
ORGANIC CHEMISTRY-PACKAGE >CUSTOM<
10th Edition
ISBN: 9781260028355
Author: Carey
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 8, Problem 66P
As a method for the preparation of
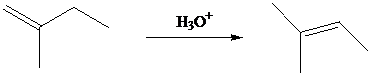
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Under strongly acidic conditions, hexane is observed to undergo an isomerization process, during which it is converted into
branched alkanes having the same molecular formula as hexane (C6H₁4). The rate at which each constitutional isomer is produced
is related to the type of acid that is used in the reaction and the degree of mixing that occurs during the reaction (J. Chem. Soc.,
Perkins Trans. 2 1999, 12, 2715-2718).
Step 1
Draw a bond-line structure of hexane.
Draw Your Solution
There are four branched isomers of hexane. Draw bond-line structures of all four of its isomers.
Draw Your Solution
Ethyl ethanoate (commonly called ethyl acetate), CH3CO₂C₂H5, is an important solvent used in nail polish remover, lacquers,
adhesives, the manufacture of plastics, and even as a food favoring. It is produced from acetic acid and ethanol by the reaction
CH3CO₂H + C₂H5OH
CH³CO₂C₂H5 + H₂O
ethanoic acid
(acetic acid)
ethanol
ethyl ethanoate
(ethyl acetate)
At 25 °C, Kc = 4.10 for this reaction.
In a reaction mixture, the following equilibrium concentrations were observed:
[CH3CO₂H] = 0.318 M,
[H₂O] = 0.00671 M, and
[CH3CO₂C₂H5 ] = 0.702 M.
What was the concentration of C₂H5OH in the mixture?
[C2₂H5OH] =
M
Carbon–carbon bond dissociation enthalpies have been measured for many alkanes. Identify the alkane in each of the following pairs that has the lower carbon–carbon bond-dissociation enthalpy, and explain the reason for your choice. (a) Ethane or propane (b) Propane or 2-methylpropane (c) 2-Methylpropane or 2,2-dimethylpropane (d) Cyclobutane or cyclopentane
Chapter 8 Solutions
ORGANIC CHEMISTRY-PACKAGE >CUSTOM<
Ch. 8.1 - What three alkenes yield 2-methylbutane on...Ch. 8.2 - Prob. 2PCh. 8.2 - Prob. 3PCh. 8.3 - Prob. 4PCh. 8.4 - Prob. 5PCh. 8.4 - Give a structural formula for the carbocation...Ch. 8.5 - Prob. 7PCh. 8.6 - Instead of the three-step process of Mechanism...Ch. 8.6 - The rates of hydration of the two alkenes shown...Ch. 8.6 - Is the electrophilic addition of hydrogen chloride...
Ch. 8.7 - You can calculate the equilibrium constant for the...Ch. 8.7 - Does the presence or absence of a catalyst such as...Ch. 8.7 - The gas phase reaction of ethanol with hydrogen...Ch. 8.8 - Prob. 14PCh. 8.8 - Hydroborationoxidation of -pinene, like its...Ch. 8.10 - Arrange the compounds 2-methyl-1-butene,...Ch. 8.10 - Give the structure of the product formed when each...Ch. 8.11 - Prob. 18PCh. 8.11 - Prob. 19PCh. 8.12 - Prob. 20PCh. 8.12 - Prob. 21PCh. 8.13 - Prob. 22PCh. 8.14 - Prob. 23PCh. 8.14 - Prob. 24PCh. 8 - How many alkenes yield...Ch. 8 - Prob. 26PCh. 8 - Catalytic hydrogenation of...Ch. 8 - Prob. 28PCh. 8 - Prob. 29PCh. 8 - Prob. 30PCh. 8 - Prob. 31PCh. 8 - A single epoxide was isolated in 7984% yield in...Ch. 8 - Prob. 33PCh. 8 - Prob. 34PCh. 8 - On catalytic hydrogenation over a rhodium...Ch. 8 - Prob. 36PCh. 8 - Prob. 37PCh. 8 - Prob. 38PCh. 8 - Prob. 39PCh. 8 - 1-Butene has a higher heat of hydrogenation than...Ch. 8 - Match the following alkenes with the appropriate...Ch. 8 - The heats of reaction were measured for addition...Ch. 8 - Complete the following table by adding + and -...Ch. 8 - Match the heats of hydrogenation (107 kJ/mol,...Ch. 8 - The iodination of ethylene at 25 C is...Ch. 8 - Specify reagents suitable for converting...Ch. 8 - (a) Which primary alcohol of molecular formula...Ch. 8 - Identify compounds A and B in the retrosynthesis...Ch. 8 - Identify compounds A and B in the retrosynthesis...Ch. 8 - Prob. 50PCh. 8 - On being heated with a solution of sodium ethoxide...Ch. 8 - Compound A (C7H15Br) is not a primary alkyl...Ch. 8 - Prob. 53PCh. 8 - Prob. 54PCh. 8 - A mixture of three alkenes (A, B, and C) was...Ch. 8 - Reaction of 3,3-dimethyl-1-butene with hydrogen...Ch. 8 - Dehydration of 2,2,3,4,4-pentamethyl-3-pentanol...Ch. 8 - Prob. 58PCh. 8 - East Indian sandalwood oil contains a hydrocarbon...Ch. 8 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - On the basis of the mechanism of acid-catalyzed...Ch. 8 - As a method for the preparation of alkenes, a...Ch. 8 - Which of the following is the most reasonable...Ch. 8 - Prob. 68PCh. 8 - Oxymercuration Concerns about mercurys toxicity...Ch. 8 - Prob. 70DSPCh. 8 - Prob. 71DSPCh. 8 - Prob. 72DSPCh. 8 - Prob. 73DSPCh. 8 - Oxymercuration Concerns about mercurys toxicity...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- You are asked to prepare (synthesize) chloroethane using an alkane and any inorganic substance of your choice. Write a chemical equation for the synthesis of chloroethane. 3.arrow_forwardThe boiling points for the compounds are 118 °C and 35 °C respectively. The solubility for both compounds is the same (8g/100g water). Explain this observation for (i) boiling point disparity; (ii) solubility similarity H-bonds form in diethyl ether; n-butyl alcohol forms H-bonds in water H-bonds form in n-butyl alcohol; diethyl ether forms H-bonds in water H-bonds in n-butyl alcohol; Both compounds form H-bonds in water O Both compounds form H-bonds; Both compounds form H-bonds in waterarrow_forward2-chloropropane is a major product of the reaction of chlorine with propane under ultraviolet light. Write the mechanism for this reaction including the initiation step and the two propagation steps.arrow_forward
- Analyze the following structure of limonene in terms of isoprene units joined by head-to-tail linkages. (1) How many isoprene units are there in a limonene molecule? (2) Classify limonene as a monoterpene (one terpene unit), a sesquiterpene (1.5 terpene units), or a diterpene (2 terpene units).arrow_forwardGive 3 examples of combustion of alkanes and write its general reaction and mechanismarrow_forwardExplain this ? "Organic synthesis is the systematic preparation of a compound from areadily available starting material by one or many steps".arrow_forward
- Following are the steps in the industrial synthesis of glycerin. Provide structures for all intermediate compounds (AD) and describe the type of mechanism by which each is formed.arrow_forwardLipoic acid is required by many microorganisms for proper growth. As a disulfide, it functions in the living system by catalyzing certain oxidation reactions and is reduced in the process. Write the structure of the reduction product.arrow_forwardWhen bromine is added to two beakers, one containing phenyl isopropyl ether and the other containing cyclohexene, the bromine color in both beakers disappears. What observation could you make while performing this test that would allow you to distinguish the alkene from the aryl ether?arrow_forward
- CH3COOH + 2H2 = C2H5OH + H2O %3D Acetate Ethanol Acetate may be hydrogenated to ethanol in industrial reaction systems using several catalysts: Select the most effective catalyst for this reaction system if the free energy of the reaction is -34 kJ/mol; Catalist effective at the temperature range 37-45°C, O Catalist effective at the temperature range 40-55°C, O Catalist effective at the temperature range 60-72°C,arrow_forwardWhich of the following is true for the reactions of alkyl halides? (a) The characteristic reactions of alkyl halides are oxidation and reduction.(b) The characteristic reactions of alkyl halides are elimination and substitutionc. The characteristic reactions of alkyl halides are addition and substitutiond. Characteristic reactions of alkyl halides are addition and elimination.arrow_forward1. Consider the solubility and boiling point of the following pair of compounds: n-butyl alcohol and diethyl ether. The boiling points for the compounds are 118 °C and 35 °C respectively. The solubility for both compounds is the same (8g/100g water). Explain this observation for (i) boiling point disparity; (ii) solubility similarity a. H-bonds form in diethyl ether; n-butyl alcohol forms H-bonds in water b. H-bonds form in n-butyl alcohol; diethyl ether forms H-bonds in water c. H-bonds in n-butyl alcohol; Both compounds form H-bonds in water d. Both compounds form H-bonds; Both compounds form H-bonds in water 2. Account for the bond angle differences between (i) H-C-H (109.5°) in methane and H-S-H (90°); H-C-H (109.5°) and H-O-H (107.5°) in water. a. The H-S-H has two lone pairs; The H-O-H has two lone pairs b. The H-S-H has no hybridization at p-orbitals; The H-O-H has two lone pairs c. The H-S-H has two lone pairs; The H-O-H has no hybridization…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY