
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 22.62P
A newer generation of antipsychotics, among them clozapine, are now used to treat the symptoms of schizophrenia. These drugs are more effective than earlier drugs in improving patient response in the areas of social withdrawal, apathy, memory, comprehension, and judgment. They also produce fewer side effects such as seizures and tardive dyskinesia (involuntary body movements). In the following synthesis of clozapine, Step 1 is an Ullmann coupling, a type of nucleophilic
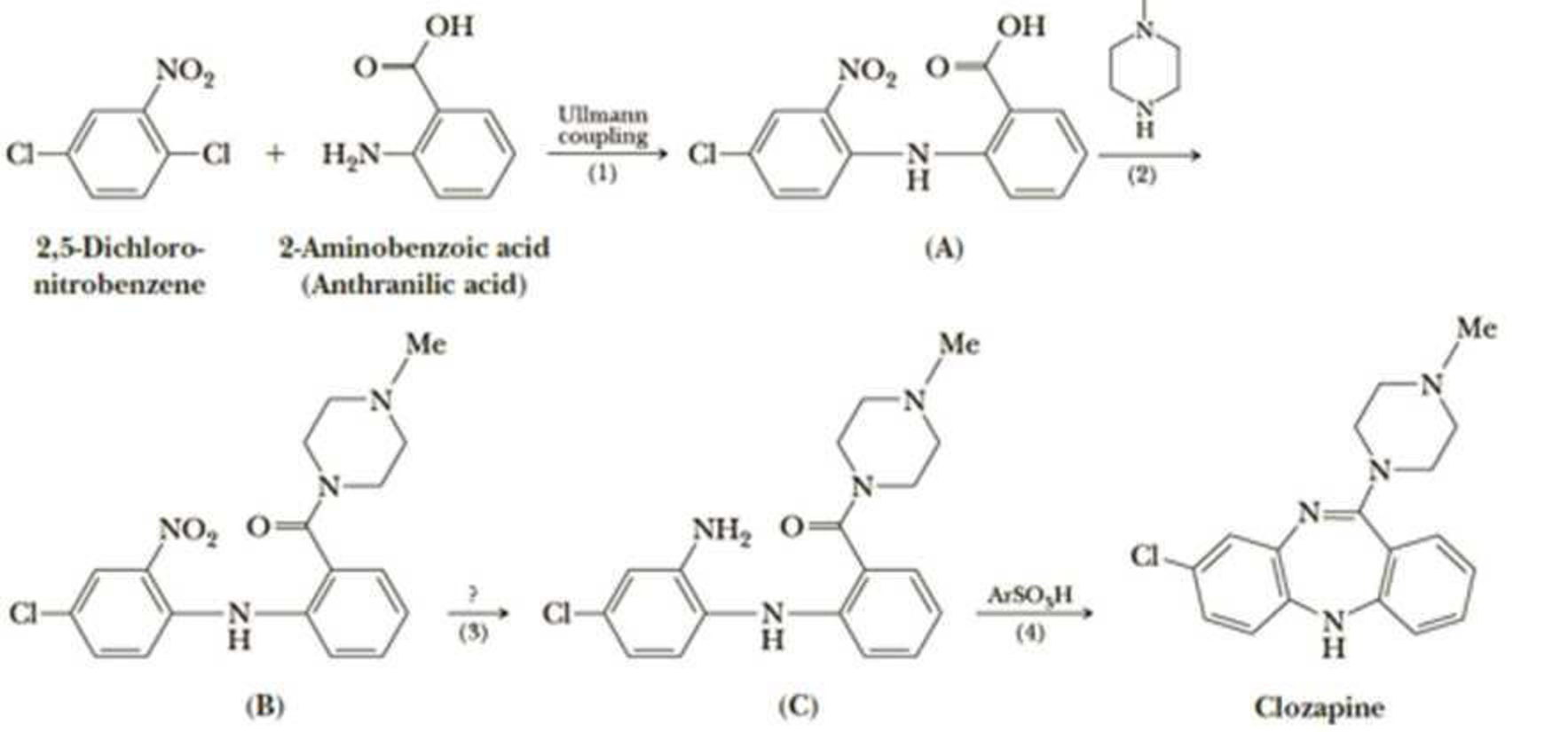
- (a) Show how you might bring about formation of the amide in Step 2.
- (b) Propose a reagent for Step 3.
- (c) Propose a mechanism for Step 4.
- (d) Is clozapine chiral? If so, how many of the possible stereoisomers are formed in this synthesis?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Using the following two half-reactions, determine the pH range in which $NO_2^-\ (aq)$ cannot be found as the predominant chemical species in water.* $NO_3^-(aq)+10H^+(aq)+8e^-\rightarrow NH_4^+(aq)+3H_2O(l),\ pE^{\circ}=14.88$* $NO_2^-(aq)+8H^+(aq)+6e^-\rightarrow NH_4^+(aq)+2H_2O(l),\ pE^{\circ}=15.08$
Indicate characteristics of oxodec acid.
What is the final product when hexanedioic acid reacts with 1º PCl5 and 2º NH3.
Chapter 22 Solutions
Organic Chemistry
Ch. 22.1 - Prob. 22.1PCh. 22.1 - Write a structural formula for the product from...Ch. 22.1 - Prob. 22.3PCh. 22.2 - Prob. 22.4PCh. 22.2 - Predict the major produce(s) of each electrophilic...Ch. 22.3 - In SN2 reactions of haloalkanes, the order of...Ch. 22 - Prob. 22.8PCh. 22 - Prob. 22.9PCh. 22 - Addition of m-xylene to the strongly acidic...Ch. 22 - Addition of tert-butylbenzene to the strongly...
Ch. 22 - What product do you predict from the reaction of...Ch. 22 - Other groups besides H+ can act as leaving groups...Ch. 22 - Prob. 22.14PCh. 22 - Prob. 22.15PCh. 22 - Prob. 22.16PCh. 22 - Prob. 22.17PCh. 22 - Suggest a reason why the nitroso group, N=O, is...Ch. 22 - Prob. 22.19PCh. 22 - Prob. 22.20PCh. 22 - The following molecules each contain two aromatic...Ch. 22 - Prob. 22.22PCh. 22 - Prob. 22.23PCh. 22 - The insecticide DDT is prepared by the following...Ch. 22 - Prob. 22.25PCh. 22 - Prob. 22.26PCh. 22 - Prob. 22.27PCh. 22 - Prob. 22.28PCh. 22 - Prob. 22.29PCh. 22 - Prob. 22.32PCh. 22 - Show how to prepare each compound from...Ch. 22 - Prob. 22.34PCh. 22 - Show reagents and conditions to bring about the...Ch. 22 - Prob. 22.36PCh. 22 - Propose a synthesis for each compound from...Ch. 22 - The first widely used herbicide for the control of...Ch. 22 - The first widely used herbicide for the control of...Ch. 22 - Prob. 22.40PCh. 22 - Prob. 22.41PCh. 22 - Prob. 22.42PCh. 22 - Prob. 22.43PCh. 22 - Cancer of the prostate is the second leading cause...Ch. 22 - Prob. 22.45PCh. 22 - Prob. 22.46PCh. 22 - Prob. 22.47PCh. 22 - When certain aromatic compounds are treated with...Ch. 22 - Prob. 22.49PCh. 22 - Following is the structure of miconazole, the...Ch. 22 - Prob. 22.51PCh. 22 - Prob. 22.52PCh. 22 - Prob. 22.53PCh. 22 - Show how the antidepressant venlafaxine (Effexor)...Ch. 22 - Prob. 22.57PCh. 22 - Given this retrosynthetic analysis, propose a...Ch. 22 - Prob. 22.59PCh. 22 - Prob. 22.60PCh. 22 - Prob. 22.61PCh. 22 - A newer generation of antipsychotics, among them...Ch. 22 - Prob. 22.63P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the final product when D-galactose reacts with hydroxylamine?arrow_forwardIndicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.arrow_forwardIn the two chair conformations of glucose, the most stable is the one with all the OH groups in the equatorial position. Is this correct?arrow_forward
- please help me with my homeworkarrow_forwardhelparrow_forwardThe temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forward
- QUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forwarder your payment details | bar xb Home | bartleby x + aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1 O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 1 3- 0- 0 200 Explanation Check temperature (K) 400 X Q Search L G 2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Cearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY