
World of Chemistry, 3rd edition
3rd Edition
ISBN: 9781133109655
Author: Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher: Brooks / Cole / Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 20, Problem 60A
Interpretation Introduction
Interpretation:
The repeating unit of each
Concept Introduction :
The substance which is made up of the larger number of identical units that bonded together by the covalent bond is known as
Expert Solution & Answer
Answer to Problem 60A
The sketch of a repeating units of Nylon-6, 6 is
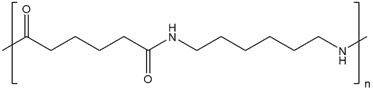
The sketch representation of a repeating unit of Dacron is

Explanation of Solution
Polymers are the substance that is made up with the help of a large number of identical units which are bonded together by the covalent bond and monomer is the smallest unit that is used to prepare polymers.
- Nylon: - Nylon is the polymer which is made up of the diamine acid and dicarboxylic acid and the repeating units are connected by the amide linkage and give the water molecule. The most common polymer of nylon is nylon-6, 6. The polymer nylon-6, 6 has the monomerhexamethylenediamineandadipic acid.
- Dacron: - Dacron is the polymer that is made up of ethylene glycol and terephthalic acid under the higher temperature.The preparation of dacron is a polycondensation reaction.
Conclusion
The monomer of nylon-6, 6 is hexamethylenediamine and adipic acid and the monomer unit of Dacron is ethylene glycol and terephthalic acid.
Chapter 20 Solutions
World of Chemistry, 3rd edition
Ch. 20.1 - Prob. 1RQCh. 20.1 - Prob. 2RQCh. 20.1 - Prob. 3RQCh. 20.1 - Prob. 4RQCh. 20.1 - Prob. 5RQCh. 20.1 - Prob. 6RQCh. 20.1 - Prob. 7RQCh. 20.1 - Prob. 8RQCh. 20.2 - Prob. 1RQCh. 20.2 - Prob. 2RQ
Ch. 20.2 - Prob. 3RQCh. 20.2 - Prob. 4RQCh. 20.2 - Prob. 5RQCh. 20.2 - Prob. 6RQCh. 20.3 - Prob. 1RQCh. 20.3 - Prob. 2RQCh. 20.3 - Prob. 3RQCh. 20.3 - Prob. 4RQCh. 20.3 - Prob. 5RQCh. 20.4 - Prob. 1RQCh. 20.4 - Prob. 2RQCh. 20.4 - Prob. 3RQCh. 20.4 - Prob. 4RQCh. 20.4 - Prob. 5RQCh. 20 - Prob. 1ACh. 20 - Prob. 2ACh. 20 - Prob. 3ACh. 20 - Prob. 4ACh. 20 - Prob. 5ACh. 20 - Prob. 6ACh. 20 - Prob. 7ACh. 20 - Prob. 8ACh. 20 - Prob. 9ACh. 20 - Prob. 10ACh. 20 - Prob. 11ACh. 20 - Prob. 12ACh. 20 - Prob. 13ACh. 20 - Prob. 14ACh. 20 - Prob. 15ACh. 20 - Prob. 16ACh. 20 - Prob. 17ACh. 20 - Prob. 18ACh. 20 - Prob. 19ACh. 20 - Prob. 20ACh. 20 - Prob. 21ACh. 20 - Prob. 22ACh. 20 - Prob. 23ACh. 20 - Prob. 24ACh. 20 - Prob. 25ACh. 20 - Prob. 26ACh. 20 - Prob. 27ACh. 20 - Prob. 28ACh. 20 - Prob. 29ACh. 20 - Prob. 30ACh. 20 - Prob. 31ACh. 20 - Prob. 32ACh. 20 - Prob. 33ACh. 20 - Prob. 34ACh. 20 - Prob. 35ACh. 20 - Prob. 36ACh. 20 - Prob. 37ACh. 20 - Prob. 38ACh. 20 - Prob. 39ACh. 20 - Prob. 40ACh. 20 - Prob. 41ACh. 20 - Prob. 42ACh. 20 - Prob. 43ACh. 20 - Prob. 44ACh. 20 - Prob. 45ACh. 20 - Prob. 46ACh. 20 - Prob. 47ACh. 20 - Prob. 48ACh. 20 - Prob. 49ACh. 20 - Prob. 50ACh. 20 - Prob. 51ACh. 20 - Prob. 52ACh. 20 - Prob. 53ACh. 20 - Prob. 54ACh. 20 - Prob. 55ACh. 20 - Prob. 56ACh. 20 - Prob. 57ACh. 20 - Prob. 58ACh. 20 - Prob. 59ACh. 20 - Prob. 60ACh. 20 - Prob. 61ACh. 20 - Prob. 62ACh. 20 - Prob. 63ACh. 20 - Prob. 64ACh. 20 - Prob. 65ACh. 20 - Prob. 66ACh. 20 - Prob. 67ACh. 20 - Prob. 68ACh. 20 - Prob. 69ACh. 20 - Prob. 70ACh. 20 - Prob. 71ACh. 20 - Prob. 72ACh. 20 - Prob. 73ACh. 20 - Prob. 1STPCh. 20 - Prob. 2STPCh. 20 - Prob. 3STPCh. 20 - Prob. 4STPCh. 20 - Prob. 5STPCh. 20 - Prob. 6STPCh. 20 - Prob. 7STPCh. 20 - Prob. 8STPCh. 20 - Prob. 9STP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Indicate whether the product of the reaction between Naphthalene and CrO3 in acetic acid at 25ºC is 1,4 naphthoquinone or phthalic anhydride.arrow_forwardIndicate the products of the reaction between CH3COCH2COOC2H5 and Na+-OC2H5.arrow_forwardPrimary, Secondary, and Tertiary Alcohols O-H O-H O-H R₁-C-H R₁-C-H R₁-C-R₁ H R₂ R₂ Primary Alcohol Secondary Alcohol ChemistryLearner.com R stands for Carbon group like ethyl methyl propyl Tertiary Alcohol If 1 carbon group with two H attached to alcoholic carbon, then primary If 2 carbon group and 1 H are attached to alcoholic carbon, then secondary IF 3 carbon group and no H attach to alcoholic carbon then tertiary. The bottom line Starting "Weak" oxidant material PCC, DMP, Swern, etc Primary alcohol Aldehyde OH Secondary alcohol Ketone OH "Strong" oxidant KMnO4, H₂CrO4 (or equivalent) OH Carboxylic acid 요 Ketone No reaction No reaction Tertiary alcohol 1. Is ethanol a primary, secondary, or tertiary alcohol? Write out the structures of ethanol and any oxidation products of ethanol. If there is more than one oxidation product, give the structure of each of the products. 2. Is 2-propanol a primary, secondary, or tertiary alcohol? Write out the structures of 2-propanol and any…arrow_forward
- Complete the following equations hand written pleasearrow_forwardComplete the following equations please hand written pleasearrow_forwardUsing the Nernst equation to calculate nonstandard cell voltage A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 3+ 3Cu²+ (aq) +2Al(s) → 3 Cu(s)+2A1³* (aq) 2+ Suppose the cell is prepared with 5.29 M Cu in one half-cell and 2.49 M A1³+ in the other. Calculate the cell voltage under these conditions. Round your answer to 3 significant digits. x10 μ ☑ 00. 18 Ar Иarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY