
(a)
Interpretation: The given compounds in each group are to be ranked in the order of increasing boiling point.
Concept introduction: Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
Intermolecular forces are also known as non-covalent interactions. The interactions present between molecules are known as intermolecular forces. A
Therefore, the increasing order of boiling point is as follows:
Answer to Problem 3.32P
The increasing order of boiling point for the given compounds is
Explanation of Solution
The given compounds are
Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
The increasing order of intermolecular force strength is as follows:
Therefore, the increasing order of boiling point is as follows:
The boiling point increases with an increase in the surface area.
Therefore, the increasing order of boiling point for the given compounds is
The increasing order of boiling point for the given compounds is
(b)
Interpretation: The given compounds in each group are to be ranked in the order of increasing boiling point.
Concept introduction: Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
Intermolecular forces are also known as non-covalent interactions. The interactions present between molecules are known as intermolecular forces. A functional group present in a molecule decides the type of interaction.
Therefore, the increasing order of boiling point is as follows:
Answer to Problem 3.32P
The increasing order of boiling point for the given compounds is
Explanation of Solution
The given compounds are
Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
In
The increasing order of intermolecular force strength is as follows:
Therefore, the increasing order of boiling point is as follows:
Therefore, the increasing order of boiling point for the given compounds is
The increasing order of boiling point for the given compounds is
(c)
Interpretation: The given compounds in each group are to be ranked in the order of increasing boiling point.
Concept introduction: Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
Intermolecular forces are also known as non-covalent interactions. The interactions present between molecules are known as intermolecular forces. A functional group present in a molecule decides the type of interaction.
Therefore, the increasing order of boiling point is as follows:
Answer to Problem 3.32P
The increasing order of boiling point for the given compounds is
Explanation of Solution
The given compounds are
Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
In
The increasing order of intermolecular force strength is as follows:
Therefore, the increasing order of boiling point is as follows:
The boiling point increases with an increase in the surface area. Branched chain alkanes have low boiling point than straight chain alkanes because in branched chain alkanes, surface area is less.
Therefore, the increasing order of boiling point for the given compounds is
The increasing order of boiling point for the given compounds is
(d)
Interpretation: The given compounds in each group are to be ranked in the order of increasing boiling point.
Concept introduction: Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
Intermolecular forces are also known as non-covalent interactions. The interactions present between molecules are known as intermolecular forces. A functional group present in a molecule decides the type of interaction.
Therefore, the increasing order of boiling point is as follows:
Answer to Problem 3.32P
The increasing order of intermolecular force strength for the given compounds is shown below.
![]()
Explanation of Solution
The given compounds are,
![]()
Figure 1
Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
The compounds containing hydroxyl groups show hydrogen bonding.
The increasing order of intermolecular force strength is as follows:
Therefore, the increasing order of boiling point is as follows:
The boiling point increases with an increase in the surface area. Branched chain
Therefore, the increasing order of boiling point for the given compounds is,
![]()
Figure 2
The increasing order of boiling point for the given compounds is shown in Figure 2.
(e)
Interpretation: The given compounds in each group are to be ranked in the order of increasing boiling point.
Concept introduction: Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
Intermolecular forces are also known as non-covalent interactions. The interactions present between molecules are known as intermolecular forces. A functional group present in a molecule decides the type of interaction.
Therefore, the increasing order of boiling point is as follows:
Answer to Problem 3.32P
The increasing order of intermolecular force strength for the given compounds is shown below.
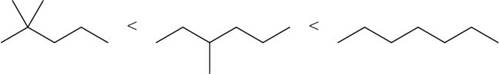
Explanation of Solution
The given compounds are,
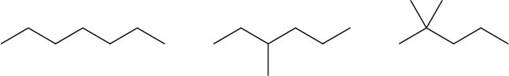
Figure 3
Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
The increasing order of intermolecular force strength is as follows:
Therefore, the increasing order of boiling point is as follows:
The boiling point increases with an increase in the surface area. Branched chain alkanes have low boiling point than straight chain alkanes because in branched chain alkanes, surface area is less.
Therefore, the increasing order of boiling point for the given compounds is,

Figure 4
The increasing order of boiling point for the given compounds is rightfully stated.
(f)
Interpretation: The given compounds in each group are to be ranked in the order of increasing boiling point.
Concept introduction: Boiling point depends upon the intermolecular forces. Greater is the intermolecular forces, greater will be the boiling point.
Intermolecular forces are also known as non-covalent interactions. The interactions present between molecules are known as intermolecular forces. A functional group present in a molecule decides the type of interaction. The increasing order of intermolecular force strength is as follows:
Therefore, the increasing order of boiling point is as follows:
Answer to Problem 3.32P
The increasing order of intermolecular force strength for the given compounds is shown below.
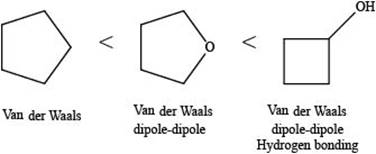
Explanation of Solution
The given compounds are,
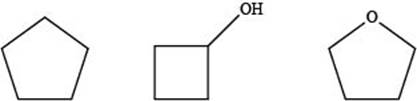
Figure 5
A hydrogen bond is a strong electrostatic attraction which takes place when hydrogen atom is bonded to an electronegative atom (
Dipole-dipole interactions are the forces present between two polar molecules.
Van der Waals forces are the weak forces that are present between non-polar compounds or molecules.
The increasing order of intermolecular force strength is as follows:
The interaction present in cyclopentane is Van der Waals forces because it is a non-polar compound.
The interactions present in cyclobutanolare Van der Waals forces, hydrogen bonding, and dipole-dipole interactions.
Due to electronegativity difference between carbon and oxygen, ethers are polar molecule. Thus, the interaction present in polar molecules is Dipole-dipole interaction. The interactions present in tetrahydrofuran are Van der Waals forces and dipole-dipole interactions.
Therefore, the increasing order of boiling point for the given compounds is,
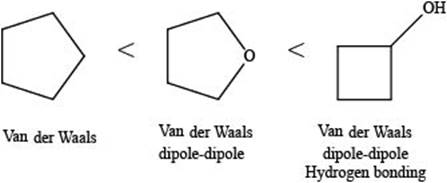
Figure 6
The increasing order of boiling point for the given compounds is rightfully stated.
Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry
- Show how to convert ethyl benzene to (a) 2,5-dichlorobenzoic acid and (b) 2,4-dichlorobenzoic acid.arrow_forwardHelp me solve this problem. Thank you in advance.arrow_forward22.7 Predict the monoalkylated products of the following reactions with benzene. (a) AlCl3 Ya (b) AlCl3 (c) H3PO4 (d) 22.8 Think-Pair-Share AICI3 The reaction below is a common electrophilic aromatic substitution. SO3 H₂SO4 SO₂H (a) Draw the reaction mechanism for this reaction using HSO,+ as the electrophile. (b) Sketch the reaction coordinate diagram, where the product is lower in energy than the starting reactant. (c) Which step in the reaction mechanism is highest in energy? Explain. (d) Which of the following reaction conditions could be used in an electrophilic aro- matic substitution with benzene to provide substituted phenyl derivatives? (i) AICI3 HNO3 H₂SO4 K2Cr2O7 (iii) H₂SO4 (iv) H₂PO₁arrow_forward
- Is an acid-base reaction the only type of reaction that would cause leavening products to rise?arrow_forwardHelp me understand this! Thank you in advance.arrow_forward22.22 For each compound, indicate which group on the ring is more strongly activating and then draw a structural formula of the major product formed by nitration of the compound. Br CHO (a) CH3 (b) (c) CHO CH3 SO₂H (d) ☑ OCHS NO₂ (e) (f) CO₂H NHCOCH3 NHCOCH, (h) CHS 22.23 The following molecules each contain two aromatic rings. (b) 000-100- H3C (a) (c) Which ring in each undergoes electrophilic aromatic substitution more readily? Draw the major product formed on nitration.arrow_forward
- V Consider this step in a radical reaction: Br: ? What type of step is this? Check all that apply. Draw the products of the step on the right-hand side of the drawing area below. If more than one set of products is possible, draw any set. Also, draw the mechanism arrows on the left-hand side of the drawing area to show how this happens. ⚫ionization termination initialization neutralization none of the abc Explanation Check 80 Ο F3 F1 F2 2 F4 01 % do5 $ 94 #3 X 5 C MacBook Air 25 F5 F6 66 ©2025 ˇ F7 29 & 7 8arrow_forwardShow how to convert ethyl benzene to (a) 2,5-dichlorobenzoic acid and (b) 2,4-dichlorobenzoic acid.arrow_forwardno aiarrow_forward
- Polymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer formed in this reaction. Assume there are hydrogen atoms there are hydrogen atoms on the two ends of the trimer. Ignore inorganic byproducts.arrow_forwardDraw a tetramer if this alternating copolymer pleasearrow_forwardDraw the monomers required to synthesize this condensation polymer.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning




