
Concept explainers
Convert each shorthand structure to a complete structure with all atoms and lone pairs drawn in.
-
a.
b.
c.
d. 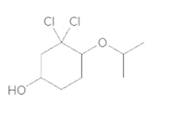
(a)
Interpretation:
To convert the following shorthand structure to complete structure with all atoms and lone pairs drawn.
Concept Introduction:
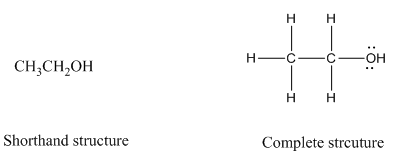
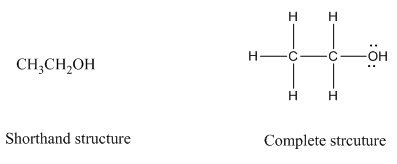
Complete structure of a compound is the one in which all the bonds, atoms and lone pairs are shown. Example of complete structure is given below.
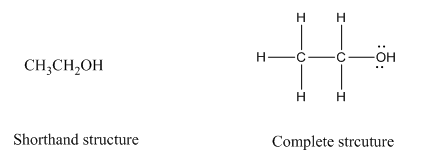
In shorthand structure, bond between the atoms and lone pairs of atoms are not shown. But, in complete structure, all the bonds between atoms and lone pairs are shown.
Answer to Problem 37P
Complete structure of the compound is,
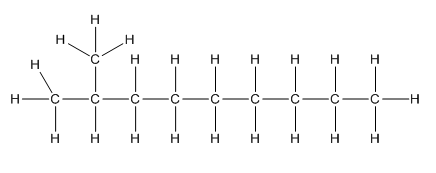
Explanation of Solution
Given compound is as follows:
To −CH group two methyl
(b)
Interpretation:
To convert the following shorthand structure to complete structure with all atoms and lone pairs drawn.
Concept Introduction:
Complete structure of a compound is the one in which all the bonds, atoms and lone pairs are shown. Example of complete structure is given below.
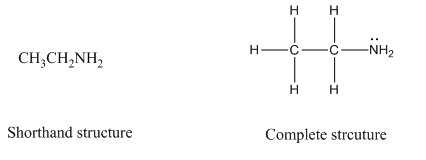
In shorthand structure, bond between the atoms and lone pairs of atoms are not shown. But, in complete structure, all the bonds between atoms and lone pairs are shown.
Answer to Problem 37P
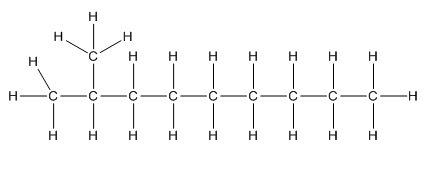
Complete structure of the compound is as follows:
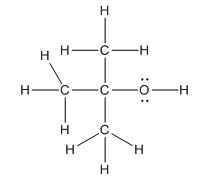
Explanation of Solution
In the compound
Hence, complete structure of the compound is as follows:
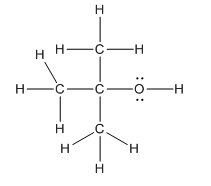
(c)
Interpretation:
To convert the following shorthand structure to complete structure with all atoms and lone pairs drawn.
Concept Introduction:
Complete structure of a compound is the one in which all the bonds, atoms and lone pairs are shown. Example of complete structure is given below.
In shorthand structure, bond between the atoms and lone pairs of atoms are not shown. But, in complete structure, all the bonds between atoms and lone pairs are shown.
Answer to Problem 37P
Complete structure of the compound is as follows:
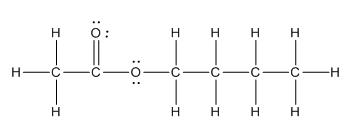
Explanation of Solution
Given compound is as follows:
In the complete structure, all the bonds between atoms and lone pairs are to be shown. Two oxygen atoms present. Both the oxygen atoms have two lone pairs (as the total number of valence electron of O is 6 and out of these 2 are involved in bond formation) on it. Hence, complete structure of the compound is as follows:
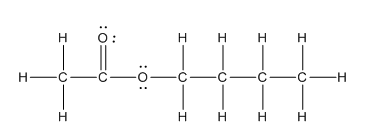
(d)
Interpretation:
To convert the following shorthand structure to complete structure with all atoms and lone pairs drawn.
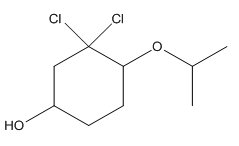
Concept Introduction:
Complete structure of a compound is the one in which all the bonds, atoms and lone pairs are shown. Example of complete structure is given below.
In shorthand structure, bond between the atoms and lone pairs of atoms are not shown. But, in complete structure, all the bonds between atoms and lone pairs are shown.
Answer to Problem 37P
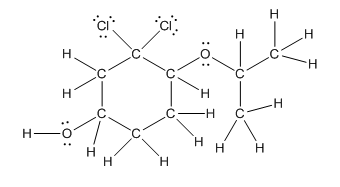
Complete structure is as follows:
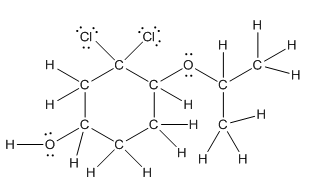
Explanation of Solution
Given compound is as follows:
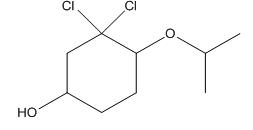
In complete structure, all the carbon atoms, hydrogen atoms and all the lone pairs should be shown. In the compound two chlorine atoms (as the total number of valence electron of Cl is 7 and out of these 1 is involved in bond formation), one oxygen atom and one −OH group(as the total number of valence electron of O is 6 and out of these 2 are involved in bond formation) present which has lone pairs. So, complete structure is as follows:
Want to see more full solutions like this?
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Synthesis of Ibuprofen-Part 2: 1. Some pain relievers including ibuprofen (MotrinⓇ) and naproxen (Aleve®) are "α-arylpropanoic acids." Look up the structure of naproxen (AleveⓇ), another a-arylpropionic acid. Using the same reactions that we used for making ibuprofen, show how to make naproxen from the compound below. Show all intermediates and reagents in your synthesis. Show how you would prepare ibuprofen starting from p-isobutylbenzene rather than p-isobutylacetophenenone. What reaction steps would need to change/add? 3. What signals appeared/disappeared/shifted that indicate that you have your intended product and not starting material? What other impurities are present in your product and how do you know?arrow_forwardAcid Catalyzed Aromatization of Carvone: 1. Starting with the ketone, below, draw a mechanism for the reaction to give the phenol as shown. H2SO4 HO- H₂O 2. Why do we use CDCl instead of CHCl, for acquiring our NMR spectra? 3. Why does it not matter which enantiomer of carvone is used for this reaction? What signals appeared/disappeared/shifted that indicate that you have your intended product and not starting material? What other impurities are present in your product and how do you know?arrow_forwardAssign this H NMRarrow_forward
- Please complete these blanks need that asaparrow_forwardNitration of Methyl Benzoate: 1. Predict the major product for the reaction below AND provide a mechanism. Include ALL resonance structures for the intermediate. C(CH3)3 NO₂* ? 2. Assuming the stoichiometry is 1:1 for the reaction above, what volume of concentrated nitric acid would be required to mononitrate 0.50 grams of the compound above? What product(s) might you expect if you nitrated phenol instead of methyl benzoate? Explain your reasoning. What signals appeared/disappeared/shifted that indicate that you have your intended product and not starting material? What other impurities are present in your product and how do you know?arrow_forwardSodium Borohydride Reduction (continued on the next page): 1. Draw the product of each of the reactions below and give the formula mass to the nearest whole number. ? (1) NaBH (2) acid (1) NaBD4 (2) acid ? 2. In mass spectra, alcohols typically break as shown in equation 8 in chapter 11 (refer to your lab manual). The larger group is generally lost and this gives rise to the base peak in the mass spectrum. For the products of each of the reactions in question # 1, draw the ion corresponding to the base peak for that product and give its mass to charge ratio (m/z). 3. Given the reaction below, calculate how many mg of 1-phenyl-1-butanol that can be produced using 31 mg NaBH4 and an excess of butyrophenone. 4. + NaBH4 OH (after workup with dilute HCI) What signals appeared/disappeared/shifted that indicate that you have your intended product and not starting material? What other impurities are present in your product and how do you know?arrow_forward
- Aspirin from Wintergreen: 1. In isolating the salicylic acid, why is it important to press out as much of the water as possible? Write a step-by-step mechanism for the esterification of salicylic acid with acetic anhydride catalyzed by concentrated H₂SO4. 3. Calculate the exact monoisotopic mass of aspirin showing your work. What signals appeared/disappeared/shifted that indicate that you have your intended product and not starting material? What other impurities are present in your product and how do you know?arrow_forwardSynthesis of Ibuprofen-Part 1: 1. What characteristic absorption band changes would you expect in the IR spectrum on going from p-isobutylacetophenone to 1-(4-isobutylphenyl)-ethanol and then to 1-(4-isobutylphenyl)-1-choroethane as you did in the experiment today? Give approximate wavenumbers associated with each functional group change. Given that the mechanism of the chlorination reaction today involves formation of a benzylic carbocation, explain why the following rearranged product is not formed. محرم محمد 3. Why do we use dilute HCl for the first step of the reaction today and concentrated HCI for the second step? What signals appeared/disappeared/shifted that indicate that you have your intended product and not starting material? What other impurities are present in your product and how do you know?arrow_forwardAssign only the C NMRarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


