
(a)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
(1) The six-membered aromatic ring is named as benzene.
(2) If a
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 1, the benzene ring contains chloro and the ethyl group. The numbering starts from the carbon atom containing chloro group since it comes first in the alphabetical series and the numbering is done according to lowest locant rule.
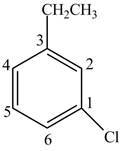
Figure 1
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
(b)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 2, the benzene ring contains two ethyl groups. So, numbering can be done from any carbon containing ethyl group using lowest locant rule.
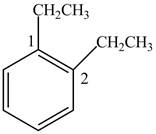
Figure 2
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
(c)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 3, the benzene ring contains nitro and the vinyl group. The numbering starts from the carbon atom containing nitrro group since it comes first in the alphabetical series and the numbering is done according to lowest locant rule.
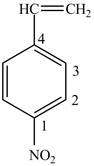
Figure 3
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
(d)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 4, the benzene ring contains two chloro groups and one functional group, hydroxyl group. The name of the benzene ring will become phenyl. The numbering starts from the carbon atom containing hydroxyl group since it is a functional group and then the substituents are numbered using lowest locant rule.
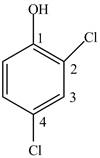
Figure 4
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
(e)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
In Figure 5, the benzene ring contains chloro, bromo, fluoro, iodo and nitro groups. The numbering is done using the lowest locant rule.
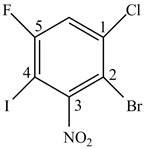
Figure 5
The rule of naming an aromatic compound according to IUPAC is
The IUPAC name of the aromatic compound is
(f)
Interpretation:
The name of the benzene derivative compound is to be stated.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
In aryl compounds, the substituents are indicated by the numbers and the aromatic ring is named as benzene.
Answer to Problem 16.1P
The IUPAC name of the aromatic compound is
Explanation of Solution
Aromatic compounds are those compounds which contains at least one aromatic ring. The naming of aromatic compounds uses the same rules as aliphatic compounds.
(1) The six-membered aromatic ring is named as benzene.
(2) If a functional group is present on the benzene ring, then the benzene is named as phenyl.
(3) The substituents on the benzene ring are indicated by the numbers and are named in alphabetical order.
(4) The lowest locant rule is used. The substituent which comes first in the alphabetical order is numbered one and then the numbering is done in the direction such that the other substituent gets the maximum lower numerical value.
(5) If a functional group is attached on the benzene ring, then that carbon is numbered one.
(6) If benzene ring is a substitutent, it is named as phenyl.
In Figure 6, the benzene rings are subtituents on the methyl group.
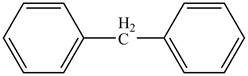
Figure 6
The rule of naming an aromatic compound according to IUPAC is
Therefore, the IUPAC name of the aromatic compound is
The IUPAC name of the aromatic compound is
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry Study Guide and Solutions
- please help fill in the tablearrow_forwardAnswer F pleasearrow_forward4. Refer to the data below to answer the following questions: The octapeptide saralasin is a specific antagonist of angiotensin II. A derivative of saralasin is used therapeutically as an antihypertensive. Amino acid analysis of saralasin show the presence of the following amino acids: Ala, Arg, His, Pro, Sar, Tyr, Val, Val A.Sar is the abbreviation for sarcosine, N-methyl aminoethanoic acid. Draw the structure of sarcosine. B. N-Terminal analysis by the Edman method shows saralasin contains sarcosine at the N-terminus. Partial hydrolysis of saralasin with dilute hydrochloric acid yields the following fragments: Tyr-Val-His Sar-Arg-Val His-Pro-Ala Val-Tyr-Val Arg-Val-Tyr What is the structure of saralasin?arrow_forward
- What is the structure of the DNA backbone?arrow_forwardPLEASE PLEASE PLEASE use hand drawn structures when possarrow_forward. M 1- MATCH each of the following terms to a structure from the list below. There is only one correct structure for each term and structures may be used more than once. Place the letter of the structure in the blank to the left of the corresponding term. A. Sanger dideoxy method C. Watson-Crick B. GAUCGUAAA D. translation E. HOH2C OH OH G. transcription I. AUGGCUGAG 0 K. OPOH2C 0- OH N- H NH2 F. -OPOH2C 0- OH OH H. Maxam-Gilbert method J. replication N L. HOH2C a. b. C. d. e. f. g. B M. AGATCGCTC a pyrimidine nucleoside RNA base sequence with guanine at the 3' end. DNA base sequence with cytosine at the 3' end. a purine nucleoside DNA sequencing method for the human genome 2'-deoxyadenosine 5'-phosphate process by which mRNA directs protein synthesis OH NH2arrow_forward
- Please use hand drawn structures when neededarrow_forwardB. Classify the following amino acid. Atoms other than carbon and hydrogen are labeled. a. acidic b. basic C. neutral C. Consider the following image. Which level of protein structure is shown here? a. primary b. secondary c. tertiary d. quaternary D. Consider the following image. H RH H HR H R HR HR RH Which level of protein structure is shown in the box? a. primary b. secondary R c. tertiary d. quaternary コー Rarrow_forwardBriefly answer three from the followings: a. What are the four structures of the protein? b. Why is the side chain (R) attached to the alpha carbon in the amino acids is important for the function? c. What are the types of amino acids? And how is it depend on the (R) structure? d. Write a reaction to prepare an amino acid. prodarrow_forward
- Answe Answer A and B pleasearrow_forward3. Refer to the data below to answer the following questions: Isoelectric point Amino Acid Arginine 10.76 Glutamic Acid 3.22 Tryptophan 5.89 A. Define isoelectric point. B. The most basic amino acid is C. The most acidic amino acid is sidizo zoarrow_forward3. A gas mixture contains 50 mol% H2 and 50 mol% He. 1.00-L samples of this gas mixture are mixed with variable volumes of O2 (at 0 °C and 1 atm). A spark is introduced to allow the mixture to undergo complete combustion. The final volume is measured at 0 °C and 1 atm. Which graph best depicts the final volume as a function of the volume of added O2? (A) 2.00 1.75 Final Volume, L 1.50 1.25 1.00 0.75 0.50 0.25 0.00 0.00 0.25 0.50 2.00 (B) 1.75 1.50 Final Volume, L 1.25 1.00 0.75 0.50- 0.25 0.00 0.75 1.00 0.00 0.25 Volume O₂ added, L 2 0.50 0.75 1.00 Volume O₂ added, L 2 2.00 2.00 (C) (D) 1.75 1.75 1.50 1.50 Final Volume, L 1.25 1.00 0.75 0.50 Final Volume, L 1.25 1.00 0.75 0.50 0.25 0.25 0.00 0.00 0.00 0.25 0.50 0.75 1.00 0.00 0.25 Volume O₂ added, L 0.50 0.75 1.00 Volume O₂ added, L 2arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





