
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
4th Edition
ISBN: 9781265982959
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 54P
What type of interaction occur at each of the labeled sites in the portion of the protein ribbon shown?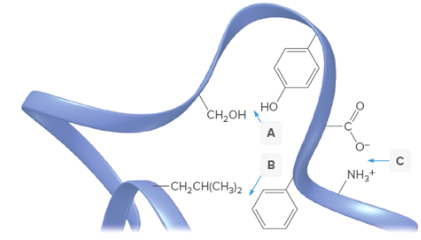
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider the following gas chromatographs of Compound A, Compound B, and a mixture of Compounds A and B.
Inject
A
B
mixture
Area= 9
Area = 5
Area = 3
Area
Inject
.
མི།
Inject
J2
What is the percentage of Compound B in the the mixture?
Rank these according to stability.
CH3
H3C
CH3
1
CH3
H3C
1 most stable, 3 least stable
O 1 most stable, 2 least stable
2 most stable, 1 least stable
O2 most stable, 3 least stable
O3 most stable, 2 least stable
O3 most stable, 1 least stable
CH3
2
CH3
CH3
H₂C
CH3
3
CH3
CH
Consider this IR and NMR:
INFRARED SPECTRUM
TRANSMITTANCE
0.8-
0.6
0.4
0.2
3000
10
9
8
00
HSP-00-541
7
CO
6
2000
Wavenumber (cm-1)
сл
5
ppm
4
M
Which compound gave rise to these spectra?
N
1000
1
0
Chapter 21 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
Ch. 21.2 - In addition to the amino and carboxyl groups, what...Ch. 21.2 - Draw both enantiomers of each amino acid in...Ch. 21.2 - Which of the following amino acids is naturally...Ch. 21.3 - Draw the structure of the amino acid valine at...Ch. 21.3 - Identify the amino acid shown with all uncharged...Ch. 21.4 - Identify the N-terminal and C-terminal amino acid...Ch. 21.4 - Prob. 21.4PCh. 21.4 - Prob. 21.4PPCh. 21.4 - Prob. 21.5PCh. 21.4 - Prob. 21.5PP
Ch. 21.4 - Prob. 21.6PPCh. 21.5 - Prob. 21.6PCh. 21.6 - Prob. 21.7PCh. 21.6 - Prob. 21.8PCh. 21.6 - Prob. 21.9PCh. 21.7 - Why is hemoglobin more water soluble than ...Ch. 21.8 - Prob. 21.7PPCh. 21.8 - Prob. 21.11PCh. 21.9 - Prob. 21.8PPCh. 21.9 - Prob. 21.12PCh. 21.9 - Prob. 21.9PPCh. 21.9 - Prob. 21.13PCh. 21.10 - Prob. 21.14PCh. 21.10 - Prob. 21.15PCh. 21.10 - Prob. 21.16PCh. 21.10 - Prob. 21.17PCh. 21.10 - The nerve gas sarin acts as a poison by covalently...Ch. 21.10 - Prob. 21.19PCh. 21.10 - Explain why the proteins involved in blood...Ch. 21 - The amino acid alanine is a solid at room...Ch. 21 - Why is phenylalanine water soluble but...Ch. 21 - Draw the structure of a naturally occurring amino...Ch. 21 - Draw the structure of a naturally occurring amino...Ch. 21 - For each amino acid: [1] draw the L enantiomer in...Ch. 21 - For each amino acid: [1] draw the L enantiomer in...Ch. 21 - Draw both enantiomers of each amino acid and label...Ch. 21 - Which of the following Fischer projections...Ch. 21 - For each amino acid: [1] give the name; [2] give...Ch. 21 - For each amino acid: [1] give the name; [2] give...Ch. 21 - (a) Identify the amino acid shown with all...Ch. 21 - Prob. 32PCh. 21 - Prob. 33PCh. 21 - Draw the structure of the neutral, positively...Ch. 21 - Locate the peptide bond in the dipeptide shown in...Ch. 21 - Label the N-terminal and C-terminal amino acids in...Ch. 21 - Melittin, the principal toxin of bee venom,...Ch. 21 - Cobratoxin is a neurotoxin found in the venom of...Ch. 21 - (a) Draw the structure of the two possible...Ch. 21 - (a) Draw the structure of the two possible...Ch. 21 - For each tripeptide: [1] draw the structure of the...Ch. 21 - For each tripeptide: [1] draw the structure of the...Ch. 21 - Prob. 43PCh. 21 - For each tripeptide: [1] identify the amino acids...Ch. 21 - What amino acids are formed by hydrolysis of the...Ch. 21 - Prob. 46PCh. 21 - Prob. 47PCh. 21 - Draw the structures of the amino acids formed when...Ch. 21 - Prob. 49PCh. 21 - Prob. 50PCh. 21 - Prob. 51PCh. 21 - Trypsin is a digestive enzyme that hydrolyzes...Ch. 21 - What type of intermolecular forces exist between...Ch. 21 - What type of interaction occur at each of the...Ch. 21 - Which peptide in each pair contains amino acids...Ch. 21 - Decide if the side chains of the amino acid...Ch. 21 - Which type of protein structure is indicated in...Ch. 21 - Label each of the following diagrams as...Ch. 21 - Prob. 59PCh. 21 - Prob. 60PCh. 21 - Prob. 61PCh. 21 - Prob. 62PCh. 21 - Compare - keratin and hemoglobin with regard to...Ch. 21 - Compare collagen and myoglobin with regard to each...Ch. 21 - Prob. 65PCh. 21 - Prob. 66PCh. 21 - Describe the function or biological activity of...Ch. 21 - Describe the function or biological activity of...Ch. 21 - Prob. 69PCh. 21 - Prob. 70PCh. 21 - What class of enzyme catalyzes each of the...Ch. 21 - What class of enzyme catalyzes each of the...Ch. 21 - Prob. 73PCh. 21 - Prob. 74PCh. 21 - Prob. 75PCh. 21 - What kind of reaction is catalyzed by each of the...Ch. 21 - Prob. 77PCh. 21 - How will each of the following changes affect the...Ch. 21 - Prob. 79PCh. 21 - Prob. 80PCh. 21 - Prob. 81PCh. 21 - Prob. 82PCh. 21 - Prob. 83PCh. 21 - Prob. 84PCh. 21 - Prob. 85PCh. 21 - Prob. 86PCh. 21 - Why must vegetarian diets be carefully balanced?Ch. 21 - Prob. 88PCh. 21 - Sometimes an incision is cauterized (burned) to...Ch. 21 - Why is insulin administered by injection instead...Ch. 21 - Prob. 91PCh. 21 - The silk produced by a silkworm is a protein with...Ch. 21 - Explain the difference in the mechanism of action...Ch. 21 - Prob. 94PCh. 21 - Prob. 95CPCh. 21 - Suggest a reason for the following observation....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider this reaction (molecular weights are under each compound): HC=CH + 2 HCI --> C2H4Cl 2 MW = 26 36.5 99 If 4.4 g of HC=CH are reacted with 110 mL of a 2.3 M HCI solution, and 6.0 g of product are actually produced, what is the percent yield?arrow_forwardWhat is the name of the major product of this reaction? OH CH3 H₂SO4, heat 1-methylcyclohexene O2-methyl-1-cyclohexene O 3-mthylcyclohexene 1-methyl-2-cyclohexenearrow_forwardWe added a brown solution of Br2 to one of our products, and the brown color disappeared. This indicated that our product wasarrow_forward
- Rank the following according to reactivity toward nitration: a) benzene b) bromobenzene c) nitrobenzene d) phenol Od) greatest, c) least Od) greatest, b) least Od) greatest, a) least a) greatest, b) least a) greatest, c) least Oa) greatest, d) least Ob) greatest, a) least O b) greatest, c) least Ob) greatest, d) least O c) greatest, a) least O c) greatest, b) least O c) greatest, d) leastarrow_forwardO-Nitrophenol was distilled over with the steam in our experiment while the other isomer did not. This is due to: O intramolecular hydrogen bonding in the ortho isomer O intermolecular hydrogen bonding in the the ortho isomer O the ortho isomer has a lower density O the ortho isomer has a lower molecular weightarrow_forwardK 44% Problem 68 of 15 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. :6: :: :CI: CI CI: :0:0 Select to Add Arrows Select to Add Arrows H H Cl CI: CI CI: Select to Add Arrows Select to Add Arrows H :CI: Alarrow_forward
- I I H :0: Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. 0:0 :0: CI ΑΙ :CI: :CI: :0: CI Select to Add Arrows Select to Add Arrows cl. :0: Cl © ハ CI:: CI H CO Select to Add Arrows Select to Add Arrows 10: AI ::arrow_forwardOrder the following compounds from slowest to fastest in a nucleophilic acyl substitution reaction. ii 요 OB D A E C OCE Darrow_forwardI need the most help figuring out how to find [I^-] mol/ L, [S2O8^2-] mol/L. 1st and 2nd Blank columns.arrow_forwardCan someone help me whats the issue?arrow_forwarda. The change in the Gibbs energy of a certain constant pressure process is found to fit the expression: AG-85.1 J mol −1 +36.5 J mol ¹K-1 × T A. Calculate the value of AS for the process. B. Next, use the Gibbs-Helmholtz equation: (a(AG/T)) ΔΗ - T2 to calculate the value of AH for the process.arrow_forwardNonearrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY