
Chemical Principles
8th Edition
ISBN: 9781305581982
Author: Steven S. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 26E
The allene molecule has the following Lewis structure:
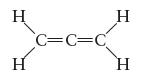
Must all four hydrogen atoms lie in the same plane? If not, what is the spatial relationship among them? Why?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
None
What are the reagents needed for this and the third structure I only got the top right structure right
Please label this COZY spectra
Chapter 14 Solutions
Chemical Principles
Ch. 14 - Prob. 1DQCh. 14 - Prob. 2DQCh. 14 - Prob. 3DQCh. 14 - Prob. 4DQCh. 14 - Prob. 5DQCh. 14 - Prob. 6DQCh. 14 - Compare and contrast the MO model with the LE...Ch. 14 - Prob. 8DQCh. 14 - Prob. 9ECh. 14 - Prob. 10E
Ch. 14 - Prob. 11ECh. 14 - Prob. 12ECh. 14 - Prob. 13ECh. 14 - Use the localized electron model to describe the...Ch. 14 - Prob. 15ECh. 14 - Use the LE model to describe the bonding in H2CO...Ch. 14 - Prob. 17ECh. 14 - The space-filling models of hydrogen cyanide and...Ch. 14 - Prob. 19ECh. 14 - Prob. 20ECh. 14 - Prob. 21ECh. 14 - Indigo is the dye used in coloring blue jeans. The...Ch. 14 - Prob. 23ECh. 14 - Prob. 24ECh. 14 - Why must all six atoms in C2H4 be in the same...Ch. 14 - The allene molecule has the following Lewis...Ch. 14 - Biacetyl and acetoin are added to margarine to...Ch. 14 - Many important compounds in the chemical industry...Ch. 14 - Prob. 29ECh. 14 - Hot and spicy foods contain molecules that...Ch. 14 - Two molecules used in the polymer industry are...Ch. 14 - Prob. 32ECh. 14 - The three most stable oxides of carbon are carbon...Ch. 14 - Prob. 34ECh. 14 - Prob. 35ECh. 14 - What are the relationships among bond order, bond...Ch. 14 - Prob. 37ECh. 14 - A Lewis structure obeying the octet rule can be...Ch. 14 - Prob. 39ECh. 14 - Why does the molecular orbital model do a better...Ch. 14 - Prob. 41ECh. 14 - Prob. 42ECh. 14 - Prob. 43ECh. 14 - In which of the following diatomic molecules would...Ch. 14 - Prob. 45ECh. 14 - Using the molecular orbital model to describe the...Ch. 14 - The transport of O2 in the blood is carried out by...Ch. 14 - Prob. 48ECh. 14 - Prob. 49ECh. 14 - Consider the following electron configuration:...Ch. 14 - Prob. 51ECh. 14 - Using an MO energy-level diagram, would you expect...Ch. 14 - Use Figs.14.45 and 14.46 to answer the following...Ch. 14 - The diatomic molecule OH exists in the gas phase....Ch. 14 - Prob. 55ECh. 14 - Describe the bonding in the O3 molecule and the...Ch. 14 - Prob. 57ECh. 14 - The space-filling model for benzoic acid is shown...Ch. 14 - Prob. 59ECh. 14 - Prob. 60ECh. 14 - The microwave spectrum of 12C16O shows that the...Ch. 14 - Prob. 62ECh. 14 - Prob. 63ECh. 14 - Prob. 64ECh. 14 - Draw the Lewis structures, predict the molecular...Ch. 14 - Prob. 66AECh. 14 - Prob. 67AECh. 14 - Prob. 68AECh. 14 - Prob. 69AECh. 14 - Prob. 70AECh. 14 - Prob. 71AECh. 14 - Prob. 72AECh. 14 - Prob. 73AECh. 14 - Vitamin B6 is an organic compound whose deficiency...Ch. 14 - Prob. 75AECh. 14 - Prob. 76AECh. 14 - Prob. 77AECh. 14 - Prob. 78AECh. 14 - Prob. 79AECh. 14 - Draw the Lewis structures for TeCl4 , ICl5 , PCl5...Ch. 14 - Prob. 81AECh. 14 - Pelargondin is the molecule responsible for the...Ch. 14 - Prob. 83AECh. 14 - Prob. 84AECh. 14 - Prob. 85AECh. 14 - Prob. 86AECh. 14 - Given that the ionization energy of F2 is...Ch. 14 - Bond energy has been defined in the text as the...Ch. 14 - a.A flask containing gaseous N2 is irradiated with...Ch. 14 - Use the MO model to determine which of the...Ch. 14 - Cholesterol (C27H46O) has the following structure:...Ch. 14 - Arrange the following from lowest to highest...Ch. 14 - Carbon monoxide (CO) forms bonds to a variety of...Ch. 14 - Prob. 94CPCh. 14 - In Exercise71 in Chapter13 , the Lewis structures...Ch. 14 - Prob. 96CPCh. 14 - Prob. 97CPCh. 14 - Prob. 98MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please label this HNMRarrow_forwardConsider the following gas chromatographs of Compound A, Compound B, and a mixture of Compounds A and B. Inject A B mixture Area= 9 Area = 5 Area = 3 Area Inject . མི། Inject J2 What is the percentage of Compound B in the the mixture?arrow_forwardRank these according to stability. CH3 H3C CH3 1 CH3 H3C 1 most stable, 3 least stable O 1 most stable, 2 least stable 2 most stable, 1 least stable O2 most stable, 3 least stable O3 most stable, 2 least stable O3 most stable, 1 least stable CH3 2 CH3 CH3 H₂C CH3 3 CH3 CHarrow_forward
- Consider this IR and NMR: INFRARED SPECTRUM TRANSMITTANCE 0.8- 0.6 0.4 0.2 3000 10 9 8 00 HSP-00-541 7 CO 6 2000 Wavenumber (cm-1) сл 5 ppm 4 M Which compound gave rise to these spectra? N 1000 1 0arrow_forwardConsider this reaction (molecular weights are under each compound): HC=CH + 2 HCI --> C2H4Cl 2 MW = 26 36.5 99 If 4.4 g of HC=CH are reacted with 110 mL of a 2.3 M HCI solution, and 6.0 g of product are actually produced, what is the percent yield?arrow_forwardWhat is the name of the major product of this reaction? OH CH3 H₂SO4, heat 1-methylcyclohexene O2-methyl-1-cyclohexene O 3-mthylcyclohexene 1-methyl-2-cyclohexenearrow_forward
- We added a brown solution of Br2 to one of our products, and the brown color disappeared. This indicated that our product wasarrow_forwardRank the following according to reactivity toward nitration: a) benzene b) bromobenzene c) nitrobenzene d) phenol Od) greatest, c) least Od) greatest, b) least Od) greatest, a) least a) greatest, b) least a) greatest, c) least Oa) greatest, d) least Ob) greatest, a) least O b) greatest, c) least Ob) greatest, d) least O c) greatest, a) least O c) greatest, b) least O c) greatest, d) leastarrow_forwardO-Nitrophenol was distilled over with the steam in our experiment while the other isomer did not. This is due to: O intramolecular hydrogen bonding in the ortho isomer O intermolecular hydrogen bonding in the the ortho isomer O the ortho isomer has a lower density O the ortho isomer has a lower molecular weightarrow_forward
- K 44% Problem 68 of 15 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. :6: :: :CI: CI CI: :0:0 Select to Add Arrows Select to Add Arrows H H Cl CI: CI CI: Select to Add Arrows Select to Add Arrows H :CI: Alarrow_forwardI I H :0: Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. 0:0 :0: CI ΑΙ :CI: :CI: :0: CI Select to Add Arrows Select to Add Arrows cl. :0: Cl © ハ CI:: CI H CO Select to Add Arrows Select to Add Arrows 10: AI ::arrow_forwardOrder the following compounds from slowest to fastest in a nucleophilic acyl substitution reaction. ii 요 OB D A E C OCE Darrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
NEET Chemistry | Group 14 Carbon Family | Theory & Problem Solving | In English | Misostudy; Author: Misostudy;https://www.youtube.com/watch?v=enOGIrcHh54;License: Standard YouTube License, CC-BY