
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 26.1, Problem 26.1P
(a)
Interpretation Introduction
Interpretation:
The numbers of possible constitutional isomers are in a triglyceride, which contains one molecule each of palmitic acid, oleic acid, and stearic acid has to be identified.
Concept Introduction:
Constitutional isomers: These are compounds having same molecular formula but having different structural arrangements that is different connectivity of atoms or group of atoms in it
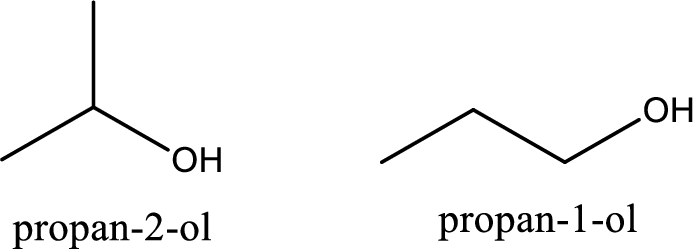
(b)
Interpretation Introduction
Interpretation:
Among the constitutional isomers, the chiral ones have to be identified.
Concept Introduction:
Chiral molecule: Molecules containing atoms which are surrounded by different atoms or group of atoms are called chiral molecule.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
2. Draw the missing structure(s) in each of the following reactions. The missing
structure(s) can be a starting material or the major reaction product(s).
C5H10
H-CI
CH2Cl2
CI
Draw the products of the stronger acid protonating the other reactant.
དའི་སྐད”“
H3C
OH
H3C
CH
CH3
KEq
Product acid
Product base
Draw the products of the stronger acid protonating the other reactant.
H3C
NH2
NH2
KEq
H3C-CH₂
1.
Product acid
Product base
Chapter 26 Solutions
Organic Chemistry
Ch. 26.1 - Prob. 26.1PCh. 26.6 - Prob. AQCh. 26.6 - Prob. BQCh. 26.6 - Prob. CQCh. 26.6 - Prob. DQCh. 26.6 - Prob. FQCh. 26 - Prob. 26.2PCh. 26 - Identify the hydrophobic and hydrophilic region(s)...Ch. 26 - Prob. 26.4PCh. 26 - Prob. 26.5P
Ch. 26 - Prob. 26.6PCh. 26 - Prob. 26.7PCh. 26 - Prob. 26.8PCh. 26 - Prob. 26.9PCh. 26 - How many moles of H2 are used in the catalytic...Ch. 26 - Prob. 26.11PCh. 26 - Prob. 26.12PCh. 26 - Prob. 26.13PCh. 26 - Prob. 26.14PCh. 26 - Prob. 26.15PCh. 26 - Prob. 26.16PCh. 26 - Prob. 26.17PCh. 26 - Prob. 26.18PCh. 26 - Prob. 26.20PCh. 26 - Prob. 26.21PCh. 26 - Prob. 26.22PCh. 26 - Prob. 26.23PCh. 26 - Following is a structural formula for cortisol...Ch. 26 - Prob. 26.25PCh. 26 - Draw the structural formula of a lecithin...Ch. 26 - Prob. 26.27PCh. 26 - Prob. 26.28PCh. 26 - Prob. 26.29PCh. 26 - Prob. 26.30PCh. 26 - Prob. 26.31PCh. 26 - Prob. 26.32PCh. 26 - Prob. 26.33P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What alkene or alkyne yields the following products after oxidative cleavage with ozone? Click the "draw structure" button to launch the drawing utility. draw structure ... andarrow_forwardDraw the products of the stronger acid protonating the other reactant. H3C-C=C-4 NH2 KEq CH H3C `CH3 Product acid Product basearrow_forward2. Draw the missing structure(s) in each of the following reactions. The missing structure(s) can be a starting material or the major reaction product(s). C5H10 Br H-Br CH2Cl2 + enant.arrow_forward
- Draw the products of the stronger acid protonating the other reactant. KEq H₂C-O-H H3C OH Product acid Product basearrow_forwardDraw the products of the stronger acid protonating the other reactant. OH KEq CH H3C H3C `CH3 Product acid Product basearrow_forward2. Draw the missing structure(s) in each of the following reactions. The missing structure(s) can be a starting material or the major reaction product(s). Ph H-I CH2Cl2arrow_forward
- 3 attempts left Check my work Draw the products formed in the following oxidative cleavage. [1] 03 [2] H₂O draw structure ... lower mass product draw structure ... higher mass productarrow_forward2. Draw the missing structure(s) in each of the following reactions. The missing structure(s) can be a starting material or the major reaction product(s). H-Br CH2Cl2arrow_forwardWrite the aldol condensation mechanism and product for benzaldehyde + cyclohexanone in a base. Then trans-cinnamaldehyde + acetone in base. Then, trans-cinnamaldehyde + cyclohexanone in a base.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning