
Concept explainers
How many
How many yield 2,3-dimethylbutane?
How many yield methylcyclobutane?
Interpretation:
The number of alkenes which are required to produce the given alkanes on catalytic hydrogenation is to be determined.
Concept introduction:
On catalytic hydrogenation, alkenes get converted to the corresponding alkanes with the same number of carbon atoms.
In hydrogenation reaction, one hydrogen atom gets attached to each of the double bonded carbon atoms.
To accelerate the rate of hydrogenation, the metal catalyst provides an alternative pathway involving low activation energy steps.
The alkane that is formed contains two hydrogen atoms more than the corresponding alkene and these two hydrogen atoms are added on the adjacent carbon atoms in the alkene.
Answer to Problem 25P
Solution:
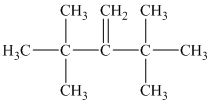
a) Only one alkene can produce
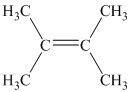
b) Two alkenes can produce
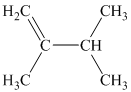
c) Three alkenes can produce methylcyclobutane upon catalytic hydrogenation.
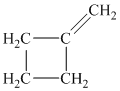
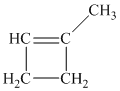
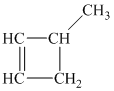
Explanation of Solution
a) The structure of
Hydrogenation of alkenes adds one hydrogen atom on each carbon atom of the double bonded carbon atoms. So in the alkane, these two carbon atoms (previously double bonded) should be attached to at least one hydrogen atom each.
In
The alkane structure is symmetric, and carbon atoms
b) The structure of
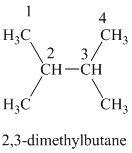
The hydrogen of alkenes adds one hydrogen atom on each carbon atom of the double bonded carbon atoms. So in the alkane, these two carbon atoms (previously double bonded) should be attached to at least one hydrogen atom each.
In
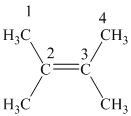
The
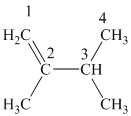
The alkane structure is symmetric, and the carbon atoms
Hence, two alkenes can produce
c) The structure of methylcyclobutane is shown below:
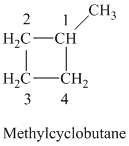
The hydrogen of alkenes adds one hydrogen atom on each carbon atom of the double bonded carbon atoms. So in the alkane, these two carbon atoms (previously double bonded) should be attached to one at least one hydrogen atom each.
In methylcyclobutane, the
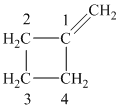
The
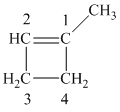
The
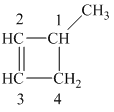
The alkane structure is symmetric. This means, there cannot be any other distinct alkene structure which would produce methylcyclobutane upon catalytic hydrogenation.
Hence, three alkenes can produce methylcyclobutane upon catalytic hydrogenation.
Want to see more full solutions like this?
Chapter 8 Solutions
ORGANIC CHEMISTRY-PACKAGE >CUSTOM<
- (ME EX1) Prblm #9/10 Can you explain in detail (step by step) I'm so confused with these problems. For turmber 13 can u turn them into lewis dot structures so I can better understand because, and then as well explain the resonance structure part. Thanks for the help.arrow_forwardProblems 19 and 20: (ME EX1) Can you please explain the following in detail? I'm having trouble understanding them. Both problems are difficult for me to explain in detail, so please include the drawings and answers.arrow_forward(ME EX1) Prblm #4-11 Can you please help me and explain these I'm very confused in detail please. Prblm number 9 I don't understand at all (its soo confusing to me and redraw it so I can better depict it).arrow_forward
- ME EX1) Prblm #19-20 I'm so confused with these problems. Can you please help me solve them and explain them? Problems number 19-20, and thanks! step by step and in detail for me please helparrow_forwardCalculate the flux of oxygen between the ocean and the atmosphere, given that: Temp = 18°C Salinity = 35 ppt Density = 1025 kg/m3 Oxygen concentration measured in bulk water = 263.84 mmol/m3 Wind speed = 7.4 m/s Oxygen is observed to be about 10% initially supersaturatedarrow_forward( ME EX1) Prblm 27-28: Can you explain to me both prblms in detail and for prblm 28 what do you mean bi conjugated bi ponds and those structures I'm confused...arrow_forward
- A. Determine the number of electrons in a system of cyclic conjugation (zero if no cyclic conjugation). B. Specify whether the species is "a"-aromatic, "aa"-anti-aromatic, or "na"-non-aromatic (neither aromatic nor anti-aromatic). (Presume rings to be planar unless structure obviously prevents planarity. If there is more than one conjugated ring, count electrons in the largest.) 1. A.Electrons in a cyclic conjugated system. 18 B.The compound is (a, aa, or na) a 2. A.Electrons in a cyclic conjugated system. 10 B.The compound is (a, aa, or na) naarrow_forwardWater is boiling at 1 atm pressure in a stainless steel pan on an electric range. It is observed that 2 kg of liquid water evaporates in 30 min. Find the rate of heat transfer to the water (kW).arrow_forwardCould you please turn this into a complete Lewis dot structure formula for me so I can visualize it more clearly? and then do the explaining for the resonance structures that were given please.arrow_forward
- Could you please turn this into a complete Lewis dot structure formula for me so I can visualize it more clearly? and then do the explaining for the question.arrow_forwardplease solve. If the answer is "no error" and it asks me to type something, and i typed a-helix, its always wrong.arrow_forwardCan you please solve and explain this for me in a simple way? I cant seem to comprehend this problem.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


