
Concept explainers
(a)
Interpretation:
The electrons in energy level are to be written and electron dot structure is to be drawn.
Concept introduction:
Electrons in energy level are calculated by writing electronic configuration.
Electron dot structure is drawn by writing the symbol of the structure and putting the dots on the symbol as there are valence electrons in that element.
Answer to Problem 110A
Electronic configuration of Kr is 2, 8, 18, 8
Electron dot structure:
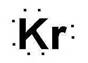
Explanation of Solution
Electronic configuration helps to draw electron dot structure. The number of dots drawn is equal to number of electrons in the valance shell.
(b)
Interpretation:
The electrons in energy level are to be written and electron dot structure is to be drawn.
Concept introduction:
Electrons in energy level are calculated by writing electronic configuration.
Electron dot structure is drawn by writing the symbol of the structure and putting the dots on the symbol as there are valence electrons in that element.
(b)
Answer to Problem 110A
Electronic configuration of Sr is 2, 8, 18, 8, 2
Electron dot structure:
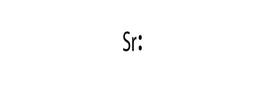
Explanation of Solution
Electronic configuration helps to draw electron dot structure. The number of dots drawn is equal to number of electrons in the valance shell.
(c)
Interpretation:
The electrons in energy level are to be written and electron dot structure is to be drawn.
Concept introduction:
Electrons in energy level are calculated by writing electronic configuration.
Electron dot structure is drawn by writing the symbol of the structure and putting the dots on the symbol as there are valence electrons in that element.
(c)
Answer to Problem 110A
Electronic configuration of P is 2, 8, 5
Electron dot structure:
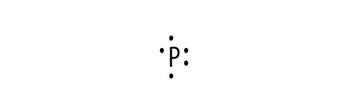
Explanation of Solution
Electronic configuration helps to draw electron dot structure. The number of dots drawn is equal to number of electrons in the valance shell.
(d)
Interpretation:
The electrons in energy level are to be written and electron dot structure is to be drawn.
Concept introduction:
Electrons in energy level are calculated by writing electronic configuration.
Electron dot structure is drawn by writing the symbol of the structure and putting the dots on the symbol as there are valence electrons in that element.
(d)
Answer to Problem 110A
Electronic configuration of B is 2, 3
Electron dot structure:
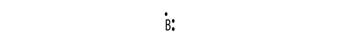
Explanation of Solution
Electronic configuration helps to draw electron dot structure. The number of dots drawn is equal to number of electrons in the valance shell.
( e)
Interpretation:
The electrons in energy level are to be written and electron dot structure is to be drawn.
Concept introduction:
Electrons in energy level are calculated by writing electronic configuration.
Electron dot structure is drawn by writing the symbol of the structure and putting the dots on the symbol as there are valence electrons in that element.
( e)
Answer to Problem 110A
Electronic configuration of Br is 2, 8, 18, 7
Electron dot structure:

Explanation of Solution
Electronic configuration helps to draw electron dot structure. The number of dots drawn is equal to number of electrons in the valance shell.
( f)
Interpretation:
The electrons in energy level are to be written and electron dot structure is to be drawn.
Concept introduction:
Electrons in energy level are calculated by writing electronic configuration.
Electron dot structure is drawn by writing the symbol of the structure and putting the dots on the symbol as there are valence electrons in that element.
( f)
Answer to Problem 110A
Electronic configuration of Se is 2, 8, 18, 6
Electron dot structure:
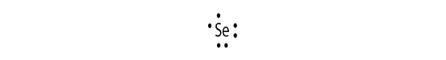
Explanation of Solution
Electronic configuration helps to draw electron dot structure. The number of dots drawn is equal to number of electrons in the valance shell.
Chapter 13 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Campbell Biology: Concepts & Connections (9th Edition)
Cosmic Perspective Fundamentals
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Human Physiology: An Integrated Approach (8th Edition)
Campbell Biology in Focus (2nd Edition)
Introductory Chemistry (6th Edition)
- A. B. b. Now consider the two bicyclic molecules A. and B. Note that A. is a dianion and B. is a neutral molecule. One of these molecules is a highly reactive compound first characterized in frozen noble gas matrices, that self-reacts rapidly at temperatures above liquid nitrogen temperature. The other compound was isolated at room temperature in the early 1960s, and is a stable ligand used in organometallic chemistry. Which molecule is the more stable molecule, and why?arrow_forwardWhere are the chiral centers in this molecule? Also is this compound meso yes or no?arrow_forwardPLEASE HELP! URGENT!arrow_forward
- Where are the chiral centers in this molecule? Also is this compound meso yes or no?arrow_forwardA mixture of C7H12O2, C9H9OCl, biphenyl and acetone was put together in a gas chromatography tube. Please decide from the GC resutls which correspond to the peak for C7,C9 and biphenyl and explain the reasoning based on GC results. Eliminate unnecessary peaks from Gas Chromatography results.arrow_forwardIs the molecule chiral, meso, or achiral? CI .CH3 H₂C CIarrow_forward
- A mixture of three compounds Phen-A, Acet-B and Rin-C was analyzed using TLC with 1:9 ethanol: hexane as the mobile phase. The TLC plate showed three spots of R, 0.1 and 0.2 and 0.3. Which of the three compounds (Phen-A; Acet-B or Rin-C) would have the highest (Blank 1), middle (Blank 2) and lowest (Blank 3) spot respectively? 0 CH: 0 CH, 0 H.C OH H.CN OH Acet-B Rin-C phen-A A A <arrow_forwardHow many chiral carbons are in the molecule? Farrow_forwardcan someone give the curly arrow mechanism for this reaction written with every intermediate and all the side products pleasearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





