
(a)
Interpretation: The more highly oxidized compound is to be identified.
Concept Introduction: In the process of oxidation, oxygen or any other electronegative element is added, and hydrogen or another electropositive element is removed. The process of an atom or ion losing one or more electrons is known as oxidation, according to the electronic notion.
(a)
Answer to Problem 110A
Ethanal is more highly oxidized compound.
Explanation of Solution
The compound that is highly oxidized can be detected with the help of its structure.
The compound that has a low number of hydrogen atoms has the tendency to get more oxidized.
The structure of ethanol and ethanal is given below:
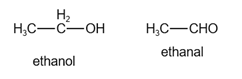
In ethanol, there are six hydrogen atoms.
In ethanal, there are four hydrogen atoms.
Since ethanal has few number of hydrogen atoms, it is highly oxidized.
(b)
Interpretation: The more highly oxidized compound is to be identified.
Concept Introduction: In the process of oxidation, oxygen or any other electronegative element is added, and hydrogen or another electropositive element is removed. The process of an atom or ion losing one or more electrons is known as oxidation, according to the electronic notion.
(b)
Answer to Problem 110A
Ethene is more highly oxidized compound.
Explanation of Solution
The compound that is highly oxidized can be detected with the help of its structure.
The compound that has a low number of hydrogen atoms has the tendency to get more oxidized.
The structure of ethane and ethene is given below:
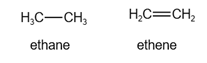
In ethane, there are six hydrogen atoms.
In ethene, there are four hydrogen atoms.
Since ethene has few hydrogen atoms, it is highly oxidized.
(c)
Interpretation: The more highly oxidized compound is to be identified.
Concept Introduction: In the process of oxidation, oxygen or any other electronegative element is added, and hydrogen or another electropositive element is removed. The process of an atom or ion losing one or more electrons is known as oxidation, according to the electronic notion.
(c)
Answer to Problem 110A
Ethanoic acid is a more highly oxidized compound.
Explanation of Solution
The compound that is highly oxidized can be detected with the help of its structure.
The compound that has a low number of hydrogen atoms has the tendency to get more oxidized.
The structure of ethanoic acid and ethanal is given below:
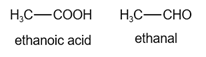
In ethanoic acid, there are four hydrogen atoms.
In ethanal, there are four hydrogen atoms.
Since
(d)
Interpretation: The more highly oxidized compound is to be identified.
Concept Introduction: In the process of oxidation, oxygen or any other electronegative element is added, and hydrogen or another electropositive element is removed. The process of an atom or ion losing one or more electrons is known as oxidation, according to the electronic notion.
(d)
Answer to Problem 110A
Ethyne is more highly oxidized compound.
Explanation of Solution
The compound that is highly oxidized can be detected with the help of its structure.
The compound that has a low number of hydrogen atoms has the tendency to get more oxidized.
The structure of ethyne and ethane is given below:
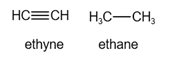
In ethyne, there are two hydrogen atoms.
In ethane, there are six hydrogen atoms.
Since ethyne has a few number of hydrogen atoms, it is highly oxidized.
Chapter 25 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
- b) Certain cyclic compounds are known to be conformationally similar to carbohydrates, although they are not themselves carbohydrates. One example is Compound C shown below, which could be imagined as adopting four possible conformations. In reality, however, only one of these is particularly stable. Circle the conformation you expect to be the most stable, and provide an explanation to justify your choice. For your explanation to be both convincing and correct, it must contain not only words, but also "cartoon" orbital drawings contrasting the four structures. Compound C Possible conformations (circle one): Детarrow_forwardLab Data The distance entered is out of the expected range. Check your calculations and conversion factors. Verify your distance. Will the gas cloud be closer to the cotton ball with HCI or NH3? Did you report your data to the correct number of significant figures? - X Experimental Set-up HCI-NH3 NH3-HCI Longer Tube Time elapsed (min) 5 (exact) 5 (exact) Distance between cotton balls (cm) 24.30 24.40 Distance to cloud (cm) 9.70 14.16 Distance traveled by HCI (cm) 9.70 9.80 Distance traveled by NH3 (cm) 14.60 14.50 Diffusion rate of HCI (cm/hr) 116 118 Diffusion rate of NH3 (cm/hr) 175.2 175.2 How to measure distance and calculate ratearrow_forwardFor the titration of a divalent metal ion (M2+) with EDTA, the stoichiometry of the reaction is typically: 1:1 (one mole of EDTA per mole of metal ion) 2:1 (two moles of EDTA per mole of metal ion) 1:2 (one mole of EDTA per two moles of metal ion) None of the abovearrow_forward
- Please help me solve this reaction.arrow_forwardIndicate the products obtained by mixing 2,2-dimethylpropanal with acetaldehyde and sodium ethoxide in ethanol.arrow_forwardSynthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- If possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forwardSynthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





