
EBK ORGANIC CHEMISTRY
8th Edition
ISBN: 8220102744127
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 95P
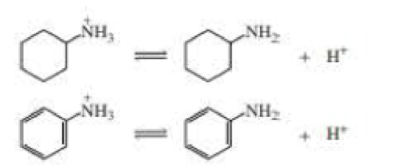
Protonated cyclohexylamine has a Ka = 1 × 10–11 Using the same sequence of steps as in Problem 94, determine which is a stronger base: cyclohexylamine or aniline.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Briefly describe the compounds called carboranes.
Please don't use Ai solution
None
Chapter 8 Solutions
EBK ORGANIC CHEMISTRY
Ch. 8.1 - Prob. 1PCh. 8.1 - Prob. 2PCh. 8.4 - Prob. 3PCh. 8.5 - Prob. 4PCh. 8.5 - Prob. 6PCh. 8.6 - a. Predict the relative bond lengths of the three...Ch. 8.6 - Prob. 8PCh. 8.6 - Prob. 9PCh. 8.6 - Prob. 10PCh. 8.7 - Prob. 11P
Ch. 8.7 - Prob. 12PCh. 8.7 - Prob. 13PCh. 8.8 - Prob. 14PCh. 8.8 - Prob. 15PCh. 8.8 - Prob. 16PCh. 8.9 - Which member of each pair is the stronger acid?Ch. 8.9 - Which member of each pair is the stronger base? a....Ch. 8.9 - Rank the following compounds from strongest acid...Ch. 8.10 - Prob. 20PCh. 8.10 - Which acid in each of the following pairs is...Ch. 8.10 - Prob. 23PCh. 8.11 - Prob. 24PCh. 8.11 - Prob. 26PCh. 8.12 - Prob. 27PCh. 8.12 - Prob. 28PCh. 8.12 - Prob. 29PCh. 8.12 - Prob. 30PCh. 8.12 - Prob. 31PCh. 8.12 - Prob. 32PCh. 8.13 - Prob. 33PCh. 8.13 - Prob. 34PCh. 8.13 - Prob. 35PCh. 8.13 - What are the major 1,2- and 1,4-addition products...Ch. 8.13 - Prob. 38PCh. 8.14 - Prob. 39PCh. 8.14 - Prob. 40PCh. 8.14 - Prob. 41PCh. 8.14 - Prob. 42PCh. 8.14 - Prob. 43PCh. 8.14 - Prob. 44PCh. 8.14 - Prob. 46PCh. 8.15 - Prob. 47PCh. 8.17 - Prob. 48PCh. 8.17 - Prob. 49PCh. 8.18 - Prob. 50PCh. 8.18 - Prob. 52PCh. 8.18 - Prob. 53PCh. 8.18 - Prob. 54PCh. 8.19 - Prob. 55PCh. 8.20 - Prob. 56PCh. 8.20 - What orbitals contain the electrons represented as...Ch. 8.20 - Prob. 59PCh. 8.20 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - Prob. 65PCh. 8 - Prob. 66PCh. 8 - Prob. 67PCh. 8 - Prob. 68PCh. 8 - Prob. 69PCh. 8 - Prob. 70PCh. 8 - Prob. 71PCh. 8 - Prob. 72PCh. 8 - Prob. 73PCh. 8 - Which compound is the strongest base?Ch. 8 - Prob. 75PCh. 8 - Prob. 76PCh. 8 - a. The A ring (Section 3.16) of cortisone (a...Ch. 8 - Prob. 78PCh. 8 - Prob. 79PCh. 8 - Prob. 80PCh. 8 - Prob. 81PCh. 8 - Purine is a heterocyclic compound with four...Ch. 8 - Prob. 83PCh. 8 - Why is the delocalization energy of pyrrole (21...Ch. 8 - Prob. 85PCh. 8 - Prob. 86PCh. 8 - Prob. 87PCh. 8 - A student obtained two products from the reaction...Ch. 8 - Prob. 89PCh. 8 - a. How could each of the following compounds be...Ch. 8 - Draw the products obtained from the reaction of...Ch. 8 - How would the following substituents affect the...Ch. 8 - Prob. 93PCh. 8 - The acid dissociation constant (Ka) for loss of a...Ch. 8 - Protonated cyclohexylamine has a Ka = 1 1011...Ch. 8 - Draw the product or products that would be...Ch. 8 - Prob. 97PCh. 8 - Prob. 98PCh. 8 - Prob. 99PCh. 8 - Prob. 100PCh. 8 - Prob. 101PCh. 8 - a. Propose n mechanism for the following reaction:...Ch. 8 - Prob. 103PCh. 8 - As many as 18 different Diels-Alder products can...Ch. 8 - Prob. 105PCh. 8 - Prob. 106PCh. 8 - Prob. 107PCh. 8 - Prob. 108PCh. 8 - The experiment shown next and discussed in Section...Ch. 8 - Prob. 110PCh. 8 - Prob. 111PCh. 8 - Prob. 112PCh. 8 - Prob. 1PCh. 8 - Prob. 2PCh. 8 - Prob. 3PCh. 8 - Prob. 4PCh. 8 - Prob. 5PCh. 8 - Prob. 6PCh. 8 - Prob. 7PCh. 8 - Prob. 8PCh. 8 - Prob. 9PCh. 8 - Prob. 10PCh. 8 - Prob. 11PCh. 8 - Prob. 12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonearrow_forwardJON Determine the bund energy for UCI (in kJ/mol Hcl) using me balanced chemical equation and bund energies listed? का (My (9) +36/2(g)-(((3(g) + 3(g) A Hryn = -330. KJ bond energy и-н 432 bond bond C-1413 C=C 839 N-H 391 C=O 1010 S-H 363 б-н 467 02 498 N-N 160 N=N 243 418 C-C 341 C-0 358 C=C C-C 339 N-Br 243 Br-Br C-Br 274 193 614 (-1 214||(=olin (02) 799 C=N 615 AALarrow_forwardDetermine the bond energy for HCI ( in kJ/mol HCI) using he balanced cremiculequecticnand bund energles listed? also c double bond to N is 615, read numbets carefully please!!!! Determine the bund energy for UCI (in kJ/mol cl) using me balanced chemical equation and bund energies listed? 51 (My (9) +312(g)-73(g) + 3(g) =-330. KJ спод bond energy Hryn H-H bond band 432 C-1 413 C=C 839 NH 391 C=O 1010 S-1 343 6-H 02 498 N-N 160 467 N=N C-C 341 CL- 243 418 339 N-Br 243 C-O 358 Br-Br C=C C-Br 274 193 614 (-1 216 (=olin (02) 799 C=N 618arrow_forward
- Differentiate between single links and multicenter links.arrow_forwardI need help on my practice final, if you could explain how to solve this that would be extremely helpful for my final thursday. Please dumb it down chemistry is not my strong suit. If you could offer strategies as well to make my life easier that would be beneficialarrow_forwardNonearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY