
(a)
Interpretation:
The order of the reaction with respect to substance
(a)
Explanation of Solution
The rate law can be represented as given below.
Where,
Carefully examining the table, it can be said that
By taking logarithm on both the sides, it can be written as follows,
The above equation is in the form of
Similarly, by keeping
By taking logarithm on both the sides, it can be written as follows,
The above equation is in the form of
Four data sets have constant
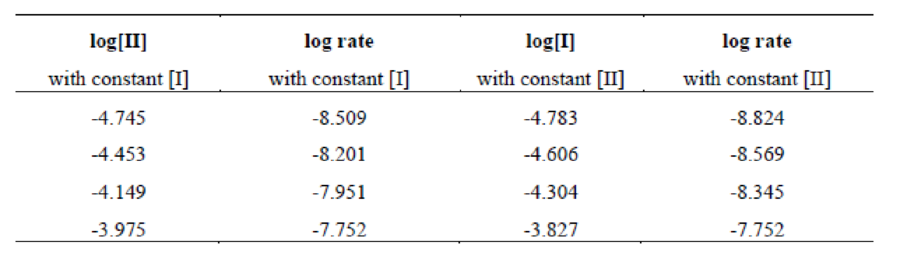
Figure 1
The graph of
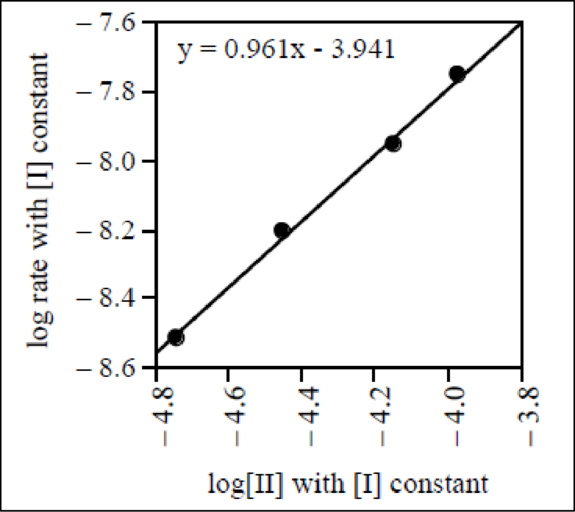
Figure 2
Therefore, the reaction order of reactant
The graph of
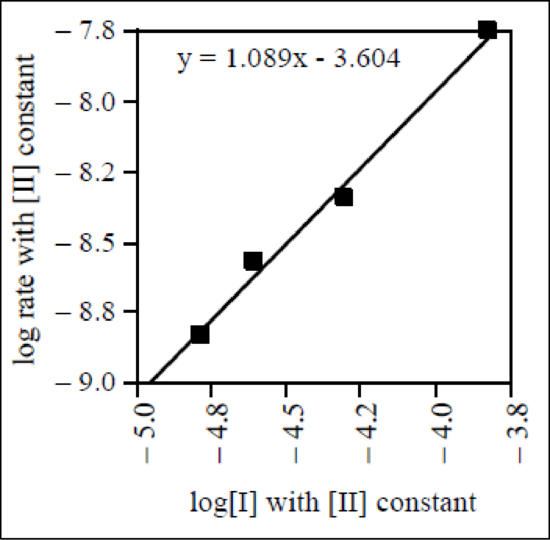
Figure 3
Therefore, the reaction order of reactant
(b)
Interpretation:
The rate law for the reaction has to be derived.
(b)
Explanation of Solution
The order of the reaction with respect to substance
(c)
Interpretation:
The rate constant
(c)
Answer to Problem 25QRT
The average of the rate constant is
Explanation of Solution
The rate constant can be expressed as shown below.
For first experiment the rate constant can be calculated by plugging all the data given in the table.
In first experiment, the initial concentration of
Now, the rate constant for first experiment is given below.
Similarly, the rate constant for rest of the experiments can be calculated. The calculated rate constant values are given in the table below.
|
Initial rate |
Rate constant | ||
The average of the rate constant is
(d)
Interpretation:
The initial rate of the reaction has to be calculated.
(d)
Answer to Problem 25QRT
The initial
Explanation of Solution
The rate law can be expressed as shown below.
Given that,
Therefore, the initial rate of reaction is
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry: The Molecular Science
- Indicate the products obtained by mixing 2,2-dimethylpropanal with acetaldehyde and sodium ethoxide in ethanol.arrow_forwardSynthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIf possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forward
- Synthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forward
- Indicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

