
(a)
Interpretation:
The electron dot formula and structural formula of
Concept introduction:
An electron dot formula is a way of representing the molecular structure in which electrons are represented by a dot. Structural formula is a way in which atoms are linked together through a solid line. This solid line represents the covalent bond. An electron dot structure is known as Lewis structure. Electron dot structure indicates the valence electrons of an atom which are involved in bonding.
Answer to Problem 38E
Electron dot structure of
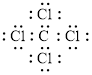
The structural formula of
Explanation of Solution
In a molecule
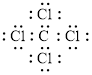
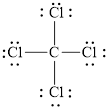
Figure 1

Figure 2
Each solid line, in Figure 2, between the carbon and chlorine atom is the covalent bond which is made up of two electrons. This bond is formed by sharing of electrons between the central atom carbon and the surrounding chlorine atom.
An electron dot structure and structural formula of
(b)
Interpretation:
The electron dot formula and structural formula of
Concept introduction:
An electron dot formula is a way of representing the molecular structure in which electrons are represented by a dot. Structural formula is a way in which atoms are linked together through a solid line. This solid line represents the covalent bond. An electron dot structure is known as Lewis structure. Electron dot structure indicates the valence electrons of an atom which are involved in bonding.
Answer to Problem 38E
Electron dot structure of
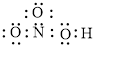
The structural formula of
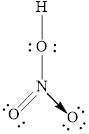
Explanation of Solution
In molecule
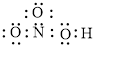
Figure 3

Figure 4
Each solid line, in Figure 4, between the nitrogen and oxygen atom is the covalent bond which is made up of two electrons. This bond is formed by sharing of electrons between the central atom nitrogen and the surrounding oxygen atom. Bond between oxygen and hydrogen is also a covalent bond. Bond between nitrogen and oxygen with red arrow is the coordinate bond. This shows the sharing of nitrogen electrons between the oxygen and the nitrogen.
An electron dot structure and structural formula of
(c)
Interpretation:
The electron dot formula and structural formula of
Concept introduction:
An electron dot formula is a way of representing the molecular structure in which electrons are represented by a dot. Structural formula is a way in which atoms are linked together through a solid line. This solid line represents the covalent bond. An electron dot structure is known as Lewis structure. Electron dot structure indicates the valence electrons of an atom which are involved in bonding.
Answer to Problem 38E
Electron dot structure
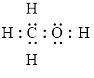
The structural formula of
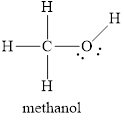
Explanation of Solution
In molecule
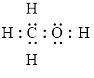
Figure 5

Figure 6
Each solid line, in Figure 6, between the carbon-hydrogen, carbon-oxygen and oxygen –hydrogen is the covalent bond which is made up of two electrons. This bond is formed by sharing of electrons between the two atoms present in that bond. Lone pair of oxygen is shown with
An electron dot structure and structural formula of
(d)
Interpretation:
The electron dot formula and structural formula of
Concept introduction:
An electron dot formula is a way of representing the molecular structure in which electrons are represented by a dot. Structural formula is a way in which atoms are linked together through a solid line. This solid line represents the covalent bond. An electron dot structure is known as lewis structure. Electron dot structure indicates the valence electrons of an atom which are involved in bonding.
Answer to Problem 38E
Electron dot structure of
![]()
The structural formula of
![]()
Explanation of Solution
In a molecule
![]()
Figure 7
![]()
Figure 8
Each solid line, in Figure 8, between the oxygen-hydrogen, carbon-oxygen and carbon –nitrogen is the covalent bond which is made up of two electrons. This bond is formed by sharing of electrons between the two atoms present in that bond. Lone pair of oxygen and nitrogen is shown with
An electron dot structure and structural formula of
Want to see more full solutions like this?
Chapter 12 Solutions
Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- Can I please get help with this.arrow_forwardDetermine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. N₂H₅ClO₄arrow_forwardPlease help me with identifying these.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





