCh. 17.1 - Prob. 17.6PCh. 17.1 - In the following pairs of compounds, which would...Ch. 17.1 - Write both condensed and line structures for (a)...Ch. 17.1 - Prob. 17.9PCh. 17.1 - Draw structures corresponding to these names: (a)...
Ch. 17.1 - Prob. 17.11PCh. 17.1 - Prob. 17.12PCh. 17.1 - Prob. 17.13KCPCh. 17.2 - Salsalate, which is an ester formed by the...Ch. 17.2 - Prob. 17.2CIAPCh. 17.2 - Prob. 17.3CIAPCh. 17.2 - Prob. 17.14PCh. 17.2 - Prob. 17.15PCh. 17.2 - Prob. 17.16PCh. 17.3 - Prob. 17.17PCh. 17.3 - Raspberry oil contains an ester that is made by...Ch. 17.3 - Prob. 17.19PCh. 17.3 - Prob. 17.20PCh. 17.3 - Prob. 17.21PCh. 17.4 - If a bottle of aspirin tablets has the aroma of...Ch. 17.4 - Prob. 17.23PCh. 17.4 - What carboxylic acids and amines result from...Ch. 17.5 - Prob. 17.25PCh. 17.5 - Prob. 17.26KCPCh. 17.6 - Prob. 17.27PCh. 17.6 - Prob. 17.28PCh. 17.6 - Prob. 17.4CIAPCh. 17.6 - Prob. 17.5CIAPCh. 17.6 - Prob. 17.29PCh. 17 - Prob. 17.30UKCCh. 17 - Prob. 17.31UKCCh. 17 - One phosphorylated form of glycerate is...Ch. 17 - Prob. 17.33UKCCh. 17 - Prob. 17.34UKCCh. 17 - Prob. 17.35UKCCh. 17 - Prob. 17.36UKCCh. 17 - For the following compounds, give the systematic...Ch. 17 - Write the equation for the ionization of hexanoic...Ch. 17 - Prob. 17.39APCh. 17 - Prob. 17.40APCh. 17 - Prob. 17.41APCh. 17 - Give systematic names for the following carboxylic...Ch. 17 - Give systematic names for the following carboxylic...Ch. 17 - Prob. 17.44APCh. 17 - Prob. 17.45APCh. 17 - Draw structures corresponding to the following...Ch. 17 - Draw structures corresponding to the following...Ch. 17 - Malic acid, a dicarboxylic acid found in apples,...Ch. 17 - Prob. 17.49APCh. 17 - Prob. 17.50APCh. 17 - Prob. 17.51APCh. 17 - Prob. 17.52APCh. 17 - Prob. 17.53APCh. 17 - Give systematic names for the following structures...Ch. 17 - Give systematic names for the following structures...Ch. 17 - Prob. 17.56APCh. 17 - Prob. 17.57APCh. 17 - Give systematic names for the following structures...Ch. 17 - Give systematic names for the following structures...Ch. 17 - Prob. 17.60APCh. 17 - What compounds are produced from hydrolysis of...Ch. 17 - Procaine, a local anesthetic whose hydrochloride...Ch. 17 - Prob. 17.63APCh. 17 - Lactones are cyclic esters in which the carboxylic...Ch. 17 - When both the carboxylic acid and the amine are in...Ch. 17 - LSD (lysergic acid diethylamide), a semisynthetic...Ch. 17 - Prob. 17.67APCh. 17 - Prob. 17.68APCh. 17 - Prob. 17.69APCh. 17 - Prob. 17.70APCh. 17 - Prob. 17.71APCh. 17 - Prob. 17.72APCh. 17 - Prob. 17.73APCh. 17 - Prob. 17.74APCh. 17 - Prob. 17.75APCh. 17 - Three amide isomers, N,N-dimethylformamide,...Ch. 17 - Prob. 17.77CPCh. 17 - Prob. 17.78CPCh. 17 - Mention at least two simple chemical tests by...Ch. 17 - Prob. 17.80CPCh. 17 - Name the following compounds.Ch. 17 - Each of the following materials has an ester that...Ch. 17 - Prob. 17.83GPCh. 17 - Prob. 17.84GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Problem 15 of 15 Submit Using the following reaction data points, construct Lineweaver-Burk plots for an enzyme with and without an inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Using the information from this plot, determine the type of inhibitor present. 1 mM-1 1 s mM -1 [S]' V' with 10 μg per 20 54 10 36 20 5 27 2.5 23 1.25 20 Answer: |||arrow_forward12:33 CO Problem 4 of 15 4G 54% Done On the following Lineweaver-Burk -1 plot, identify the by dragging the Km point to the appropriate value. 1/V 40 35- 30- 25 20 15 10- T Км -15 10 -5 0 5 ||| 10 15 №20 25 25 30 1/[S] Г powered by desmosarrow_forward1:30 5G 47% Problem 10 of 15 Submit Using the following reaction data points, construct a Lineweaver-Burk plot for an enzyme with and without a competitive inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. 1 -1 1 mM [S]' s mM¹ with 10 mg pe 20 V' 54 10 36 > ст 5 27 2.5 23 1.25 20 Answer: |||arrow_forward
- Problem 14 of 15 Submit Using the following reaction data points, construct Lineweaver-Burk plots for an enzyme with and without an inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Using the information from this plot, determine the type of inhibitor present. 1 mM-1 1 s mM -1 [S]' V' with 10 μg per 20 54 10 36 20 5 27 2.5 23 1.25 20 Answer: |||arrow_forward12:36 CO Problem 9 of 15 4G. 53% Submit Using the following reaction data points, construct a Lineweaver-Burk plot by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Based on the plot, determine the value of the catalytic efficiency (specificity constant) given that the enzyme concentration in this experiment is 5.0 μ.Μ. 1 [S] ¨‚ μM-1 1 V sμM-1 100.0 0.100 75.0 0.080 50.0 0.060 15.0 0.030 10.0 0.025 5.0 0.020 Answer: ||| O Гarrow_forwardProblem 11 of 15 Submit Using the following reaction data points, construct a Lineweaver-Burk plot for an enzyme with and without a noncompetitive inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. 1 -1 1 mM [S]' 20 V' s mM¹ with 10 μg per 54 10 36 > ст 5 27 2.5 23 1.25 20 Answer: |||arrow_forward
- Problem 13 of 15 Submit Using the following reaction data points, construct Lineweaver-Burk plots for an enzyme with and without an inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Using the information from this plot, determine the type of inhibitor present. 1 mM-1 1 s mM -1 [S]' V' with 10 μg per 20 54 10 36 20 5 27 2.5 23 1.25 20 Answer: |||arrow_forward12:33 CO Problem 8 of 15 4G. 53% Submit Using the following reaction data points, construct a Lineweaver-Burk plot by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Based on the plot, determine the value of kcat given that the enzyme concentration in this experiment is 5.0 μM. 1 [S] , мм -1 1 V₁ s μM 1 100.0 0.100 75.0 0.080 50.0 0.060 15.0 0.030 10.0 0.025 5.0 0.020 Answer: ||| Гarrow_forward1:33 5G. 46% Problem 12 of 15 Submit Using the following reaction data points, construct a Lineweaver-Burk plot for an enzyme with and without an uncompetitive inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. 1 -1 1 mM [S]' 20 V' s mM¹ with 10 μg per 54 10 36 > ст 5 27 2.5 23 1.25 20 Answer: |||arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College




Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning

Anatomy & Physiology
Biology
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:OpenStax College

Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license