
General, Organic, and Biological Chemistry
7th Edition
ISBN: 9781285853918
Author: H. Stephen Stoker
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 9, Problem 9.94EP
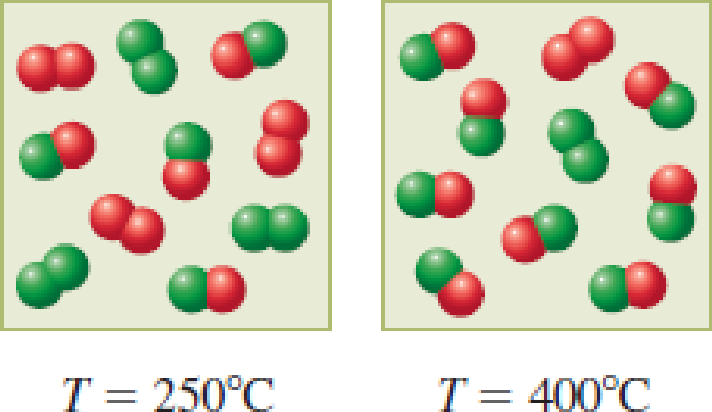
The following two diagrams represent the composition of an equilibrium mixture for the reaction A2 + B2 → 2AB at two different temperatures. Based on the diagrams, is the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
General, Organic, and Biological Chemistry
Ch. 9.1 - Prob. 1QQCh. 9.1 - Prob. 2QQCh. 9.1 - Prob. 3QQCh. 9.2 - The proper assignment of oxidation numbers to the...Ch. 9.2 - The proper assignment of oxidation numbers to the...Ch. 9.2 - Prob. 3QQCh. 9.3 - Prob. 1QQCh. 9.3 - Prob. 2QQCh. 9.3 - Prob. 3QQCh. 9.3 - Prob. 4QQ
Ch. 9.3 - Prob. 5QQCh. 9.4 - Prob. 1QQCh. 9.4 - Prob. 2QQCh. 9.4 - Prob. 3QQCh. 9.5 - Prob. 1QQCh. 9.5 - Prob. 2QQCh. 9.5 - For endothermic chemical reactions the energy...Ch. 9.6 - Prob. 1QQCh. 9.6 - Prob. 2QQCh. 9.6 - Prob. 3QQCh. 9.7 - Prob. 1QQCh. 9.7 - Prob. 2QQCh. 9.7 - Prob. 3QQCh. 9.8 - Which of the following is the correct equilibrium...Ch. 9.8 - Prob. 2QQCh. 9.8 - Prob. 3QQCh. 9.9 - Prob. 1QQCh. 9.9 - Prob. 2QQCh. 9.9 - Prob. 3QQCh. 9.9 - Prob. 4QQCh. 9 - What is the general chemical equation for each of...Ch. 9 - What is the general chemical equation for each of...Ch. 9 - Classify each of the following reactions as a...Ch. 9 - Classify each of the following reactions as a...Ch. 9 - Write the chemical formulas for the products...Ch. 9 - Write the chemical formulas for the products...Ch. 9 - Indicate whether or not each of the following...Ch. 9 - Indicate whether or not each of the following...Ch. 9 - Indicate to which of the following types of...Ch. 9 - Indicate to which of the following types of...Ch. 9 - What is the oxidation number of S in each of the...Ch. 9 - Prob. 9.12EPCh. 9 - Determine the oxidation number of the indicated...Ch. 9 - Determine the oxidation number of the indicated...Ch. 9 - Prob. 9.15EPCh. 9 - Prob. 9.16EPCh. 9 - What is the oxidation number of each element...Ch. 9 - What is the oxidation number of each element...Ch. 9 - Classify each of the following reactions as a...Ch. 9 - Classify each of the following reactions as a...Ch. 9 - Classify each of the following reactions as (1) a...Ch. 9 - Prob. 9.22EPCh. 9 - Classify each of the following reactions using one...Ch. 9 - Classify each of the following reactions using one...Ch. 9 - Prob. 9.25EPCh. 9 - In each of the following changes is the reactant...Ch. 9 - Identify which substance is oxidized and which...Ch. 9 - Identify which substance is oxidized and which...Ch. 9 - Prob. 9.29EPCh. 9 - Prob. 9.30EPCh. 9 - Indicate whether each of the following substances...Ch. 9 - Indicate whether each of the following substances...Ch. 9 - Prob. 9.33EPCh. 9 - Prob. 9.34EPCh. 9 - What are the three central concepts associated...Ch. 9 - Why are most chemical reactions carried out either...Ch. 9 - What two factors determine whether a collision...Ch. 9 - What happens to the reactants in an ineffective...Ch. 9 - Which of the following reactions are endothermic,...Ch. 9 - Prob. 9.40EPCh. 9 - Should heat be added as a reactant or as a product...Ch. 9 - Should heat be added as a reactant or as a product...Ch. 9 - Prob. 9.43EPCh. 9 - Indicate whether each of the following is a...Ch. 9 - Sketch an energy diagram graph representing an...Ch. 9 - Sketch an energy diagram graph representing an...Ch. 9 - Using collision theory, indicate why each of the...Ch. 9 - Using collision theory, indicate why each of the...Ch. 9 - Substances burn more rapidly in pure oxygen than...Ch. 9 - Milk will sour in a couple of days when left at...Ch. 9 - Will each of the changes listed increase or...Ch. 9 - Will each of the changes listed increase or...Ch. 9 - For each of the changes listed will the rate of...Ch. 9 - For each of the changes listed will the rate of...Ch. 9 - Prob. 9.55EPCh. 9 - Draw an energy diagram graph for an endothermic...Ch. 9 - The characteristics of four reactions, each of...Ch. 9 - The characteristics of four reactions, each of...Ch. 9 - What condition must be met in order for a system...Ch. 9 - What relationship exists between the rates of the...Ch. 9 - What does the term reversible reaction mean?Ch. 9 - What does the notation denote when it is used in...Ch. 9 - Consider the following equilibrium system....Ch. 9 - Consider the following equilibrium system....Ch. 9 - Prob. 9.65EPCh. 9 - Sketch a graph showing how the rates of the...Ch. 9 - The following series of diagrams represent the...Ch. 9 - The following series of diagrams represent the...Ch. 9 - For the reaction A2 + 2B 2AB, diagram I depicts...Ch. 9 - For the reaction A2 + B2 2AB, diagram I depicts...Ch. 9 - Write equilibrium constant expressions for the...Ch. 9 - Write equilibrium constant expressions for the...Ch. 9 - Write equilibrium constant expressions for the...Ch. 9 - Prob. 9.74EPCh. 9 - Calculate the value of the equilibrium constant...Ch. 9 - Calculate the value of the equilibrium constant...Ch. 9 - Prob. 9.77EPCh. 9 - Use the given Keq value and the terminology in...Ch. 9 - Write a balanced chemical equation for a totally...Ch. 9 - Write a balanced chemical equation for a totally...Ch. 9 - The following four diagrams represent gaseous...Ch. 9 - Based on the diagrams, chemical reaction, and...Ch. 9 - The following four diagrams represent gaseous...Ch. 9 - Based on the diagrams, chemical reaction, and...Ch. 9 - Indicate whether or not each of the following...Ch. 9 - Indicate whether or not each of the following...Ch. 9 - For the generalized chemical reaction...Ch. 9 - For the generalized chemical reaction...Ch. 9 - Prob. 9.89EPCh. 9 - For the reaction C6H6(g)+3H2(g)C6H12(g)+heat...Ch. 9 - Consider the following chemical system at...Ch. 9 - Prob. 9.92EPCh. 9 - The following two diagrams represent the...Ch. 9 - The following two diagrams represent the...Ch. 9 - Indicate whether or not product formation...Ch. 9 - Prob. 9.96EPCh. 9 - Prob. 9.97EPCh. 9 - Indicate whether or not product formation...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- . Explain what it means that a reaction has reached a state of chemical equilibrium. Explain why equilibrium is a dynamic state: Does a reaction really “stop” when the system reaches a state of equilibrium? Explain why, once a chemical system has reached equilibrium, the concentrations of all reactants remain constant with time. Why does this constancy of concentration not contradict our picture of equilibrium as being dynamic? What happens to the rates of the forward and reverse reactions as a system proceeds to equilibrium from a starting point where only reactants are present?arrow_forwardThe boxes shown below represent a set of initial conditions for the reaction: Draw a quantitative molecular picture that shows what this system looks like after the reactants are mixed in one of the boxes and the system reaches equilibrium. Support your answer with calculations. Consider an equilibrium mixture of four chemicals (A, B, C, and D, all gases) reacting in a closed flask according to the foll owing equation: A+BC+D a. You add more A to the flask. How does the concentration of each chemical compare to its original concentration after equilibrium is re-established? Justify your answer. b. You have the original set-up at equilibrium, and add more D to the flask. How does the concentration of each chemical compare to its original concentration after equilibrium is re-established? Justify your answer.arrow_forwardThe boxes shown below represent a set of initial conditions for the reaction: Draw a quantitative molecular picture that shows what this system looks like after the reactants are mixed in one of the boxes and the system reaches equilibrium. Support your answer with calculations.arrow_forward
- For the equilibrium reaction in Question 4, write the expression for the equilibrium constant. (a) How would this equilibrium constant change if the total pressure on the system were doubled? (b) How would the equilibrium constant change if the temperature were increased?arrow_forwardWhen a mixture of hydrogen and bromine is maintained at normal atmospheric pressure and heated above 200. °C in a closed container, the hydrogen and bromine react to form hydrogen bromide and a gas-phase equilibrium is established. Write a balanced chemical equation for the equilibrium reaction. Use bond enthalpies from Table 6.2 ( Sec. 6-6b) to estimate the enthalpy change for the reaction. Based on your answers to parts (a) and (b), which is more important in determining the position of this equilibrium, the entropy effect or the energy effect? In which direction will the equilibrium shift as the temperature increases above 200. °C? Explain. Suppose that the pressure were increased to triple its initial value. In which direction would the equilibrium shift? Why is the equilibrium not established at room temperature?arrow_forwardIndicate whether or not each of the following changes would affect the value of a systems equilibrium constant. a. Addition of a product to the equilibrium mixture b. Increase in the systems total pressure c. Increase in the systems temperature d. Addition of both a reactant and a product to the equilibrium mixturearrow_forward
- For the reaction C6H6(g)+3H2(g)C6H12(g)+heat determine in what direction the equilibrium will be shifted by each of the following changes. Decreasing the concentration of H2 a. Increasing the concentration of C6H6 b. Decreasing the temperature c. Increasing the pressure by decreasing the volume of the containerarrow_forwardWrite equilibrium constant expressions for the following generalized reactions. a. 2X(g)+3Y(g)2Z(g) b. 2X(g)+3Y(s)2Z(g) c. 2X(s)+3Y(s)2Z(g) d. 2X(g)+3Y(g)2Z(s)arrow_forwardAccording to the Resource Conservation and Recovery Act (RCRA), waste material is classified as toxic and must be handled as hazardous if the lead concentration exceeds 5 mg/L. By adding chloride ion, the lead ion will precipitate as PbCl2, which can be separated from the liquid portion. Once the lead has been removed, the rest of the waste can be sent to a conventional waste treatment facility. How many grams of sodium chloride must be added to 500 L of a waste solution to reduce the concentration of the Pb2+ ion from 10 to 5 mg/L?arrow_forward
- Describe a nonchemical system that is in equilibrium, and explain how the principles of equilibrium apply to the system.arrow_forwardExplain that equilibrium is dynamic, and that at equilibrium the forward and backward reaction rates are equal.arrow_forwardTell what will happen to each equilibrium concentration in the following when the indicated stress is applied and a new equilibrium position is established. a. LiOH(s)+CO2(g)LiHCO3(s)+heat; CO2 is removed. b. 2NaHCO3(s)+heatNa2O(s)+2CO2(g)+H2O(g); The system is cooled. c. CaCO3(s)+heatCaO(s)+CO2(g); The system is cooled.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY