
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14.2, Problem 4P
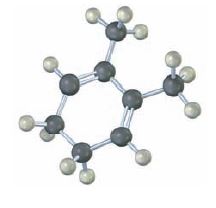
Give the structures of both 1, 2 and 1, 4 adducts resulting from reaction of 1 equivalent of HBr with the following compound:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
QUESTION: Fill in the answers in the empty green boxes
1. Step 2
2. Step 3
3. Step 4 (SUM)
4. Step 5 (df) (GIVEN)
5. Determine S y/x value
*The data values have been provided in the worksheet attached in the first image*
If the symbol A is placed in a reaction, at what temperature does it take place?
By malonic or acetylacetic synthesis, synthesize 3-methyl-4-oxopentanoic acid (indicate the formulas of the compounds).
Chapter 14 Solutions
Organic Chemistry
Ch. 14.1 - Prob. 1PCh. 14.2 - Give the structures of both 1, 2 and 1, 4 adducts...Ch. 14.2 - Prob. 3PCh. 14.2 - Give the structures of both 1, 2 and 1, 4 adducts...Ch. 14.3 - Prob. 5PCh. 14.3 - Prob. 6PCh. 14.5 - Predict the product of the following Diels–Alder...Ch. 14.5 - Prob. 8PCh. 14.5 - Which of the following dienes have an s-cis...Ch. 14.5 - Predict the product of the following Diels–Alder...
Ch. 14.6 - Prob. 11PCh. 14.6 - Prob. 12PCh. 14.7 - Prob. 13PCh. 14.7 - Prob. 14PCh. 14.8 - Which of the following compounds would you expect...Ch. 14.SE - Prob. 16VCCh. 14.SE - Show the product of the Diels–Alder reaction of...Ch. 14.SE - Prob. 18VCCh. 14.SE - Prob. 19VCCh. 14.SE - Prob. 20MPCh. 14.SE - Prob. 21MPCh. 14.SE - In light of your answer to Problem 14-21 propose...Ch. 14.SE - Luminol, which is used by forensic scientists to...Ch. 14.SE - Prob. 24MPCh. 14.SE - Give IUPAC names for the following compounds:Ch. 14.SE - Prob. 26APCh. 14.SE - Prob. 27APCh. 14.SE - Electrophilic addition of Br2 to isoprene...Ch. 14.SE - Prob. 29APCh. 14.SE - Prob. 30APCh. 14.SE - Predict the products of the following...Ch. 14.SE - 2,3-Di-tert-butyl-1,3-butadiene does not undergo...Ch. 14.SE - Prob. 33APCh. 14.SE - Prob. 34APCh. 14.SE - Prob. 35APCh. 14.SE - Prob. 36APCh. 14.SE - Rank the following dienophiles in order of their...Ch. 14.SE - Prob. 38APCh. 14.SE - Prob. 39APCh. 14.SE - Prob. 40APCh. 14.SE - Although the Diels–Alder reaction generally...Ch. 14.SE - Prob. 42APCh. 14.SE - Tires whose sidewalls are made of natural rubber...Ch. 14.SE - Prob. 44APCh. 14.SE - Prob. 45APCh. 14.SE - Prob. 46APCh. 14.SE - Would you expect allene, H2C = C = CH2, to show a...Ch. 14.SE - The following ultraviolet absorption maxima have...Ch. 14.SE - Prob. 49APCh. 14.SE - -Ocimene is a pleasant-smelling hydrocarbon found...Ch. 14.SE - Draw the resonance forms that result when the...Ch. 14.SE - Prob. 52APCh. 14.SE - Treatment of 3,4-dibromohexane with strong base...Ch. 14.SE - Prob. 54APCh. 14.SE - Prob. 55APCh. 14.SE - Prob. 56APCh. 14.SE - Prob. 57APCh. 14.SE - Prob. 58APCh. 14.SE - Hydrocarbon A, C10H14, has a UV absorption at...Ch. 14.SE - Prob. 60APCh. 14.SE - Prob. 61APCh. 14.SE - Prob. 62APCh. 14.SE - Prob. 63APCh. 14.SE - Prob. 64APCh. 14.SE - The double bond of an enamine (alkene + amine) is...Ch. 14.SE - Prob. 66AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- oalmitic acid is a 16 carbon acid. In a balanced equation, the products of the sponification of tripalmitin (glyceryl tripalmitate are blank.arrow_forwardWrite the esterification reaction mechanism of salicylic acid and acetic acid to produce aspirin (acetylsalicylic acid). Note: salicylic acid will act as the alcoholarrow_forwardWhat type of interaction would you expect between the following R groups in the tertiary structure of a protein? O -CH2-CO and -CH2-CH2-CH2-CH2-NH3+ a. disulfide bonds b. salt bridges c. hydrogen bonds HO abios vist anisinoo tedt bigil s ai loistaslor sale! 10 OUT d. hydrophobic interactions e. peptide bondsarrow_forward
- 4. True or false: This skeletal structure represents a saturated fatty acid. Ini to 0 fale) me OH faistong starrow_forwardBy malonic or acetylacetic synthesis, synthesize 5-Methyl-2-hexanone (with the formulas of the compounds).arrow_forwardQUESTION: Answer Question 5: 'Calculating standard error of regression' by filling in all the empty green boxes *The values are all provided in the first photo attached*arrow_forward
- Draw the formula for 3-chlorobenzoic acetic anhydride.arrow_forwardBy malonic or acetylacetic synthesis, synthesize 2-methylbutanoic acid (indicate the formulas of the compounds).arrow_forwardObtain 2-methylbutanoic acid by malonic or acetylacetic synthesis (indicate the formulas of the compounds involved).arrow_forward
- EFFICIENTS SAMPLE READINGS CONCENTRATIONS Pigiadient) TOMATO SAUCE (REGULAR) TOMATO (REDUCED SALT) TOMATO SAUCE (REGULAR) TOMATO (REDUCED SALT) 58 6.274 3.898 301.7 151.2 14150 5.277 3.865 348.9 254.8 B 5.136 3.639 193.7 85.9 605 4.655 3.041 308.6 199.6 05 5.135 3.664 339.5 241.4 0139 4.676 3.662 160.6 87.6 90148 5.086 3.677 337.7 242.5 0092 6.348 3.775 464.7 186.4 PART3 5.081 3.908 223.5 155.8 5.558 3.861 370.5 257.1 4.922 3.66 326.6 242.9 4.752 3.641 327.5 253.3 50 5.018 3.815 336.1 256.0 84 4.959 3.605 317.9 216.6 38 4.96 3.652 203.8 108.7 $3 5.052 3.664 329.8 239.0 17 5.043 3.767 221.9 149.7 052 5.058 3.614 331.7 236.4 5.051 4.005 211.7 152.1 62 5.047 3.637 309.6 222.7 5.298 3.977 223.4 148.7 5.38 4.24 353.7 278.2 5 5.033 4.044 334.6 268.7 995 4.706 3.621 305.6 234.4 04 4.816 3.728 340.0 262.7 16 4.828 4.496 304.3 283.2 0.011 4.993 3.865 244.7 143.6 AVERAGE STDEV COUNT 95% CI Confidence Interval (mmol/L) [Na+] (mg/100 mL) 95% Na+ Confidence Interval (mg/100 mL)arrow_forwardIf we have two compounds: acetone (CH₃COCH₃) and acetic acid (CH₃COOH), applying heat to them produces an aldol condensation of the two compounds. If this is correct, draw the formula for the final product.arrow_forwardIf we have two compounds: acetone (CH3COCH3) and acetic acid (CH3COOH); if we apply heat (A), what product(s) are obtained?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY