
Concept explainers
Interpretation:
Among the given reactions, the one which gives two different halogenated hydrocarbons has to be chosen from the given options.
Concept Introduction:
In this reaction no atoms or group of atoms are removed. Instead the unsaturated bond is reduced to saturated bond. A general scheme for addition reaction of
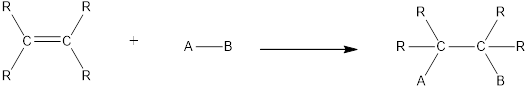
Addition reactions can be classified broadly into two types. They are asymmetrical addition reaction and symmetrical addition reaction.
Symmetrical addition reactions is the one in which the same atom or same group of atoms are added across the carbon‑carbon multiple bonds.
Unsymmetrical addition reactions is the one in which the different atom or different group of atoms are added across the carbon‑carbon multiple bonds.
Hydrohalogenation is an unsymmetrical addition reaction. In this reaction a hydrogen atom and a halogen atom are added across the multiple bond. If the alkene considered is a symmetrical one, then only one product is formed. If the alkene considered is an unsymmetrical one, then two products are formed which depends on which side of the double bond the atoms gets attached.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
GENERAL,ORGANIC,+BIO.CHEM.-MINDTAP
- Would the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forwardBelow is the SN1 reaction of (S)-3-chlorocyclohexene and hydroxide (OH). Draw the missing curved arrows, lone pairs of electrons, and nonzero formal charges. In the third box, draw the two enantiomeric products that will be produced. 5th attempt Please draw all four bonds at chiral centers. Draw the two enantiomeric products that will be produced. Draw in any hydrogen at chiral centers. 1000 4th attempt Feedback Please draw all four bonds at chiral centers. 8. R5 HO: See Periodic Table See Hint H Cl Br Jid See Periodic Table See Hintarrow_forwardShow that a molecule with configuration π4 has a cylindrically symmetric electron distribution. Hint: Let the π orbitals be equal to xf and yf, where f is a function that depends only on the distance from the internuclear axis.arrow_forward
- (a) Verify that the lattice energies of the alkali metal iodides are inversely proportional to the distances between the ions in MI (M = alkali metal) by plotting the lattice energies given below against the internuclear distances dMI. Is the correlation good? Would a better fit be obtained by plotting the lattice energies as a function of (1 — d*/d)/d, as theoretically suggested, with d* = 34.5 pm? You must use a standard graphing program to plot the graph. It generates an equation for the line and calculates a correlation coefficient. (b) From the graph obtained in (a), estimate the lattice energy of silver iodide. (c) Compare the results of (b) with the experimental value of 886 kJ/mol. If they do not agree, explain the deviation.arrow_forwardCan I please get help with #3 & 4? Thanks you so much!arrow_forwardA solution consisting of 0.200 mol methylbenzene, C,H,CH,, in 500. g of nitrobenzene, CH,NO₂, freezes at 3.2°C. Pure nitrobenzene freezes at 6.0°C. The molal freezing point constant of nitrobenzene is _ °C/m. a) 2.8 b) 3.2 c) 5.6 d) 7.0 e) 14.0arrow_forward
- Below is the SN1 reaction of (S)-3-chlorocyclohexene and hydroxide ("OH). Draw the missing curved arrows, lone pairs of electrons, and nonzero formal charges. In the third box, draw the two enantiomeric products that will be produced. 2nd attempt Please draw all four bonds at chiral centers. 0 D Draw the missing curved arrow notation. Add lone pairs of electrons and nonzero formal charges. + 노 V 1st attempt Feedback Please draw all four bonds at chiral centers. See Periodic Table See Hint F P 41 H Br See Periodic Table See Hint H Larrow_forwardHow close are the Mulliken and Pauling electronegativity scales? (a) Now that the ionization energies and electron affinities have been defined, calculate the Mulliken and Pauling electronegativities for C, N, O and F. Compare them. (Make the necessary adjustments to the values, such as dividing the ionization energies and electron affinities by 230kj/mol) (b) Plot both sets of electronegativities against atomic number (use the same graph). (c) Which scale depends most consistently on position in the Periodic Table?arrow_forwardBelow is the SN2 reaction between 2-bromopropane and iodide (I). Draw the mechanism arrows in the first box to reflect electron movements. In both boxes, add lone pairs of electrons and nonzero formal charges. 4th attempt Feedback 3rd attempt Feedback 1 -Br H :Bri :Br: ili See Periodic Table See Hint ini See Periodic Table See Hintarrow_forward
- When 4-chloro-1-butanol is placed in sodium hydride, a cyclization reaction occurs. 3rd attempt 2 HO NaH CI D Draw the curved arrow notation to form the intermediate. 4 2 H₂ See Periodic Table See Hint =arrow_forwardSketch, qualitatively, the potential energy curves of the N-N bond of N2H4, N2 and N3- graph. Explain why the energy at the minimum of each curve is not the same.arrow_forward(a) Show that the lattice energies are inversely proportional to the distance between ions in MX (M = alkali metal, X = halide ions) by plotting the lattice energies of KF, KCl, and KI against the internuclear distances, dMX. The lattice energies of KF, KCl, and KI are 826, 717, and 645 kJ/mol, respectively. Does the correlation obtained correlate well? You will need to use a standard graphing program to construct the graph (such as a spreadsheet program). It will generate an equation for the line and calculate a correlation coefficient. (b) Estimate the lattice energy of KBr from your graph. (c) Find an experimental value for the lattice energy of KBr in the literature, and compare this value with the one calculated in (b). Do they agree?arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

