
General, Organic, & Biological Chemistry
3rd Edition
ISBN: 9780073511245
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16.84P
Answer each question about phenylacetaldehyde, depicted in the ball-and-stick model, and Page 590 one of the components responsible for the fragrance of the plumeria flower.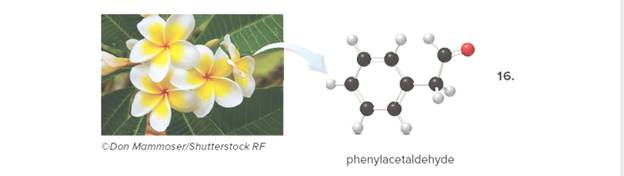
a. Draw a skeletal structure for phenylacetaldehyde.
b. How many trigonal planar carbons are present in phenylacetaldehyde?
c. What product is formed when phenylacetaldehyde is treated with two equivalents of
d. What product is formed when phenylacetaldehyde is treated with one equivalent of hydrogen gas in the presence of a metal catalyst?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following is 3-ethyl-2-methylpentane?
х
Can you please help me with this problem and explain it step by step? I'm so confused about it
2. Identify the reagents you would need to achieve the following. You may need to consider
using a protecting group.
HO
1.
2.
3.
4.
5.
OH
Br
HO
Chapter 16 Solutions
General, Organic, & Biological Chemistry
Ch. 16.1 - Prob. 16.1PCh. 16.1 - Draw the structure of the three constitutional...Ch. 16.2 - Prob. 16.3PCh. 16.2 - Give the structure corresponding to each TUPAC...Ch. 16.2 - Prob. 16.5PCh. 16.2 - Prob. 16.6PCh. 16.2 - Give the structure corresponding to each name. a....Ch. 16.3 - Which compound in each pair has the higher boiling...Ch. 16.3 - Acetone and progesterone are two ketones that...Ch. 16.4 - Prob. 16.10P
Ch. 16.5 - What product is formed when each carbonyl compound...Ch. 16.5 - Prob. 16.12PCh. 16.5 - Prob. 16.13PCh. 16.6 - What alcohol is formed when each compound is...Ch. 16.6 - Prob. 16.15PCh. 16.6 - Prob. 16.16PCh. 16.7 - Prob. 16.17PCh. 16.7 - Prob. 16.18PCh. 16.8 - Prob. 16.19PCh. 16.8 - Prob. 16.20PCh. 16.8 - Prob. 16.21PCh. 16.8 - Label the three acetals in solanine, the toxic...Ch. 16.8 - Prob. 16.23PCh. 16.8 - Prob. 16.24PCh. 16 - Prob. 16.25PCh. 16 - Prob. 16.26PCh. 16 - Prob. 16.27PCh. 16 - Prob. 16.28PCh. 16 - Prob. 16.29PCh. 16 - Prob. 16.30PCh. 16 - Prob. 16.31PCh. 16 - Prob. 16.32PCh. 16 - Prob. 16.33PCh. 16 - Prob. 16.34PCh. 16 - Give an acceptable name for each ketone. a. b. c....Ch. 16 - Prob. 16.36PCh. 16 - Prob. 16.37PCh. 16 - Draw the structure corresponding to each name. a....Ch. 16 - Prob. 16.39PCh. 16 - Prob. 16.40PCh. 16 - Prob. 16.41PCh. 16 - Prob. 16.42PCh. 16 - Draw out the structure of benzaldehyde, including...Ch. 16 - Prob. 16.44PCh. 16 - Which compound in each pair has the higher boiling...Ch. 16 - Prob. 16.46PCh. 16 - Prob. 16.47PCh. 16 - Prob. 16.48PCh. 16 - Prob. 16.49PCh. 16 - Prob. 16.50PCh. 16 - Prob. 16.51PCh. 16 - Prob. 16.52PCh. 16 - Prob. 16.53PCh. 16 - Prob. 16.54PCh. 16 - Prob. 16.55PCh. 16 - Consider the following ball-and-stick model....Ch. 16 - Prob. 16.57PCh. 16 - Prob. 16.58PCh. 16 - Prob. 16.59PCh. 16 - Prob. 16.60PCh. 16 - Prob. 16.61PCh. 16 - Prob. 16.62PCh. 16 - Prob. 16.63PCh. 16 - Prob. 16.64PCh. 16 - Prob. 16.65PCh. 16 - Prob. 16.66PCh. 16 - Prob. 16.67PCh. 16 - Prob. 16.68PCh. 16 - Prob. 16.69PCh. 16 - Label the functional group(s) in each compound as...Ch. 16 - Prob. 16.71PCh. 16 - Prob. 16.72PCh. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - Prob. 16.75PCh. 16 - Prob. 16.76PCh. 16 - Prob. 16.77PCh. 16 - Prob. 16.78PCh. 16 - Prob. 16.79PCh. 16 - Prob. 16.80PCh. 16 - Prob. 16.81PCh. 16 - Prob. 16.82PCh. 16 - Prob. 16.83PCh. 16 - Answer each question about phenylacetaldehyde,...Ch. 16 - Prob. 16.85PCh. 16 - Prob. 16.86PCh. 16 - Prob. 16.87PCh. 16 - Prob. 16.88PCh. 16 - Three constitutional isomers of molecular formula...Ch. 16 - Identify A—C in the following reaction sequenceCh. 16 - Androsterone is a male sex hormone that controls...Ch. 16 - Prob. 16.92PCh. 16 - Prob. 16.93PCh. 16 - Prob. 16.94PCh. 16 - Prob. 16.95PCh. 16 - Paraldehyde, a hypnotic and sedative once commonly...Ch. 16 - Prob. 16.97PCh. 16 - Prob. 16.98PCh. 16 - Prob. 16.99CPCh. 16 - Prob. 16.100CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- BeF2 exists as a linear molecule. Which kind of hybrid orbitals does Be use in this compound? Use Orbital Diagrams to show how the orbitals are formed. (6)arrow_forwardPlease answer the questions and provide detailed explanations as well as a drawing to show the signals in the molecule.arrow_forwardPropose an efficient synthesis for the following transformation: EN The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagents in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A. t-BuOK B. Na2Cr2O7, H2SO4, H2O C. NBS, heat F. NaCN D. MeOH E. NaOH G. MeONa H. H2O I. 1) O3; 2) DMSarrow_forward
- Stereochemistry Identifying the enantiomer of a simple organic molecule 1/5 Check the box under each structure in the table that is an enantiomer of the molecule shown below. If none of them are, check the none of t above box under the table. Br ま HO H 0 Molecule 1 Molecule 2 Molecule 3 OH H Br H H" Br OH Br Molecule 4 Br H OH + + OH Molecule 5 Br H OH none of the above Molecule 6 Br H... OHarrow_forwardPlease answer the questions and provide detailed explanations.arrow_forwardQuestion 16 0/1 pts Choose the correct option for the following cycloaddition reaction. C CF3 CF3 CF3 CF3 The reaction is suprafacial/surafacial and forbidden The reaction is antarafacial/antarafacial and forbidden The reaction is antarafacial/antarafacial and allowed The reaction is suprafacial/surafacial and allowedarrow_forward
- 1. Give the structures of the products obtained when the following are heated. Include stereochemistry where relevant. A NO2 + NO2 B + C N=C CEN + { 2. Which compounds would you heat together in order to synthesize the following?arrow_forwardExplain how myo-inositol is different from D-chiro-inositol. use scholarly sources and please hyperlink.arrow_forwardWhat is the molarisuty of a 0.396 m glucose solution if its density is 1.16 g/mL? MM glucose 180.2 /mol.arrow_forward
- Provide the proper IUPAC or common name for the following compound. Dashes, commas, and spaces must be used correctly. Br ......Im OHarrow_forwardCan you please help me solve this problems. The top one is just drawing out the skeletal correct and then the bottom one is just very confusing to me and its quite small in the images. Can you enlarge it and explain it to me please. Thank You much (ME EX1) Prblm #33arrow_forwardI'm trying to memorize VESPR Shapes to solve problems like those. I need help making circles like the second image in blue or using an x- and y-axis plane to memorize these and solve those types of problems, especially the ones given in the top/first image (180, 120, 109.5). Can you help me with this? or is their any other efficient method do soarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY