
Concept explainers
Name each of the following compounds.
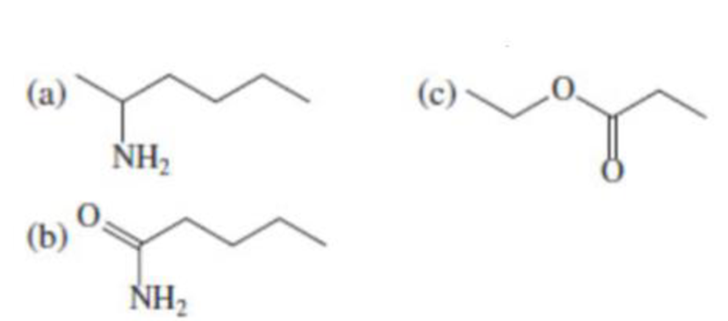
(a)
Interpretation:
The IUPAC name of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted alkane:
- (1) Name the parent alkane (long alkyl chain)
- (2) Number the carbon
- (3) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
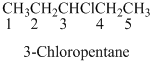
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below


The given compound is an acid (
 ssw
ssw
The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
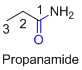
If the molecule is ester,
Esters end with “ate”
Example

The given compound is an aldehyde (
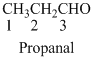
The given compound is a ketone (
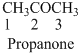
The given compound is an amine (
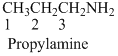
Answer to Problem 23.10QP
Answer
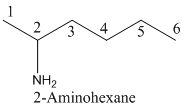
- (1) Name of the given organic compounds is shown below (a).
Explanation of Solution
To find: The name of the given organic compounds
Name of the given organic compounds is shown below.

Parent chain is identified and numbering is given for the compound.
Second carbon is bearing one amino group and it has six carbon atom in the molecule. The name of the given organic compound is 2-Aminohexane.
(b)
Interpretation:
The IUPAC name of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted alkane:
- (1) Name the parent alkane (long alkyl chain)
- (2) Number the carbon
- (3) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
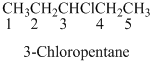
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below


The given compound is an acid (
 ssw
ssw
The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
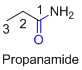
If the molecule is ester,
Esters end with “ate”
Example

The given compound is an aldehyde (
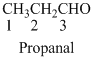
The given compound is a ketone (
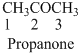
The given compound is an amine (
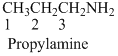
Answer to Problem 23.10QP
Name of the given organic compounds is shown below (b)
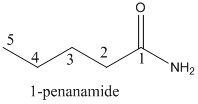
Explanation of Solution
To find: The name of the given organic compounds
Name of the given organic compounds is shown below (b)

Parent chain is identified and numbering is given for the compound.
Fifth carbon is bearing amide group and it has five carbon atom in the molecule. The name of the given organic compound is pentanamide.
(c)
Interpretation:
The IUPAC name of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted alkane:
- (1) Name the parent alkane (long alkyl chain)
- (2) Number the carbon
- (3) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
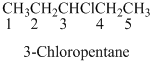
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below


The given compound is an acid (
 ssw
ssw
The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
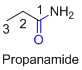
If the molecule is ester,
Esters end with “ate”
Example

The given compound is an aldehyde (
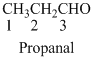
The given compound is a ketone (
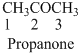
The given compound is an amine (
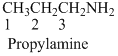
Answer to Problem 23.10QP
Answer
Name of the given organic compounds is shown below (c)
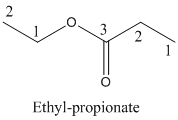
Explanation of Solution
To find: The name of the given organic compounds
Name of the given organic compounds is shown below (c)
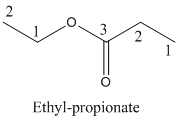
Parent chain is identified and numbering is given for the compound.
Third carbon is bearing ester group and it has five carbon atoms in the molecule. The name of the given organic compound is ethyl propionate.
Want to see more full solutions like this?
Chapter 23 Solutions
Chemistry: Atoms First
- On the next page is an LC separation of the parabens found in baby wash. Parabens are suspected in a link to breast cancer therefore an accurate way to quantitate them is desired. a. In the chromatogram, estimate k' for ethyl paraben. Clearly indicate what values you used for all the terms in your calculation. b. Is this a "good" value for a capacity factor? Explain. c. What is the resolution between n-Propyl paraben and n-Butyl paraben? Again, indicate clearly what values you used in your calculation. MAU | Methyl paraben 40 20 0 -2 Ethyl paraben n-Propyl paraben n-Butyl paraben App ID 22925 6 8 minarrow_forwardd. In Figure 4, each stationary phase shows some negative correlation between plate count and retention factor. In other words, as k' increases, N decreases. Explain this relationship between k' and N. Plate Count (N) 4000 3500 2500 2000 1500 1000 Figure 4. Column efficiency (N) vs retention factor (k') for 22 nonionizable solutes on FMS (red), PGC (black), and COZ (green). 3000 Eluent compositions (acetonitrile/water, A/W) were adjusted to obtain k' less than 15, which was achieved for most solutes as follows: FMS (30/70 A/W), PGC (60/40), COZ (80/20). Slightly different compositions were used for the most highly retained solutes. All columns were 50 mm × 4.6 mm id and packed with 5 um particles, except for COZ, which was packed with 3 um particles. All other chromatographic conditions were constant: column length 5 cm, column j.§. 4.6 mm, flow rate 2 mL/min, column temperature 40 °C, and injection volume 0.5 μL Log(k'x/K'ethylbenzene) FMS 1.5 1.0 0.5 0.0 ཐྭ ཋ ཤྩ བྷྲ ; 500 0 5 10…arrow_forwardf. Predict how the van Deemter curve in Figure 7 would change if the temperature were raised from 40 °C to 55 °C. Figure 7. van Desmter curves in reduced coordinates for four nitroalkane homologues (nitropropane, black; nitrobutane, red; nitropentane, blue; and nitrohexane, green) separated on the FMS phase. Chromatographic conditions: column dimensions 50 mm × 4.6 mm id, eluent 30/70 ACN/water, flow rates 0.2-5.0 mL/min, injection volume 0.5 and column temperature 40 °C. No corrections to the plate heights have been made to account for extracolumn dispersion. Reduced Plate Height (h) ° 20 40 60 Reduced Velocity (v) 8. (2) A water sample is analyzed for traces of benzene using headspace analysis. The sample and standard are spiked with a fixed amount of toluene as an internal standard. The following data are obtained: Ppb benzene Peak area benzene Peak area toluene 10.0 252 376 Sample 533 368 What is the concentration of benzene in the sample?arrow_forward
- Liquid chromatography has been used to track the concentration of remdesivir (a broad-spectrum antiviral drug, structure shown at right) in COVID patients undergoing experimental treatments. Intensity The authors provide the following details regarding standard solutions preparation: HN CN HO OH NH2 Remdesivir (RDV) stock solution (5000 µg/mL) was prepared by dissolving RDV drug powder using the mixture of DMSO: MeOH (30:70 v/v). The RDV working standard solutions for calibration and quality controls were prepared using methanol in concentrations of 100, 10, 1, 0.1, 0.01 µg/mL. 1, 2.5, 5, 7.5, 10, 25, 50, 75, 100, 250, 500, 1000, and 5000 ng/mL sample solutions were prepared freshly by spiking calibration standard solutions into the blank human plasma samples for method calibration. a) What type of calibration method is being described? Why do you think the authors chose this method as opposed to another? b) Based on the details provided in part a, describe an appropriate method blank…arrow_forwardRecent advancements in liquid chromatography include the development of ultrahigh pressure liquid chromatography (UHPLC) and an increased use of capillary columns that had previously only been used with gas chromatography. Both of these advances have made the development of portable LC systems possible. For example, Axcend Corp. makes a portable system that uses a capillary column with an internal diameter of 150-μm-that is packed with 1.7-um stationary phase particles. In contrast, a traditional LC column has a 4.6 mm internal diameter and utilizes 5-um stationary phase particles. a) Explain one advantage that is afforded by the use of a capillary column in liquid chromatographic separation. Explain one disadvantage of capillary columns. b) Explain how the use of smaller stationary phase particles can improve the resolution of a separation. Include any relevant equations that support your explanation. c) A scientist at Rowan University is using the Axcend LC to conduct analyses of F…arrow_forwardThis paper describes the use of fullerene molecules, also known as buckyballs, as a stationary phase for liquid chromatography. The performance of the fullerene-modified stationary phase (FMS) is compared to that of a more common C18 stationary phase and to two other carbon-based stationary phases, PGC and COZ. A. 10A OM B. - Figure 1. Idealized drawing of the cross-section of a pore inside a silica particle, showing the relative densities of aminopropylsilyl (red/green) and fullerene (blue) groups: (A) full cross- section; (B) detailed view of covalent bonding of fullerene to the silica surface. Surface densities of silyl and fullerene groups were inferred from elemental composition results obtained at each stage of the synthesis (see Table 1). Absorbance (mAU, 220 nm) 700 600 500 400 300 200 100 a. Define selectivity, a, with words and an equation. b. Explain how the choice of stationary phase affects selectivity. c. Calculate the resolution of the nitrobenzene and toluene peaks in…arrow_forward
- Normalized Intensity (a. u.) 0.5 1.0 A 3D-printed GC column (shown below) was created for use with "micro" gas chromatography applications. To prove its utility, it was used to separate a mixture of alkanes (C9-C18, C22, C24). For the separation shown below, the column temperature was ramped from 40 °C to 250 °C at a rate of 30 °C per minute. (a) 9 10 = 1 mm 12 13 15 22 0.0 0 100 200 300 400 Time (sec) a) What detector would you use for this analysis? Justify your selection. b) Explain how the chromatogram would change if the separation was run isothermally. c) Explain how the chromatogram would change if the temperature ramp were increased to 50 °C per minute.arrow_forwardDevise a synthesis of each compound from the indicated starting material. You may also use any organic compounds with one or two carbons and any needed inorganic reagents. a. Brarrow_forwardPlease help me with #2b & #3 using the data.arrow_forward
- Heparin is used as an anti-coagulant. A risk of heparin use is thrombocytopenia, or low platelet count. This risk is minimized with the use of low molecular weight heparins (LMWH), therefore it is desirable to separate LMWH from higher molecular weight heparins. The method of choice to do this is molecular exclusion chromatography. Below is a chromatogram from a molecular exclusion chromatographic run. Peaks ranging from A to J are clearly distinguishable. The heparin mixture that was analyzed had anywhere from 6 to 30 repeat units of monomer (where the heparin with 30 repeat units would be roughly five times the size of the heparin with six repeat units). a. Which letter most likely represents the peak with 6 repeat units given these heparin polymers were separated with molecular exclusion chromatography? b. Explain your reasoning describing the mechanism of retention in molecular exclusion chromatography. 100 80 60 60 Relative Abundance 40 40 E GH 20 20 B A 36 38 40 42 44 46 48 50 50…arrow_forwardHELP NOW PLEASE ! URGENT!arrow_forwardHELP NOW PLEASE ! URGENT!arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





