
(a)
Interpretation:
The given alcohol is classified as what type of alcohol has to be identified.
Concept Introduction:
Alcohols are classified as primary, secondary, or tertiary depending upon the number of carbon atoms that is bonded to the carbon atom bearing hydroxyl group.
Primary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to only one other carbon atom.
Secondary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to two other carbon atoms.
Tertiary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to three other carbon atoms.
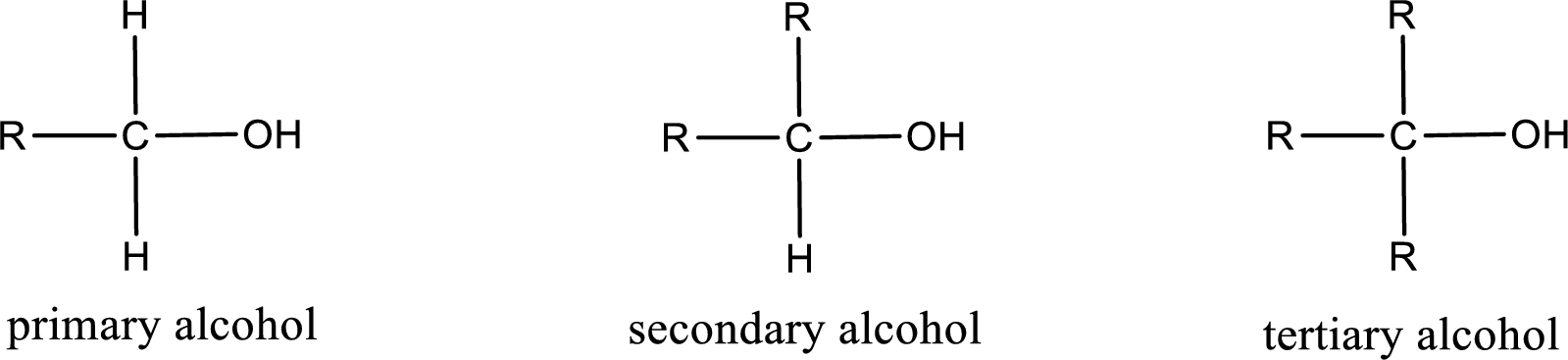
Primary is denoted as
(b)
Interpretation:
The given alcohol is classified as what type of alcohol has to be identified.
Concept Introduction:
Alcohols are classified as primary, secondary, or tertiary depending upon the number of carbon atoms that is bonded to the carbon atom bearing hydroxyl group.
Primary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to only one other carbon atom.
Secondary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to two other carbon atoms.
Tertiary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to three other carbon atoms.
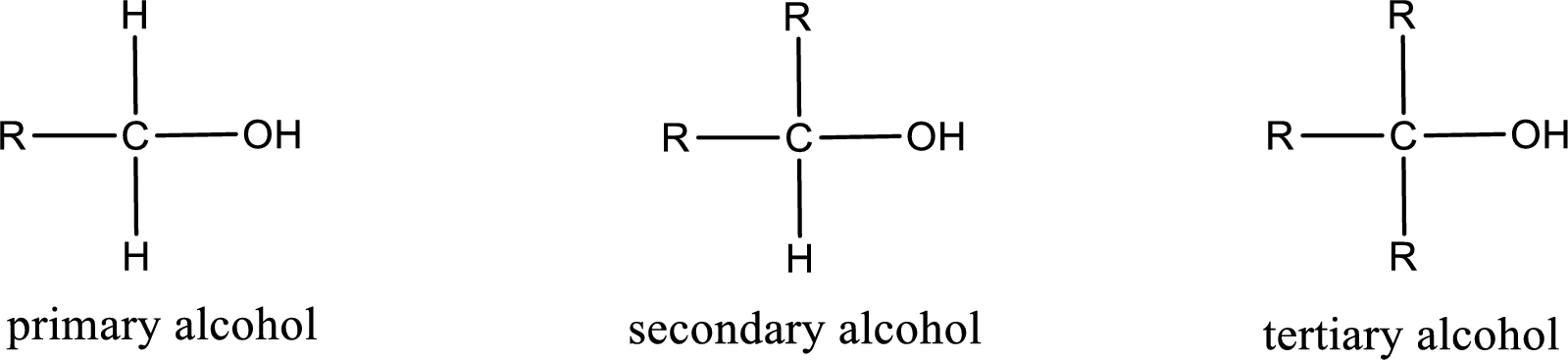
Primary is denoted as
(c)
Interpretation:
The given alcohol is classified as what type of alcohol has to be identified.
Concept Introduction:
Alcohols are classified as primary, secondary, or tertiary depending upon the number of carbon atoms that is bonded to the carbon atom bearing hydroxyl group.
Primary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to only one other carbon atom.
Secondary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to two other carbon atoms.
Tertiary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to three other carbon atoms.
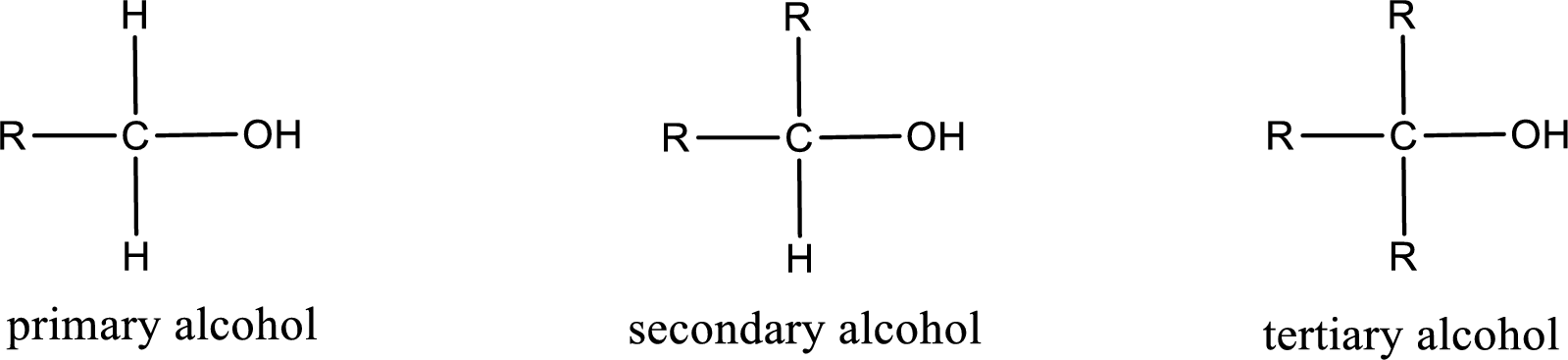
Primary is denoted as
(d)
Interpretation:
The given alcohol is classified as what type of alcohol has to be identified.
Concept Introduction:
Alcohols are classified as primary, secondary, or tertiary depending upon the number of carbon atoms that is bonded to the carbon atom bearing hydroxyl group.
Primary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to only one other carbon atom.
Secondary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to two other carbon atoms.
Tertiary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to three other carbon atoms.
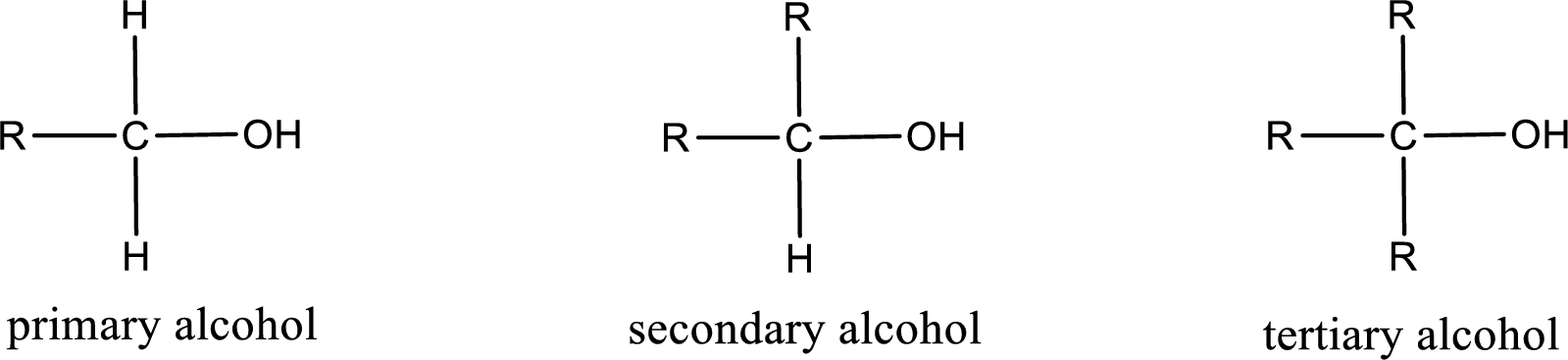
Primary is denoted as
Trending nowThis is a popular solution!

Chapter 3 Solutions
EBK ORGANIC AND BIOLOGICAL CHEMISTRY
- Provide the semi-developed formula of isooxazole obtained by reacting acetylacetone and hydroxylamine.arrow_forwardGiven a 1,3-dicarbonyl compound (R1-CO-CH2-CO-R2), indicate the formula of the compound obtaineda) if I add hydroxylamine (NH2OH) to give an isooxazole.b) if I add thiosemicarbazide (NH2-CO-NH-NH2) to give an isothiazole.arrow_forwardAn orange laser has a wavelength of 610 nm. What is the energy of this light?arrow_forward
- The molar absorptivity of a protein in water at 280 nm can be estimated within ~5-10% from its content of the amino acids tyrosine and tryptophan and from the number of disulfide linkages (R-S-S-R) between cysteine residues: Ε280 nm (M-1 cm-1) ≈ 5500 nTrp + 1490 nTyr + 125 nS-S where nTrp is the number of tryptophans, nTyr is the number of tyrosines, and nS-S is the number of disulfide linkages. The protein human serum transferrin has 678 amino acids including 8 tryptophans, 26 tyrosines, and 19 disulfide linkages. The molecular mass of the most dominant for is 79550. Predict the molar absorptivity of transferrin. Predict the absorbance of a solution that’s 1.000 g/L transferrin in a 1.000-cm-pathlength cuvet. Estimate the g/L of a transferrin solution with an absorbance of 1.50 at 280 nm.arrow_forwardIn GC, what order will the following molecules elute from the column? CH3OCH3, CH3CH2OH, C3H8, C4H10arrow_forwardBeer’s Law is A = εbc, where A is absorbance, ε is the molar absorptivity (which is specific to the compound and wavelength in the measurement), and c is concentration. The absorbance of a 2.31 × 10-5 M solution of a compound is 0.822 at a wavelength of 266 nm in a 1.00-cm cell. Calculate the molar absorptivity at 266 nm.arrow_forward
- How to calculate % of unknown solution using line of best fit y=0.1227x + 0.0292 (y=2.244)arrow_forwardGiven a 1,3-dicarbonyl compound, state the (condensed) formula of the compound obtaineda) if I add hydroxylamine (NH2OH) to give an isooxazole.b) if I add thiosemicarbazide (NH2-CO-NH-NH2) to give an isothiazole.arrow_forwardComplete the following acid-base reactions and predict the direction of equilibrium for each. Justify your prediction by citing pK values for the acid and conjugate acid in each equilibrium. (a) (b) NHs (c) O₂N NH NH OH H₁PO₁arrow_forward
- 23.34 Show how to convert each starting material into isobutylamine in good yield. ཅ ནད ཀྱི (b) Br OEt (c) (d) (e) (f) Harrow_forwardPlease help me Please use https://app.molview.com/ to draw this. I tried, but I couldn't figure out how to do it.arrow_forwardPropose a synthesis of 1-butanamine from the following: (a) a chloroalkane of three carbons (b) a chloroalkane of four carbonsarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning





