
Concept explainers
(a)
Interpretation: To characterize
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A general oxidative deamination reaction is as follows:
(a)
Answer to Problem 26.45EP
Explanation of Solution
In both transamination and oxidative deamination reaction exchange of an amino group from an
(b)
Interpretation: To characterize glutamate as a possible reactant, product, or enzyme involved in transamination, oxidative deamination, or both transamination and oxidative deamination.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A general oxidative deamination reaction is as follows:
(b)
Answer to Problem 26.45EP
Glutamate acts as a reactant in both transamination and oxidative deamination reaction.
Explanation of Solution
In both transamination and oxidative deamination reaction exchange of an amino group from an
Glutamate is an amino acid thus acts as a reactant in both transamination and oxidative deamination reaction to give corresponding keto acid
(c)
Interpretation: To characterize glutamate dehydrogenase as a possible reactant, product, or enzyme involved in transamination, oxidative deamination, or both transamination and oxidative deamination.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A general oxidative deamination reaction is as follows:
(c)
Answer to Problem 26.45EP
Glutamate dehydrogenase is the enzyme involved in the oxidative deamination reaction of glutamate to give
Explanation of Solution
Oxidative deamination reaction of glutamate requires dehydrogenase enzyme. It is an oxidoreductase enzyme and works with either
(d)
Interpretation: To characterize
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A general oxidative deamination reaction is as follows:
(d)
Answer to Problem 26.45EP
The ammonium ion is one of the products obtained in oxidative deamination reaction of glutamate.
Explanation of Solution
Glutamate is an
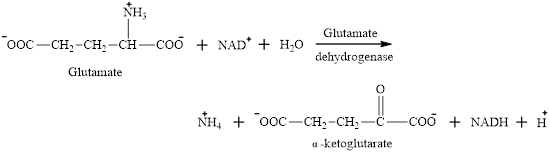
The ammonium ion formed is toxic if build up in the body. Ammonium ion acts as the “nitrogen carrier” for further reactions.
Want to see more full solutions like this?
Chapter 26 Solutions
General, Organic, and Biological Chemistry
- Please help me solve this reaction.arrow_forwardIndicate the products obtained by mixing 2,2-dimethylpropanal with acetaldehyde and sodium ethoxide in ethanol.arrow_forwardSynthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- If possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forwardSynthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forward
- We mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forwardIndicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,




