
Concept explainers
(a)
Interpretation: Themolecular structure and the bond angle for HCN needs to be determined.
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(a)
Answer to Problem 95E
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in HCN:
Hence the best Lewis structure for HCN must be:
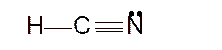
With linear stricture the bond angle must be 180°.
(b)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(b)
Answer to Problem 95E
Bond angle will be less than 120° and approximately it will be 93°.
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
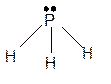
Bond angle will be less than 120° and approximately it will be 93°.
(c)
Interpretation: Interpret the molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(c)
Answer to Problem 95E
The bond angle will be 109°.
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
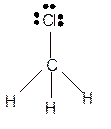
The bond angle will be 109° with tetrahedral geometry.
(d)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(d)
Answer to Problem 95E
The bond angle will be 109°.
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for

The bond angle will be 109° with tetrahedral geometry.
(e)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(e)
Answer to Problem 95E
The bond angle will be 120°.
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
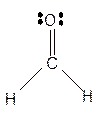
The bond angle will be 120° with trigonal planar geometry.
(f)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(f)
Answer to Problem 95E
The bond angle will be approximately 104°.
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
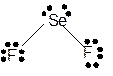
The bond angle will be 104° with bent geometry.
(g)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(g)
Answer to Problem 95E
The bond angle will be 180°.
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
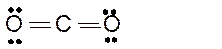
The bond angle will be 180° with linear geometry.
(h)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(h)
Answer to Problem 95E
The bond angle will be 180°.
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for

The bond angle will be 180° with linear geometry.
(i)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(i)
Answer to Problem 95E
The bond angle will be 180°.
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for

The bond angle will be 180° with linear geometry.
(j)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(j)
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
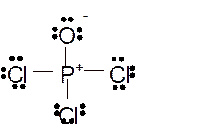
Total number of valence electrons in
Hence the best Lewis structure for
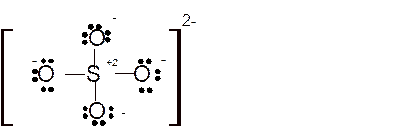
Total number of valence electrons in
Hence the best Lewis structure for
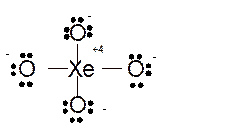
Total number of valence electrons in
Hence the best Lewis structure for
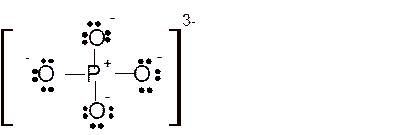
Total number of valence electrons in
Hence the best Lewis structure for
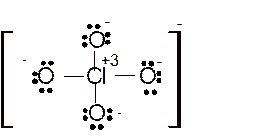
(k)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(k)
Answer to Problem 95E
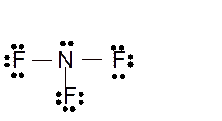
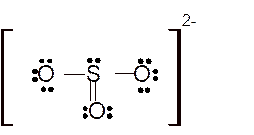
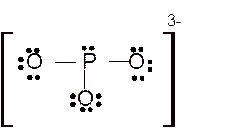
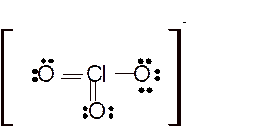
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
Total number of valence electrons in
Hence the best Lewis structure for
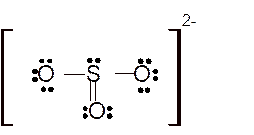
Total number of valence electrons in
Hence the best Lewis structure for
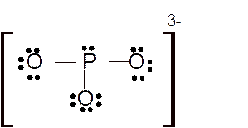
Total number of valence electrons in
Hence the best Lewis structure for
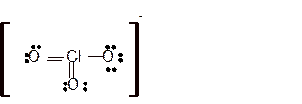
For all the above structures, bond angle will be less than 120o.
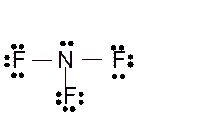
(l)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(l)
Answer to Problem 95E
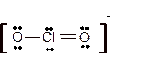
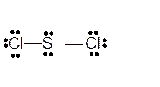
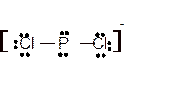
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
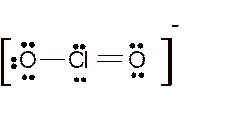
Total number of valence electrons in
Hence the best Lewis structure for
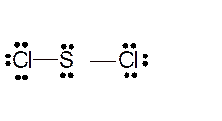
Total number of valence electrons in
Hence the best Lewis structure for
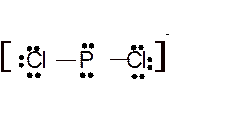
(m)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(m)
Answer to Problem 95E
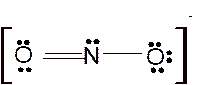
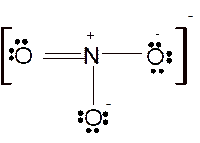
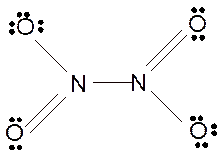
For all the above, the bond angle is approximately 120°.
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
According to the given molecular formula, the structure of
Hence the best Lewis structure for
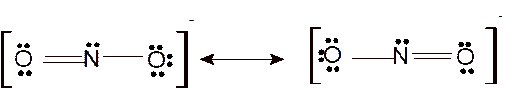
According to the given molecular formula, the structure of
Hence the best Lewis structure for
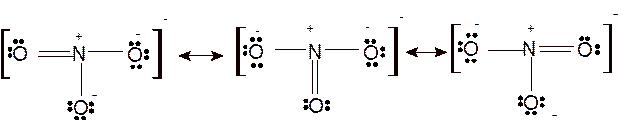
According to the given molecular formula, the structure of
Hence the best Lewis structure for
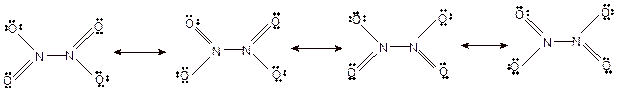
(n)
Interpretation: The molecular structure and the bond angle for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(n)
Answer to Problem 95E
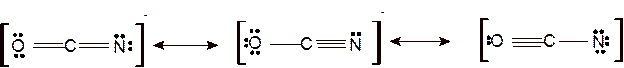
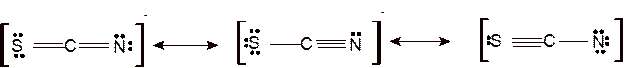
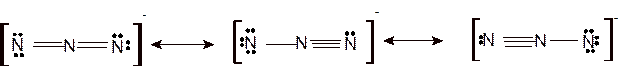
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
According to the given molecular formula, the structure of
Hence the best Lewis structure for
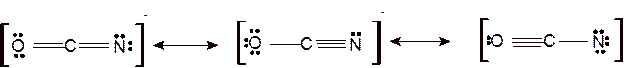
According to the given molecular formula, the structure of
Hence the best Lewis structure for
According to the given molecular formula, the structure of
Hence the best Lewis structure for
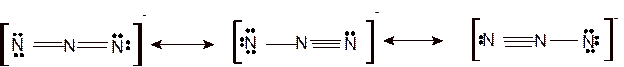
Since all are linear ions therefore the bond angle must be 180°.
Want to see more full solutions like this?
Chapter 13 Solutions
EBK CHEMICAL PRINCIPLES
- Indicate the differences between the spectra of 1-methyl-benzimidazole and benzimidazole.arrow_forwardProvide reasons as to why appropriate sampling is important in relation to food?arrow_forwardWhat is the significance of selecting a "representative" sample for chemical analysis, and how does this practice ensure accurate and reliable results with respect to chemical analyses?arrow_forward
- Identify and provide an explanation of the differences between homogeneous and heterogeneous sampling in the context of sampling methods.arrow_forwardГ C-RSA CHROMATOPAC CH=1 DATA 1: @CHRM1.C00 ATTEN=10 SPEED= 10.0 0.0 b.092 0.797 1.088 1.813 C-RSA CHROMATOPAC CH=1 Report No. =13 ** CALCULATION REPORT ** DATA=1: @CHRM1.000 11/03/05 08:09:52 CH PKNO TIME 1 2 0.797 3 1.088 4 1.813 AREA 1508566 4625442 2180060 HEIGHT 207739 701206 V 287554 V MK IDNO CONC NAME 18.1447 55.6339 26.2213 TOTAL 8314067 1196500 100 C-R8A CHROMATOPAC CH=1 DATA 1: @CHRM1.C00 ATTEN=10 SPEED= 10.0 0. 0 087 337. 0.841 1.150 C-R8A CHROMATOPAC CH=1 Report No. =14 DATA=1: @CHRM1.000 11/03/05 08:12:40 ** CALCULATION REPORT ** CH PKNO TIME AREA 1 3 0.841 1099933 41.15 4039778 HEIGHT MK IDNO 170372 649997¯¯¯ CONC NAME 21.4007 78.5993 TOTAL 5139711 820369 100 3 C-R8A CHROMATOPAC CH=1 DATA 1: @CHRM1.C00 ATTEN=10 SPEED= 10.0 0.100 0:652 5.856 3 1.165 C-RSA CHROMATOPAC CH-1 Report No. =15 DATA=1: @CHRM1.000 11/03/05 08:15:26 ** CALCULATION REPORT ** CH PKNO TIME AREA HEIGHT MK IDNO CONC NAME 1 3 3 0.856 4 1.165 TOTAL 1253386 4838738 175481 708024 V 20.5739 79.4261 6092124…arrow_forwardDraw the product of the reaction shown below. Ignore small byproducts that would evaporate please.arrow_forward
- Relative Abundance 20- Problems 501 (b) The infrared spectrum has a medium-intensity peak at about 1650 cm. There is also a C-H out-of-plane bending peak near 880 cm. 100- 80- 56 41 69 M(84) LL 15 20 25 30 35 55 60 65 70 75 80 85 90 m/zarrow_forwardPolyethylene furanoate is a polymer made from plant-based sources; it is used for packaging. Identify the monomer(s) used in the production of this polymer using a condensation process.arrow_forwardPhenol is the starting material for the synthesis of 2,3,4,5,6-pentachlorophenol, known al-ternatively as pentachlorophenol, or more simply as penta. At one time, penta was widely used as a wood preservative for decks, siding, and outdoor wood furniture. Draw the structural formula for pentachlorophenol and describe its synthesis from phenol.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





