
General Chemistry: Atoms First
2nd Edition
ISBN: 9780321809261
Author: John E. McMurry, Robert C. Fay
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11.6, Problem 11.15CP
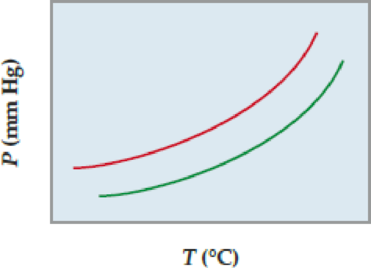
The following diagram shows a close-up view of part of the vapor-pressure curve for a pure solvent and a solution of a nonvolatile solute. Which curve represents the pure solvent, and which the solution?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
b.
H3C
Br
-CEN
H3C
Draw the most likely mechanism for the following
Draw the most likely mechanism for the following
Chapter 11 Solutions
General Chemistry: Atoms First
Ch. 11.2 - Arrange the following compounds in order of their...Ch. 11.2 - Which would you expect to have the larger (more...Ch. 11.3 - Prob. 11.3PCh. 11.3 - Prob. 11.4PCh. 11.3 - Prob. 11.5PCh. 11.3 - Prob. 11.6PCh. 11.3 - Prob. 11.7PCh. 11.3 - Prob. 11.8PCh. 11.3 - Prob. 11.9PCh. 11.3 - Prob. 11.10P
Ch. 11.4 - Prob. 11.11PCh. 11.4 - Prob. 11.12PCh. 11.6 - Prob. 11.13PCh. 11.6 - How many grams of NaBr must be added to 250 g of...Ch. 11.6 - The following diagram shows a close-up view of...Ch. 11.6 - (a) What is the vapor pressure in mm Hg of a...Ch. 11.6 - The following phase diagram shows part of the...Ch. 11.7 - What is the normal boiling point in C of a...Ch. 11.7 - What is the freezing point in C of a solution...Ch. 11.7 - Assuming complete dissociation, what is the...Ch. 11.7 - When 9.12 g of HCl was dissolved in 190 g of...Ch. 11.7 - The following phase diagram shows a close-up view...Ch. 11.8 - What osmotic pressure in atmospheres would you...Ch. 11.8 - A solution of an unknown substance in water at 300...Ch. 11.9 - A solution of 0.250 g of naphthalene (mothballs)...Ch. 11.9 - What is the molar mass of sucrose (table sugar) if...Ch. 11.10 - What is the difference between a dialysis membrane...Ch. 11 - The following phase diagram shows part of the...Ch. 11 - Prob. 11.30CPCh. 11 - The following diagram shows a close-up view of...Ch. 11 - Prob. 11.32CPCh. 11 - Prob. 11.33CPCh. 11 - Prob. 11.34CPCh. 11 - A phase diagram of temperature versus composition...Ch. 11 - The following phase diagram shows a very small...Ch. 11 - Prob. 11.38SPCh. 11 - Prob. 11.39SPCh. 11 - Prob. 11.40SPCh. 11 - Prob. 11.41SPCh. 11 - Prob. 11.42SPCh. 11 - Prob. 11.43SPCh. 11 - Prob. 11.44SPCh. 11 - Prob. 11.45SPCh. 11 - Prob. 11.46SPCh. 11 - Prob. 11.47SPCh. 11 - Prob. 11.48SPCh. 11 - Prob. 11.49SPCh. 11 - Prob. 11.50SPCh. 11 - Prob. 11.51SPCh. 11 - Prob. 11.52SPCh. 11 - Which of the following solutions has the higher...Ch. 11 - What is the mass percent concentration of the...Ch. 11 - Prob. 11.55SPCh. 11 - Prob. 11.56SPCh. 11 - Prob. 11.57SPCh. 11 - Prob. 11.58SPCh. 11 - Prob. 11.59SPCh. 11 - Prob. 11.60SPCh. 11 - Prob. 11.61SPCh. 11 - Prob. 11.62SPCh. 11 - Prob. 11.63SPCh. 11 - Prob. 11.64SPCh. 11 - Prob. 11.65SPCh. 11 - Prob. 11.66SPCh. 11 - Prob. 11.67SPCh. 11 - Prob. 11.68SPCh. 11 - Look at the solubility graph in Figure 11.6, and...Ch. 11 - Vinyl chloride (H2CCHCl), the starting material...Ch. 11 - Prob. 11.71SPCh. 11 - Prob. 11.72SPCh. 11 - Prob. 11.73SPCh. 11 - Sulfur hexafluoride, which is used as a...Ch. 11 - Prob. 11.75SPCh. 11 - Prob. 11.76SPCh. 11 - Prob. 11.77SPCh. 11 - Draw a phase diagram showing how the phase...Ch. 11 - Prob. 11.79SPCh. 11 - Prob. 11.80SPCh. 11 - Prob. 11.81SPCh. 11 - What is the boiling point in C of each of the...Ch. 11 - Prob. 11.83SPCh. 11 - Prob. 11.84SPCh. 11 - The vant Hoff factor for KCl is i = 1.85. What is...Ch. 11 - Heptane (C7H16) and octane (C8H18) are...Ch. 11 - Prob. 11.87SPCh. 11 - Acetone, C3H6O, and ethyl acetate, C4H8O2, are...Ch. 11 - The industrial solvents chloroform, CHCl3, and...Ch. 11 - What is the mole fraction of each component in the...Ch. 11 - Prob. 11.91SPCh. 11 - Prob. 11.92SPCh. 11 - Prob. 11.93SPCh. 11 - A solution of citric acid, C6H8O7, in 50.0 g of...Ch. 11 - What is the normal boiling point in C of ethyl...Ch. 11 - Prob. 11.96SPCh. 11 - Prob. 11.97SPCh. 11 - Prob. 11.98SPCh. 11 - Prob. 11.99SPCh. 11 - When salt is spread on snow-covered roads at 2 C,...Ch. 11 - Prob. 11.101SPCh. 11 - Prob. 11.102SPCh. 11 - Prob. 11.103SPCh. 11 - Prob. 11.104SPCh. 11 - Prob. 11.105SPCh. 11 - Prob. 11.106SPCh. 11 - Prob. 11.107SPCh. 11 - Prob. 11.108CHPCh. 11 - Prob. 11.109CHPCh. 11 - Prob. 11.110CHPCh. 11 - Silver chloride has a solubility of 0.007 mg/mL in...Ch. 11 - Prob. 11.112CHPCh. 11 - Prob. 11.113CHPCh. 11 - Prob. 11.114CHPCh. 11 - Prob. 11.115CHPCh. 11 - Prob. 11.116CHPCh. 11 - Prob. 11.117CHPCh. 11 - Rubbing alcohol is a 90 mass % solution of...Ch. 11 - Prob. 11.119CHPCh. 11 - Prob. 11.120CHPCh. 11 - What is the vant Hoff factor for K2SO4 in an...Ch. 11 - If the vant Hoff factor for LiCl in a 0.62 m...Ch. 11 - Prob. 11.123CHPCh. 11 - Prob. 11.124CHPCh. 11 - Many acids are partially dissociated into ions in...Ch. 11 - Prob. 11.126CHPCh. 11 - Prob. 11.127CHPCh. 11 - A solution of LiCl in a mixture of water and...Ch. 11 - Prob. 11.129CHPCh. 11 - Prob. 11.130CHPCh. 11 - Prob. 11.131CHPCh. 11 - Prob. 11.132CHPCh. 11 - Prob. 11.133CHPCh. 11 - Prob. 11.134CHPCh. 11 - Prob. 11.135CHPCh. 11 - A solution prepared by dissolving 100.0 g of a...Ch. 11 - Treatment of 1.385 g of an unknown metal M with an...Ch. 11 - A compound that contains only C and H was burned...Ch. 11 - Prob. 11.139MPCh. 11 - When 8.900 g of a mixture of an alkali metal...Ch. 11 - Prob. 11.141MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY