
General, Organic, and Biological Chemistry
7th Edition
ISBN: 9781285853918
Author: H. Stephen Stoker
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.77EP
Supply the structural formula of the product in each of the following
- a. CH3—CH=CH—CH3 + Cl2 → ?
- b. CH3—CH—CH=CH2 + HCl → ?
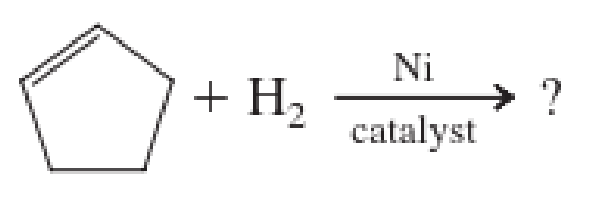
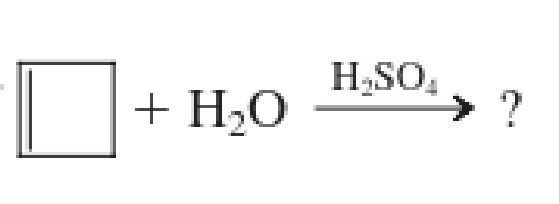
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
bre
The reaction sequence shown in Scheme 5 demonstrates the synthesis of a
substituted benzene derivative Q.
wolsd works 2
NH2
NaNO2, HCI
(apexe) 13× (1
HNO3, H2SO4
C6H5CIN2
0°C
HOTE
CHINO₂
N
O
*O₂H (
PO
Q
Я
Scheme 5
2 bag abouoqmics to sounde odi WEIC
(i)
Draw the structure of intermediate O.
[2 marks]
to noitsmot od: tot meinedogm, noit so oft listsb ni zaupaib bas wa
(ii) Draw the mechanism for the transformation of aniline N to intermediate O.
Spoilage
(b)
[6 marks]
(iii) Identify the reagent X used to convert compound O to the iodinated compound
[tom E
P.
vueimado oilovonsa ni moitos nolisbnolov ayd toes ai tedw nisiqx
(iv) Identify the possible structures of compound Q.
[2 marks]
[2 marks]
[shom 2]
(v)
bus noires goiribbeolovo xnivollot adj to subora sidab
Draw the mechanism for the transformation of intermediate P to compound Q.
[5 marks]
vi
(vi) Account for the regiochemical outcome observed in the reaction forming
compound Q.
[3 marks]
PROBLEM 4 Solved
Show how 1-butanol can be converted into the following compounds:
a.
PROBLEM 5+
b.
d.
-C=
N
Which alkene is the major product of this dehydration?
OH
H2SO4
heat
Chapter 13 Solutions
General, Organic, and Biological Chemistry
Ch. 13.1 - Prob. 1QQCh. 13.1 - Prob. 2QQCh. 13.1 - Prob. 3QQCh. 13.2 - Prob. 1QQCh. 13.2 - Prob. 2QQCh. 13.2 - Prob. 3QQCh. 13.2 - Prob. 4QQCh. 13.3 - Prob. 1QQCh. 13.3 - Prob. 2QQCh. 13.3 - Prob. 3QQ
Ch. 13.3 - Prob. 4QQCh. 13.4 - Prob. 1QQCh. 13.4 - Prob. 2QQCh. 13.5 - Prob. 1QQCh. 13.5 - Prob. 2QQCh. 13.5 - Prob. 3QQCh. 13.6 - Prob. 1QQCh. 13.6 - Prob. 2QQCh. 13.6 - Prob. 3QQCh. 13.7 - Prob. 1QQCh. 13.7 - Prob. 2QQCh. 13.7 - Prob. 3QQCh. 13.8 - Prob. 1QQCh. 13.8 - Prob. 2QQCh. 13.9 - Prob. 1QQCh. 13.9 - Prob. 2QQCh. 13.10 - Prob. 1QQCh. 13.10 - Prob. 2QQCh. 13.10 - Prob. 3QQCh. 13.10 - Prob. 4QQCh. 13.10 - Prob. 5QQCh. 13.11 - Prob. 1QQCh. 13.11 - Prob. 2QQCh. 13.11 - Prob. 3QQCh. 13.11 - Prob. 4QQCh. 13.11 - Prob. 5QQCh. 13.12 - Prob. 1QQCh. 13.12 - Prob. 2QQCh. 13.12 - Prob. 3QQCh. 13.12 - Prob. 4QQCh. 13.12 - Prob. 5QQCh. 13.13 - Prob. 1QQCh. 13.13 - Prob. 2QQCh. 13.13 - Prob. 3QQCh. 13.14 - Prob. 1QQCh. 13.14 - Prob. 2QQCh. 13.14 - Prob. 3QQCh. 13.14 - Prob. 4QQCh. 13.15 - Prob. 1QQCh. 13.15 - Prob. 2QQCh. 13.15 - Prob. 3QQCh. 13.15 - Prob. 4QQCh. 13.16 - Prob. 1QQCh. 13.16 - Prob. 2QQCh. 13 - Classify each of the following hydrocarbons as...Ch. 13 - Classify each of the following hydrocarbons as...Ch. 13 - Prob. 13.3EPCh. 13 - Prob. 13.4EPCh. 13 - Prob. 13.5EPCh. 13 - Prob. 13.6EPCh. 13 - Prob. 13.7EPCh. 13 - Prob. 13.8EPCh. 13 - Prob. 13.9EPCh. 13 - What is the molecular formula for each of the...Ch. 13 - Prob. 13.11EPCh. 13 - Prob. 13.12EPCh. 13 - What is wrong, if anything, with the following...Ch. 13 - Prob. 13.14EPCh. 13 - Prob. 13.15EPCh. 13 - Prob. 13.16EPCh. 13 - Prob. 13.17EPCh. 13 - Prob. 13.18EPCh. 13 - Draw a condensed structural formula for each of...Ch. 13 - Draw a condensed structural formula for each of...Ch. 13 - The following names are incorrect by IUPAC rules....Ch. 13 - The following names are incorrect by IUPAC rules....Ch. 13 - Prob. 13.23EPCh. 13 - Prob. 13.24EPCh. 13 - Prob. 13.25EPCh. 13 - Classify each of the following compounds as...Ch. 13 - Prob. 13.27EPCh. 13 - How many hydrogen atoms are present in a molecule...Ch. 13 - Prob. 13.29EPCh. 13 - Draw a line-angle structural formula for each of...Ch. 13 - Prob. 13.31EPCh. 13 - Prob. 13.32EPCh. 13 - Prob. 13.33EPCh. 13 - Prob. 13.34EPCh. 13 - Prob. 13.35EPCh. 13 - Prob. 13.36EPCh. 13 - Prob. 13.37EPCh. 13 - Prob. 13.38EPCh. 13 - For each of the following pairs of alkenes,...Ch. 13 - Prob. 13.40EPCh. 13 - Prob. 13.41EPCh. 13 - Prob. 13.42EPCh. 13 - Prob. 13.43EPCh. 13 - Prob. 13.44EPCh. 13 - Prob. 13.45EPCh. 13 - Prob. 13.46EPCh. 13 - For each molecule, indicate whether cistrans...Ch. 13 - For each molecule, indicate whether cistrans...Ch. 13 - Prob. 13.49EPCh. 13 - Prob. 13.50EPCh. 13 - Prob. 13.51EPCh. 13 - Draw a structural formula for each of the...Ch. 13 - Prob. 13.53EPCh. 13 - Prob. 13.54EPCh. 13 - Prob. 13.55EPCh. 13 - Prob. 13.56EPCh. 13 - Prob. 13.57EPCh. 13 - Prob. 13.58EPCh. 13 - Why is the number of carbon atoms in a terpene...Ch. 13 - How many isoprene units are present in a....Ch. 13 - Prob. 13.61EPCh. 13 - Indicate whether each of the following statements...Ch. 13 - Prob. 13.63EPCh. 13 - With the help of Figure 13-7, indicate whether...Ch. 13 - Prob. 13.65EPCh. 13 - Prob. 13.66EPCh. 13 - Prob. 13.67EPCh. 13 - Prob. 13.68EPCh. 13 - Prob. 13.69EPCh. 13 - Prob. 13.70EPCh. 13 - Prob. 13.71EPCh. 13 - Prob. 13.72EPCh. 13 - Prob. 13.73EPCh. 13 - Prob. 13.74EPCh. 13 - Prob. 13.75EPCh. 13 - Write a chemical equation showing reactants,...Ch. 13 - Supply the structural formula of the product in...Ch. 13 - Prob. 13.78EPCh. 13 - What reactant would you use to prepare each of the...Ch. 13 - Prob. 13.80EPCh. 13 - Prob. 13.81EPCh. 13 - Prob. 13.82EPCh. 13 - Prob. 13.83EPCh. 13 - Prob. 13.84EPCh. 13 - Prob. 13.85EPCh. 13 - Prob. 13.86EPCh. 13 - Prob. 13.87EPCh. 13 - Prob. 13.88EPCh. 13 - Prob. 13.89EPCh. 13 - Prob. 13.90EPCh. 13 - Prob. 13.91EPCh. 13 - Prob. 13.92EPCh. 13 - Prob. 13.93EPCh. 13 - What are the bond angles about the triple bond in...Ch. 13 - Prob. 13.95EPCh. 13 - Prob. 13.96EPCh. 13 - Prob. 13.97EPCh. 13 - Prob. 13.98EPCh. 13 - Prob. 13.99EPCh. 13 - Prob. 13.100EPCh. 13 - Prob. 13.101EPCh. 13 - Prob. 13.102EPCh. 13 - Prob. 13.103EPCh. 13 - Prob. 13.104EPCh. 13 - Prob. 13.105EPCh. 13 - Prob. 13.106EPCh. 13 - Prob. 13.107EPCh. 13 - Prob. 13.108EPCh. 13 - Assign each of the compounds in Problem 13-107 an...Ch. 13 - Assign each of the compounds in Problem 13-108 an...Ch. 13 - Prob. 13.111EPCh. 13 - Prob. 13.112EPCh. 13 - Prob. 13.113EPCh. 13 - Prob. 13.114EPCh. 13 - Prob. 13.115EPCh. 13 - Write a structural formula for each of the...Ch. 13 - Eight isomeric substituted benzenes have the...Ch. 13 - Prob. 13.118EPCh. 13 - Prob. 13.119EPCh. 13 - Prob. 13.120EPCh. 13 - Prob. 13.121EPCh. 13 - Prob. 13.122EPCh. 13 - Prob. 13.123EPCh. 13 - Prob. 13.124EPCh. 13 - Prob. 13.125EPCh. 13 - For each of the following classes of compounds,...Ch. 13 - Prob. 13.127EPCh. 13 - Prob. 13.128EPCh. 13 - Prob. 13.129EPCh. 13 - Prob. 13.130EP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please correct answer and don't used hand raiting and don't used Ai solutionarrow_forwardPlease correct answer and don't used hand raitingarrow_forwardThe vibrational contribution isa) temperature independent for internal energy and heat capacityb) temperature dependent for internal energy and heat capacityc) temperature independent for heat capacityd) temperature independent for internal energyarrow_forward
- Quantum mechanics. Explain the basis of approximating the summation to an integral in translational motion.arrow_forwardQuantum mechanics. In translational motion, the summation is replaced by an integral when evaluating the partition function. This is correct becausea) the spacing of the translational energy levels is very small compared to the product kTb) the spacing of the translational energy levels is comparable to the product kTc) the spacing of the translational energy levels is very large compared to the product kTarrow_forwardDon't used Ai solutionarrow_forward
- Please correct answer and don't used hand raiting don't used Ai solutionarrow_forwardIf the viscosity of hydrogen gas (at 0oC and 1 atm) is 8.83x10-5 P. If we assume that the molecular sizes are equal, calculate the viscosity of a gas composed of deuterium.arrow_forwardIf the viscosity of hydrogen gas (at 0oC and 1 atm) is 8.83x10-5 P. If we assume that the molecular sizes are equal, calculate the viscosity of a gas composed of deuterium.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License