
Organic Chemistry
11th Edition
ISBN: 9781118133576
Author: T. W. Graham Solomons, Craig Fryhle
Publisher: Wiley, John & Sons, Incorporated
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 43P
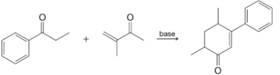
The following reaction illustrates the Robinson annulation reaction (Section 19.7A). Provide a mechanism.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When talking about the acidity of carboxylic acids, is it the same thing to say higher or stronger acidity?
Using the following two half-reactions, determine the pH range in which $NO_2^-\ (aq)$ cannot be found as the predominant chemical species in water.* $NO_3^-(aq)+10H^+(aq)+8e^-\rightarrow NH_4^+(aq)+3H_2O(l),\ pE^{\circ}=14.88$* $NO_2^-(aq)+8H^+(aq)+6e^-\rightarrow NH_4^+(aq)+2H_2O(l),\ pE^{\circ}=15.08$
Indicate characteristics of oxodec acid.
Chapter 19 Solutions
Organic Chemistry
Ch. 19 - PRACTICE PROBLEM 19.1 (a) Write a mechanism. for...Ch. 19 - Practice Problem 19.2
Since the products obtained...Ch. 19 - Prob. 3PPCh. 19 - PRACTICE PROBLEM 19.4
Write mechanisms that...Ch. 19 - Prob. 5PPCh. 19 - Prob. 6PPCh. 19 - Practice Problem 19.7 The acid-catalyzed aldol...Ch. 19 - Prob. 8PPCh. 19 - Practice Problem 19.9
(a) Provide a mechanism for...Ch. 19 - Prob. 10PP
Ch. 19 - Practice Problem 19.11 Outlined below is a...Ch. 19 - Prob. 12PPCh. 19 - Prob. 13PPCh. 19 - Prob. 14PPCh. 19 - Practice Problem 19.15
Starting with ketones and...Ch. 19 - Practice Problem 19.16 Assuming that dehydration...Ch. 19 - Practice Problem 19.17 What starting compound...Ch. 19 - Practice Problem 19.18
What experimental...Ch. 19 - Prob. 19PPCh. 19 - Practice Problem 19.20
When acrolein (propenal)...Ch. 19 - Prob. 21PPCh. 19 - PRACTICE PROBLEM 19.22
Qutline reasonable...Ch. 19 - Prob. 23PCh. 19 - Show all steps in the following syntheses. You may...Ch. 19 - Prob. 25PCh. 19 - Prob. 26PCh. 19 - Prob. 27PCh. 19 - 19.28 Show how the diketone at the right could be...Ch. 19 - Prob. 29PCh. 19 - 19.30 Write a detailed mechanism for the following...Ch. 19 - Prob. 31PCh. 19 - Prob. 32PCh. 19 - 19.33 Predict the products from each of the...Ch. 19 - Prob. 34PCh. 19 - Show how each of the following transformations...Ch. 19 - Prob. 36PCh. 19 - What reagents would you use to bring about each...Ch. 19 - Prob. 38PCh. 19 - Prob. 39PCh. 19 - 19.40 When the aldol reaction of acetaldehyde is...Ch. 19 - Prob. 41PCh. 19 - Prob. 42PCh. 19 - 19.43 The following reaction illustrates the...Ch. 19 - What is the structure of the cyclic compound that...Ch. 19 - Prob. 45PCh. 19 - Prob. 46PCh. 19 - Prob. 47PCh. 19 - Predict the products from the following reactions....Ch. 19 - Prob. 49PCh. 19 - 19.50 (+)-Fenchone is a terpenoid that can be...Ch. 19 - Prob. 51PCh. 19 - Prob. 52PCh. 19 - Prob. 53PCh. 19 - Prob. 54PCh. 19 - The Perkin condensation is an aldol-type...Ch. 19 - 19.55 (a) Infrared spectroscopy provides an easy...Ch. 19 - Prob. 57PCh. 19 - 19.61 Provide a mechanism for each of the...Ch. 19 - 19.62 (a) Deduce the structure of product A,...Ch. 19 - 19.60 Write the structures of the three products...Ch. 19 - Prob. 1LGPCh. 19 - Which hydrogen atoms in the following ester are...Ch. 19 - Prob. 2QCh. 19 - 19.3 What starting materials could be used in a...Ch. 19 - Supply the missing reagents, intermediates, and...Ch. 19 - Prob. 5QCh. 19 - Prob. 6QCh. 19 - Prob. 7QCh. 19 - Prob. 8Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
12.1 Give the IUPAC name for each of the following:
a. CH3-CH2-OH
b.
c.
d.
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
17. Anthropologists are interested in locating areas in Africa where fossils 4-8 million years old might be fou...
Campbell Biology: Concepts & Connections (9th Edition)
The air conditioner in a house or a car has a cooler that brings atmospheric air from 30Cto10C , both states at...
Fundamentals Of Thermodynamics
Choose the best answer to each of the following. Explain your reasoning. Which detection method was used by the...
Cosmic Perspective Fundamentals
The glycine cleavage system is a group of four enzymes that together catalyze the following reaction: glycine+T...
Organic Chemistry (8th Edition)
1. How many cervical, thoracic, lumbar, sacral, and coccygeal vertebrae are normally present in the vertebral ...
Human Anatomy & Physiology (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the final product when hexanedioic acid reacts with 1º PCl5 and 2º NH3.arrow_forwardWhat is the final product when D-galactose reacts with hydroxylamine?arrow_forwardIndicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.arrow_forward
- The temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forwardQUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License