
Concept explainers
(a)
Interpretation:
The predominant organic product that is formed in the given reaction has to be written.
Concept Introduction:
Alcohol is an organic compound that has hydroxyl as its
Alkenes are compounds that contain a double bond between carbon atoms. When alkenes undergo hydration in presence of sulfuric acid as catalyst, an alcohol is formed as product. The major product formed in case of unsymmetrical alkene is found by using Markovnikov’s rule. The general scheme for hydration of alkene can be given as,
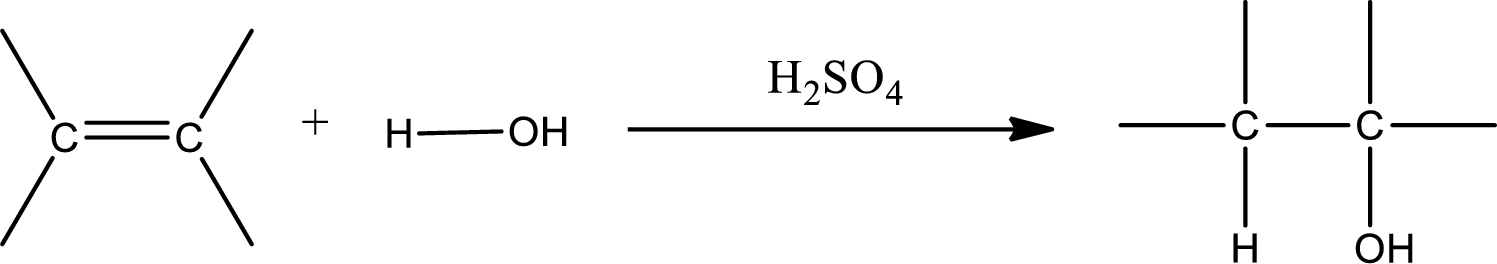
(b)
Interpretation:
The predominant organic product that is formed in the given reaction has to be written.
Concept Introduction:
Alcohol is an organic compound that has hydroxyl as its functional group. Alcohols contain both nonpolar and polar groups in it. Hydroxyl group is the polar group and the alkyl group is the nonpolar group. Physical properties of alcohol depend on which of the two groups dominate. Alcohols can be prepared in laboratory by hydration of alkenes and reduction of carbonyl compounds.
Double bond between a carbon atom and oxygen atom means that the compound is a carbonyl compound. Addition of hydrogen to this carbonyl group leads to the formation of alcohol. When hydrogen is added to the carbonyl, the oxygen of the carbonyl is converted into hydroxyl group. A scheme for the addition of hydrogen to the carbonyl group can be given as shown below,
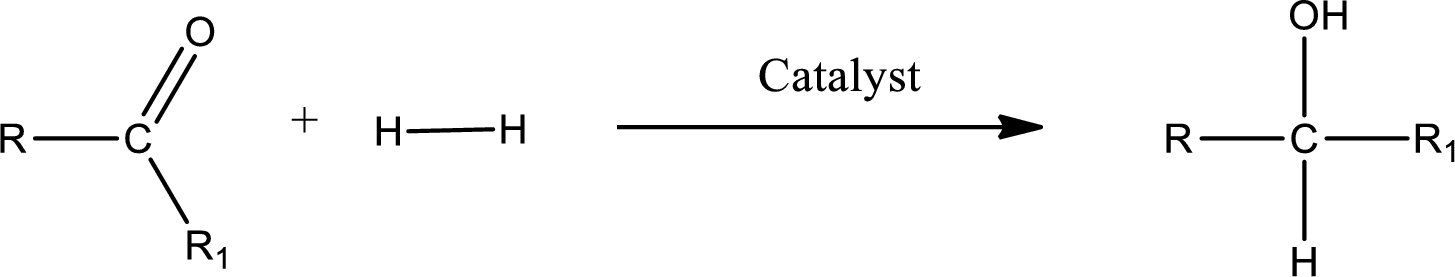
(c)
Interpretation:
The predominant organic product that is formed in the given reaction has to be written.
Concept Introduction:
Alcohol is an organic compound that has hydroxyl as its functional group. Alcohols contain both nonpolar and polar groups in it. Hydroxyl group is the polar group and the alkyl group is the nonpolar group. Physical properties of alcohol depend on which of the two groups dominate. Alcohols can be prepared in laboratory by hydration of alkenes and reduction of carbonyl compounds.
Alkenes are compounds that contain a double bond between carbon atoms. When alkenes undergo hydration in presence of sulfuric acid as catalyst, an alcohol is formed as product. The major product formed in case of unsymmetrical alkene is found by using Markovnikov’s rule. The general scheme for hydration of alkene can be given as,
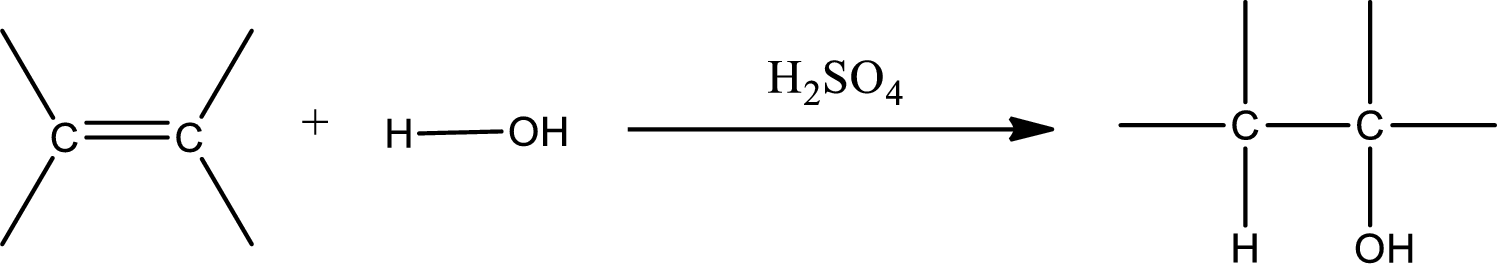
(d)
Interpretation:
The predominant organic product that is formed in the given reaction has to be written.
Concept Introduction:
Alcohol is an organic compound that has hydroxyl as its functional group. Alcohols contain both nonpolar and polar groups in it. Hydroxyl group is the polar group and the alkyl group is the nonpolar group. Physical properties of alcohol depend on which of the two groups dominate. Alcohols can be prepared in laboratory by hydration of alkenes and reduction of carbonyl compounds.
Double bond between a carbon atom and oxygen atom means that the compound is a carbonyl compound. Addition of hydrogen to this carbonyl group leads to the formation of alcohol. When hydrogen is added to the carbonyl, the oxygen of the carbonyl is converted into hydroxyl group. A scheme for the addition of hydrogen to the carbonyl group can be given as shown below,
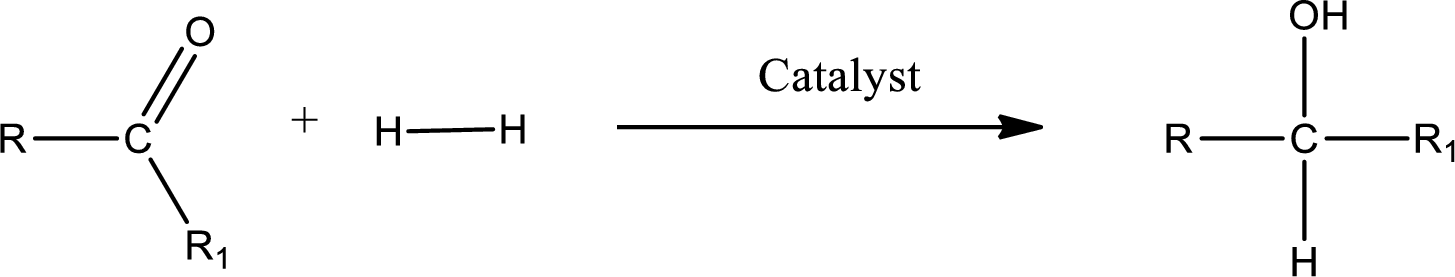
Trending nowThis is a popular solution!

Chapter 3 Solutions
Organic And Biological Chemistry
- Identify and provide a concise explanation of the concept of signal-to-noise ratio (SNR) in the context of chemical analysis. Provide specific examples.arrow_forwardIdentify and provide a concise explanation of a specific analytical instrument capable of detecting and quantifying trace compounds in food samples. Emphasise the instrumental capabilities relevant to trace compound analysis in the nominated food. Include the specific application name (eg: identification and quantification of mercury in salmon), outline a brief description of sample preparation procedures, and provide a summary of the obtained results from the analytical process.arrow_forwardIdentify and provide an explanation of what 'Seperation Science' is. Also describe its importance with the respect to the chemical analysis of food. Provide specific examples.arrow_forward
- 5. Propose a Synthesis for the molecule below. You may use any starting materials containing 6 carbons or less (reagents that aren't incorporated into the final molecule such as PhзP do not count towards this total, and the starting material can have whatever non-carbon functional groups you want), and any of the reactions you have learned so far in organic chemistry I, II, and III. Your final answer should show each step separately, with intermediates and conditions clearly drawn. H3C CH3arrow_forwardState the name and condensed formula of isooxazole obtained by reacting acetylacetone and hydroxylamine.arrow_forwardState the name and condensed formula of the isothiazole obtained by reacting acetylacetone and thiosemicarbazide.arrow_forward
- Provide the semi-developed formula of isooxazole obtained by reacting acetylacetone and hydroxylamine.arrow_forwardGiven a 1,3-dicarbonyl compound (R1-CO-CH2-CO-R2), indicate the formula of the compound obtaineda) if I add hydroxylamine (NH2OH) to give an isooxazole.b) if I add thiosemicarbazide (NH2-CO-NH-NH2) to give an isothiazole.arrow_forwardAn orange laser has a wavelength of 610 nm. What is the energy of this light?arrow_forward
- The molar absorptivity of a protein in water at 280 nm can be estimated within ~5-10% from its content of the amino acids tyrosine and tryptophan and from the number of disulfide linkages (R-S-S-R) between cysteine residues: Ε280 nm (M-1 cm-1) ≈ 5500 nTrp + 1490 nTyr + 125 nS-S where nTrp is the number of tryptophans, nTyr is the number of tyrosines, and nS-S is the number of disulfide linkages. The protein human serum transferrin has 678 amino acids including 8 tryptophans, 26 tyrosines, and 19 disulfide linkages. The molecular mass of the most dominant for is 79550. Predict the molar absorptivity of transferrin. Predict the absorbance of a solution that’s 1.000 g/L transferrin in a 1.000-cm-pathlength cuvet. Estimate the g/L of a transferrin solution with an absorbance of 1.50 at 280 nm.arrow_forwardIn GC, what order will the following molecules elute from the column? CH3OCH3, CH3CH2OH, C3H8, C4H10arrow_forwardBeer’s Law is A = εbc, where A is absorbance, ε is the molar absorptivity (which is specific to the compound and wavelength in the measurement), and c is concentration. The absorbance of a 2.31 × 10-5 M solution of a compound is 0.822 at a wavelength of 266 nm in a 1.00-cm cell. Calculate the molar absorptivity at 266 nm.arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div





