
Concept explainers
12-42 Complete these equations.
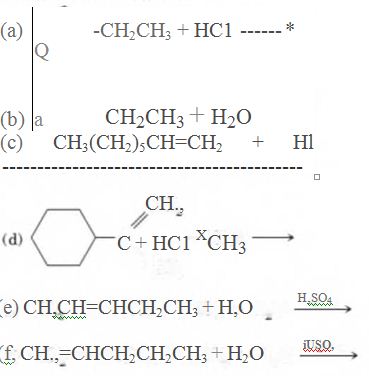
(a)
Interpretation:
Complete the below equation:
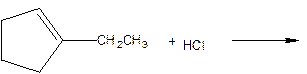
Concept introduction:
During the hydration of alkene the unsaturated alkene converted into saturated alkane with the addition of H+ and Cl- ions. This reaction is carried out in the presence of acid therefore it is called acid catalyzed hydration of alkene.
Answer to Problem 32P
Explanation of Solution
As per the given equation, the reactant is a cyclic alkene to which HCl is added. And the characteristics reactions given by the alkenes are addition reactions in which the addition takes place at the double bond. The addition follows the Markovnikov’s rule.
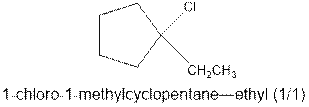
As per the Markovnikov’s rule, the hydrogen acid is added to the carbon of a double bond containing higher number of hydrogens in it. The halogen part is added to the carbon in the double bond containing fewer number of hydrogens in it. Here, the positive part is H+ and the halogen part is Cl-. On numbering the carbon atoms, the structure is.
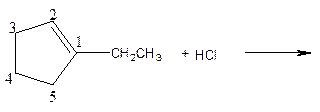
Now as per the rule, the hydrogen in the above equation attaches to carbon 2 as carbon 2 of the double bond has one hydrogen attached to it. The Cl- will attached to carbon 1 as it has no carbon attached to it. Thus, the complete reaction is:
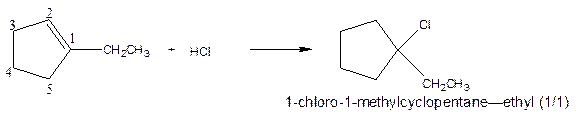
(b)
Interpretation:
Complete the below equation:
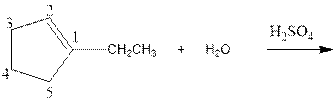
Concept introduction:
During the hydration of alkene the unsaturated alkene converted into saturated alkane with the addition of H+ and OH- ions in the presence of H2 SO4. This reaction is carried out in the presence of acid therefore it is called acid catalyzed hydration of alkene.
Answer to Problem 32P
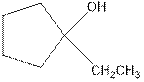
Explanation of Solution
As per the given equation, the reactant is a cyclic alkene to which H2 O is added. And the characteristics reactions given by the alkenes are addition reactions in which the addition takes place at the double bond. The addition follows the Markovnikov’s rule.
As per the Markovnikov’s rule, the hydrogen acid is added to the carbon of a double bond containing higher number of hydrogens in it. The halogen part is added to the carbon in the double bond containing fewer number of hydrogens in it. Here, the positive part is H+ and the halogen part is OH-. On numbering the carbon atoms, the structure is.
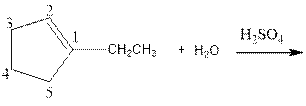
Now as per the rule, the hydrogen in the above equation attaches to carbon 2 as carbon 2 of the double bond has one hydrogen attached to it. The OH- will attached to carbon 1 as it has no carbon attached to it. Thus, the complete reaction is:
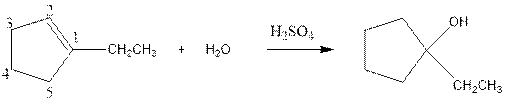
(c)
Interpretation:
Complete the below equation:
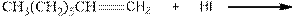
Concept introduction:
During the hydration of alkene the unsaturated alkene converted into saturated alkane with the addition of H+ and I- ions.
Answer to Problem 32P
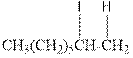
Explanation of Solution
As per the above equation, the reactant is an alkene to which HI is added. We also know that the characteristics reactions given by alkenes are addition reactions in which the addition takes place at the double bond. This addition follows the Markovnikov’s rule.
As per the Markovnikov’s rule, the hydrogen acid is added to the carbon of a double bond containing higher number of hydrogens in it. The halogen part is added to the carbon in the double bond containing fewer number of hydrogens in it. Here, the positive part is H+ and the halogen part is I-. On numbering the double bonded carbons of the alkene, the structure is.
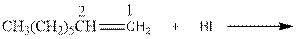
Now as per the rule, the hydrogen in the above equation attaches to carbon 1 as carbon 1 of the double bond has two hydrogen attached to it. The I- will attached to carbon 2 as it has only one carbon attached to it. Thus, the complete reaction is:
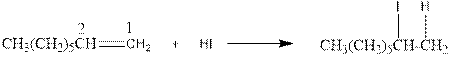
(d)
Interpretation:
Complete the below equation:

Concept introduction:
As per the Markovnikov’s rule, the hydrogen acid is added to the carbon of a double bond containing higher number of hydrogens in it. The halogen part is added to the carbon in the double bond containing fewer number of hydrogens in it.
Answer to Problem 32P
Explanation of Solution
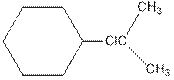
As per the above equation, the reactant is an alkene to which HCl is added. We also know that the characteristics reactions given by alkenes are addition reactions in which the addition takes place at the double bond. This addition follows the Markovnikov’s rule.
As per the Markovnikov’s rule, the hydrogen acid is added to the carbon of a double bond containing higher number of hydrogens in it. The halogen part is added to the carbon in the double bond containing fewer number of hydrogens in it. Here, the positive part is H+ and the halogen part is Cl-. On numbering the double bonded carbons of the alkene, the structure is.
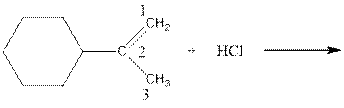
Now as per the rule, the hydrogen in the above equation attaches to carbon 1 as carbon 1 of the double bond has two hydrogen attached to it. The Cl- will attached to carbon 2 as it has only no carbon attached to it. Thus, the complete reaction is:

(e)
Interpretation:
Complete the below equation:
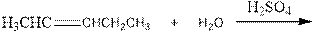
Concept introduction:
During the hydration of alkene the unsaturated alkene converted into saturated alkane with the addition of H+ and OH- ions in the presence of H2 SO4. This reaction is carried out in the presence of acid therefore it is called acid catalyzed hydration of alkene.
Answer to Problem 32P
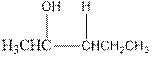
Explanation of Solution
As per the above equation, the reactant is an alkene to which H2 O is added. We also know that the characteristics reactions given by alkenes are addition reactions in which the addition takes place at the double bond. This addition follows the Markovnikov’s rule.
As per the Markovnikov’s rule, the hydrogen acid is added to the carbon of a double bond containing higher number of hydrogens in it. The halogen part is added to the carbon in the double bond containing fewer number of hydrogens in it. Here, the positive part is H+ and the halogen part is OH-. On numbering the alkene, the structure is.
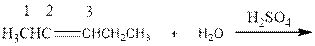
Now the below product will form where hydrogen will attach to the 2nd carbon to form more suitable product:
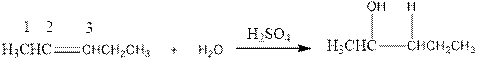
(f)
Interpretation:
Complete the below equation:
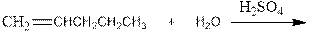
Concept introduction:
During the hydration of alkene the unsaturated alkene converted into saturated alkane with the addition of H+ and OH- ions in the presence of H2 SO4. This reaction is carried out in the presence of acid therefore it is called acid catalyzed hydration of alkene.
Answer to Problem 32P
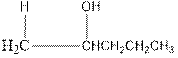
Explanation of Solution
As per the above equation, the reactant is an alkene to which H2 O is added. We also know that the characteristics reactions given by alkenes are addition reactions in which the addition takes place at the double bond. This addition follows the Markovnikov’s rule.
As per the Markovnikov’s rule, the hydrogen acid is added to the carbon of a double bond containing higher number of hydrogens in it. The halogen part is added to the carbon in the double bond containing fewer number of hydrogens in it. Here, the positive part is H+ and the halogen part is OH-. On numbering the alkene, the structure is.
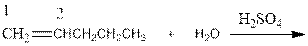
Now as per the rule, the hydrogen in the above equation attaches to carbon 1 as carbon 1 of the double bond has two hydrogen attached to it. The OH- will attached to carbon 2 as it has only one carbon attached to it. Thus, the complete reaction is:
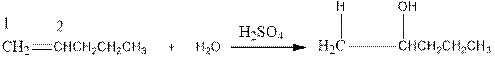
Want to see more full solutions like this?
Chapter 12 Solutions
INTRO.TO GENERAL,ORGAN...-OWLV2 ACCESS
- A pdf file of your hand drawn, stepwise mechanisms for the reactions. For each reaction in the assignment, you must write each mechanism three times (there are 10 reactions, so 30 mechanisms). (A) do the work on a tablet and save as a pdf., it is expected to write each mechanism out and NOT copy and paste the mechanism after writing it just once. Everything should be drawn out stepwise and every bond that is formed and broken in the process of the reaction, and is expected to see all relevant lone pair electrons and curved arrows. Aldol: NaOH HO H Δ NaOH Δarrow_forwardNonearrow_forwardDraw structures corresponding to the following names and give IUPAC names for the following compounds: (8 Point) a) b) c) CH3 CH2CH3 CH3CHCH2CH2CH CH3 C=C H3C H H2C=C=CHCH3 d) CI e) (3E,5Z)-2,6-Dimethyl-1,3,5,7-octatetraene f) (Z)-4-bromo-3-methyl-3-penten-1-yne g) cis-1-Bromo-2-ethylcyclopentane h) (5R)-4,4,5-trichloro-3,3-dimethyldecanearrow_forward
- Draw a Newman projection from carbon 3 to carbon 2 in the highest energy conformation for the following molecule. What is this conformation called? What kind of strain is present? Brarrow_forwardWhich of the following dienophiles is most reactive in a Diels-Alder reaction: Please explain why the correct answer to this question is option 5. Please provide a detailed explanation.arrow_forwardWhich of the following would you expect to be aromatic? Please provide a detailed explanation.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co



