
Concept explainers
(a)
Interpretation:
The structure of the
Concept Introduction:
An aldehyde refers to a chemical compound which contains -CHO
Answer to Problem 16.25P
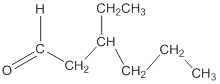
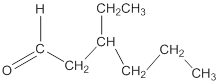
Explanation of Solution
To draw the structure of an aldehyde having the molecular formula
Thus, the structure of the aldehyde can be represented as follows:
(b)
Interpretation:
The structure of the
Concept Introduction:
In chemistry a ketone functional group refers to the structure with general molecular formula
Answer to Problem 16.25P
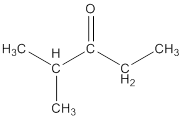
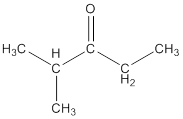
Explanation of Solution
To draw the structure of ketone having molecular formula
The structure so formed is shown below:
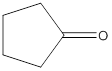
(c)
Interpretation:
The structure of ketone having the molecular formula
Concept Introduction:
In chemistry a ketone functional group refers to the structure with general molecular formula
Answer to Problem 16.25P
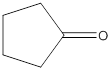
Explanation of Solution
To draw the structure of ketone having the molecular formula
(d)
Interpretation:
The structure of an aldehyde with the molecular formula
Concept Introduction:
An aldehyde refers to a chemical compound which contains -CHO functional group that consists of a carbonyl center (carbon -oxygen double bond) provided that the carbon atom is attached to hydrogen and R group of the carbon chain. A molecular formula depicts the chemical symbols for the elements constituting the given molecule such that each symbol contains a numerical subscript that describes the number of atoms of each atom present in the molecule.
Answer to Problem 16.25P

Explanation of Solution
To draw the structure of an aldehyde with the molecular formula

Want to see more full solutions like this?
Chapter 16 Solutions
General, Organic, & Biological Chemistry
- Show the mechanism steps to obtain the lowerenergy intermediate: *see imagearrow_forwardSoap is made by the previous reaction *see image. The main difference between one soap and another soap isthe length (number of carbons) of the carboxylic acid. However, if a soap irritates your skin, they mostlikely used too much lye.Detergents have the same chemical structure as soaps except for the functional group. Detergentshave sulfate (R-SO4H) and phosphate (R-PO4H2) functional groups. Draw the above carboxylic acidcarbon chain but as the two variants of detergents. *see imagearrow_forwardWhat are the reactions or reagents used? *see imagearrow_forward
- The two pKa values of oxalic acid are 1.25 and 3.81. Why are they not the same value? Show the protontransfer as part of your explanation. *see imagearrow_forwardасть Identify all the bonds that gauche interact with C-OMe in the most stable conformation of the above compound.arrow_forwardPredict the reactants used in the formation of the following compounds using Acid-Catalyzed dehydration reactionarrow_forward
- Can I please get help with this?arrow_forward.. Give the major organic product(s) for each of the following reactions or sequences of reactions. Show ll relevant stereochemistry [3 ONLY]. A H Br 1. NaCN 2 NaOH, H₂O, heat 3. H3O+ B. CH₂COOH 19000 1. LiAlH4 THF, heat 2 H₂O* C. CH Br 1. NaCN, acetone 2 H3O+, heat D. Br 1. Mg. ether 3. H₂O+ 2 CO₂ E. CN 1. (CH) CHMgBr, ether 2 H₂O+arrow_forwardAssign this COSY spectrumarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning  Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax




