
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 7IQ
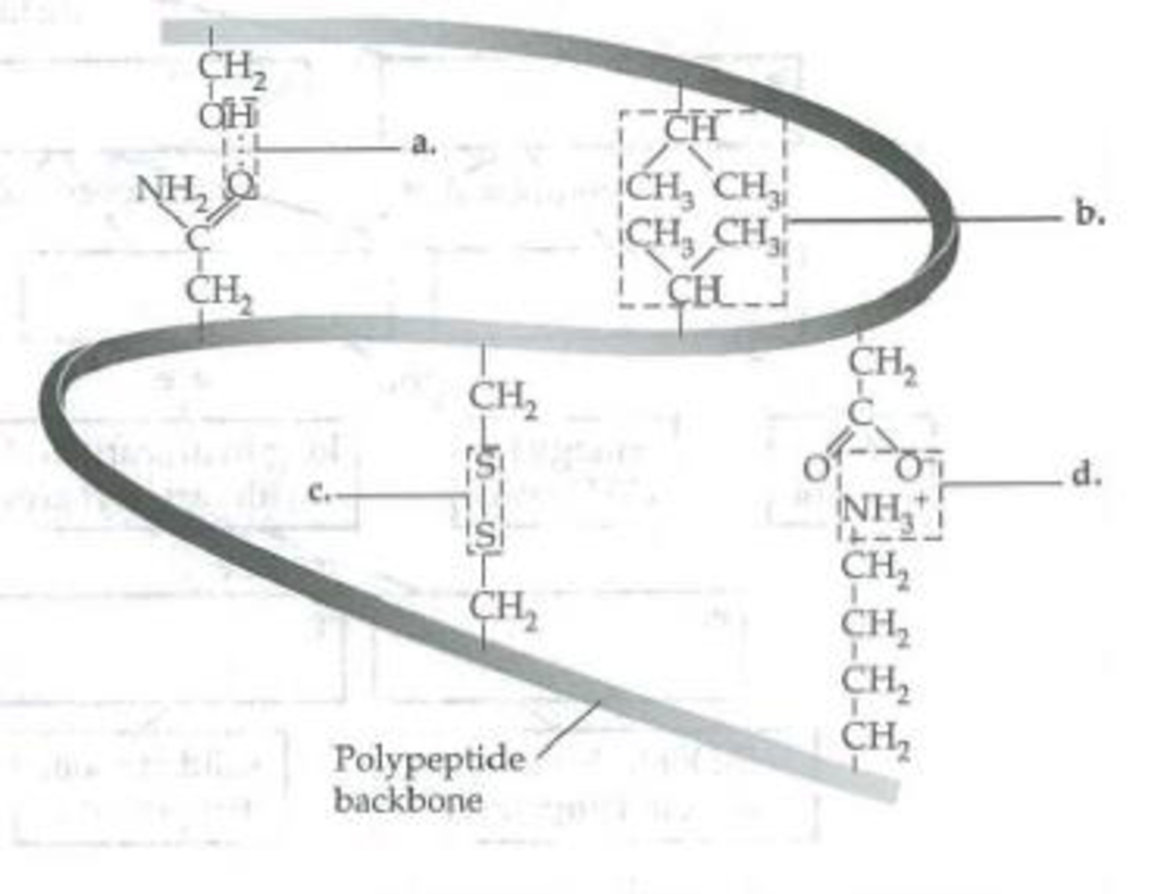
In the following diagram of a portion of a protein, label the types of interactions that are shown. What level of structure are these interactions producing?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
I have a question. I need to make 25 mL of this solution . How would I calculate the math? Please help
Introduction to blood lab report
Which of the structural components listed in the Essential terms of section 1.3 (Cell
components) could occur in a plant cell?
Paragraph
く
BIUA
川く
く
80
+
кл
K
Chapter 5 Solutions
Study Guide for Campbell Biology
Ch. 5 - Monomers are linked into polymers by ________...Ch. 5 - You can recognize a monosaccharide by its multiple...Ch. 5 - Number the carbons in the following glucose and...Ch. 5 - Prob. 4IQCh. 5 - Fill in this concept map to help you organize your...Ch. 5 - a. Draw the amino acids alanine (R group: CH3) and...Ch. 5 - In the following diagram of a portion of a...Ch. 5 - Now that you have gained experience with concept...Ch. 5 - a. Label the three parts of this nucleotide....Ch. 5 - Take the time to create a concept map that...
Ch. 5 - Prob. 1SYKCh. 5 - Prob. 2SYKCh. 5 - glycogen A. carbohydrate B. lipid C. protein D....Ch. 5 - cholesterol A. carbohydrate B. lipid C. protein D....Ch. 5 - RNA A. carbohydrate B. lipid C. protein D. nucleic...Ch. 5 - collagen A. carbohydrate B. lipid C. protein D....Ch. 5 - hemoglobin A. carbohydrate B. lipid C. protein D....Ch. 5 - A. carbohydrate B. lipid C. protein D. nucleic...Ch. 5 - Prob. 7TYKMCh. 5 - enzyme A. carbohydrate B. lipid C. protein D....Ch. 5 - cellulose A. carbohydrate B. lipid C. protein D....Ch. 5 - Chitin A. carbohydrate B. lipid C. protein D....Ch. 5 - Polymerization (the formation of polymers) is a...Ch. 5 - Which of the following statements is not true of a...Ch. 5 - Prob. 3TYKCh. 5 - Prob. 4TYKCh. 5 - Prob. 5TYKCh. 5 - Prob. 6TYKCh. 5 - A fatty acid that has the formula C16H32O2 is a....Ch. 5 - Prob. 8TYKCh. 5 - Prob. 9TYKCh. 5 - Prob. 10TYKCh. 5 - Which of the following molecules provides the most...Ch. 5 - Prob. 12TYKCh. 5 - What happens when a protein denatures? a. Its...Ch. 5 - The helix of proteins is a. part of a proteins...Ch. 5 - What is the best description of the following...Ch. 5 - Prob. 16TYKCh. 5 - Prob. 17TYKCh. 5 - Which of the following is true of the subunits of...Ch. 5 - Prob. 19TYKCh. 5 - If the nucleotide sequence of one strand of a DNA...Ch. 5 - How are nucleotide monomers connected to form a...Ch. 5 - Prob. 22TYK
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Which of the following statements refer(s) directly to the cell theory? (Note that one or more correct answers are possible.) Select 2 correct answer(s) a) There are major differences between plant and animal cells. b) There are major differences between prokaryote and eukaryote cells. c) All cells have a cell wall. d) All cells have a cell membrane. e) Animals are composed of cells. f) When a bacterial cell divides, it produces two daughter cells.arrow_forwardPreoperative Diagnosis: Torn medial meniscus, left knee Postoperative Diagnosis: Combination horizontal cleavage tear/flap tear, posterior horn, medial meniscus, left knee. Operation: Arthroscopic subtotal medial meniscectomy, left knee Anesthetic: General endotracheal Description of Procedure: The patient was placed on the operating table in the supine position and general endotracheal anesthesia was administered. After an adequate level of anesthesia was achieved, the patient's left lower extremity was prepped with Betadine scrubbing solution, then draped in a sterile manner. Several sites were then infiltrated with 1% Xylocaine solution with Epinephrine to help control bleeding from stab wounds to be made at these sites. These stab wounds were made anterolaterally at the level of the superior pole of the patella for insertion of an irrigation catheter into the suprapatellar pouch area, anterolaterally at the level of the joint line for insertion of the scope and anteromedially at…arrow_forwardUARDIAN SIGNA Life Sciences/ Baseline Test Grade 10 ry must be written in point form. pot in full sentences using NO MORE than 70 words sentences from 1 to 7. only ONE point per sentence. words as far as possible. number of words you have used in brackets at the end GDE/2024 QUESTION 3 The table below shows the results of an investigation in which the effect of temperature and light on the yield of tomatoes in two greenhouses on a farm was investigated. TEMPERATURE (°C) AVERAGE YIELD OF TOMATOES PER 3.1 PLANT (kg) LOW LIGHT LEVELS HIGH LIGHT LEVELS 5 0,5 0,5 10 1,5 2,5 15 3,0 5,0 20 3,6 8,5 25 3,5 7,8 30 2,5 6,2 State TWO steps the investigator may have taken into consideration during the planning stage of the investigation. (2) 3.2 Identify the: a) Independent variables (2) b) Dependent variable (1) 3.3 Plot a line graph showing the results of the average yield of the tomatoes from 5°C to 30°C for low light levels. (6) 3.4 State ONE way in which the scientists could have improved the…arrow_forward
- Explain why you chose this mutation. Begin by transcribing and translating BOTH the normal and abnormal DNA sequences. The genetic code below is for your reference. SECOND BASE OF CODON כ FIRST BASE OF CODON O THIRD BASE OF CODON SCAGUCAGUGAGUCAG UUU UUC UCU UAU UGU Phenylalanine (F) Tyrosine (Y) Cysteine (C) UCC UAC UGC Serine (S) UUA UUG Leucine (L) UCA UCG_ UAA UGA Stop codon -Stop codon UAG UGG -Tryptophan (W) CUU CUC CCU CAU CGU Histidine (H) CCC CAC CGC -Leucine (L) Proline (P) CUA CCA CAA CUG CCG CAG-Glutamine (Q) -Arginine (R) CGA CGG AUU ACU AAU AGU AUC Isoleucine (1) Asparagine (N) ACC AAC Threonine (T) AUA ACA AAA Methionine (M) Lysine (K) AUG ACG Start codon AAG AGC-Serine (S) -Arginine (R) AGA AGG GUU GCU GAU GUC GUA GUG GCC Valine (V) -Alanine (A) GCA GCG GAC GAA GAG Aspartic acid (D) GGU Glutamic acid (E) GGC GGA GGG Glycine (G) In order to provide a complete answer to the question stated above, fill in the mRNA bases and amino acid sequences by using the Genetic Code…arrow_forwardidentify the indicated cell in white arrowarrow_forwardGloeocaspa Genus - diagram a colony and label the sheath, cell wall, and cytoplasm. Oscillatoria Genus - Diagram a trichome, and label the shealth and individual cells Nostoc Genus- diagram a sketch of the colonoy microscopically from low power to the left of the drawing. Draw a filament showing intercalary heterocysts, and vegatative cells to the right of the drawing Merismopedia Genus- diagram a sketch of the colony. draw and label a filament showing the colony, cell wall, and sheath. Gloeotrichia Genus- diagram a habit sketch of the colony. draw a filament showing the heterocyst, akimetes and vegatative cells of the filamentarrow_forward
- What Genus is this?arrow_forwardAs a medical professional, it is important to be able to discuss how genetic processes such as translation regulation can directly affect patients. Think about some situations that might involve translation regulation. Respond to the following in a minimum of 175 words: Why is translation regulation important? What are some examples of translation regulation in humans? Select one of the examples you provided and explain what happens when translation regulation goes wrong.arrow_forwardThe metabolic pathway below is used for the production of the purine nucleotides adenosine monophosphate (AMP) and guanosine monophosphate (GMP) in eukaryotic cells. Assume each arrow represents a reaction catalyzed by a different enzyme. Using the principles of feedback inhibition, propose a regulatory scheme for this pathway that ensures an adequate supply of both AMP and GMP, and prevents the buildup of Intermediates A through G when supplies of both AMP and GMP are adequate.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning

Human Heredity: Principles and Issues (MindTap Co...
Biology
ISBN:9781305251052
Author:Michael Cummings
Publisher:Cengage Learning

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:9781305112100
Author:Cecie Starr, Beverly McMillan
Publisher:Cengage Learning

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY