
Concept explainers
(a)
Interpretation:
In this subpart one must identify the functional group that is present in each molecule and thereby indicate the family of organic compounds to which the compound specifically belongs to.
Concept Introduction:
The International Union of Pure and Applied Chemistry-IUPAC has developed a method for systematically naming organic compounds. The name given to a compound according to the
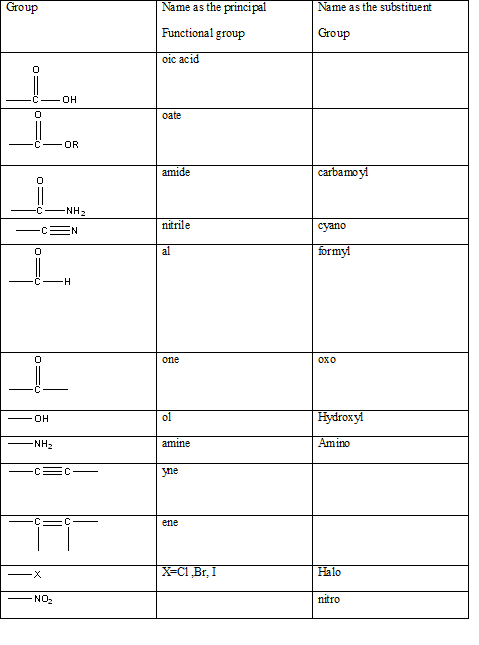
Table 1: The series of functional groups arranged in the decreasing order of the priority.
Answer to Problem 57QAP
Explanation of Solution
- The name given to a compound according to the IUPAC nomenclature consists of several parts.
- The suffix that is used to indicate the main functional group of the structure.
- Name of the chain that is used to identify the main carbon chain of the compound.
- The prefixes that are used to indicate the substituent groups.
- The numbers that are used to indicate the places at which the substituent groups, additional groups and the main functional groups are attached to the chain.
The IUPAC name of an aliphatic compound can easily be developed by following the steps stated below in the given order.
- The main functional group should be identified first.
- The root name for the principal chain should be selected.
- In the name of the chain, the suffix for the double or triple bond in the parent chain should be added.
- The substituent groups should be named.
- The name of the substituent groups should be added in the name of chain.
- The carbon atoms in the chain should be numbered.
- The minimum possible number is given to the main functional group present as a substituent in the chain.
| Number of carbon atoms | Root name | Name of the corresponding |
| 1 | meth | Methane |
| 2 | eth | Ethane |
| 3 | prop | Propane |
| 4 | but | Butane |
| 5 | pent | Pentane |
| 6 | hex | Hexane |
Table 2: The root names used for the compounds according to the number of carbon atoms and the names of the corresponding hydrocarbons.
According to this explanation-NH2 is the functional group of
(b)
Interpretation:
In this subpart one must identify the functional group that is present in each molecule and thereby indicate the family of organic compounds to which the compound specifically belongs to.
Concept Introduction:
The International Union of Pure and Applied Chemistry-IUPAC has developed a method for systematically naming organic compounds. The name given to a compound according to the IUPAC nomenclature consists of several parts the critical or the first part is to identify the main functional group present in a molecule in order to name it correctly. It allows the identification of the class of the compound easily possible without any ambiguity. The table indicating the main functional groups present in organic molecules can be summarized in the table below.
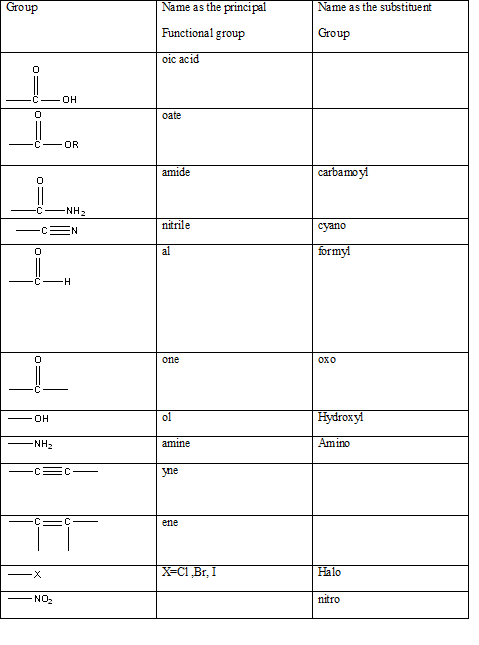
Table 1: The series of functional groups arranged in the decreasing order of the priority.
Answer to Problem 57QAP
Explanation of Solution
- The name given to a compound according to the IUPAC nomenclature consists of several parts.
- The suffix that is used to indicate the main functional group of the structure.
- Name of the chain that is used to identify the main carbon chain of the compound.
- The prefixes that are used to indicate the substituent groups.
- The numbers that are used to indicate the places at which the substituent groups, additional groups and the main functional groups are attached to the chain.
- The IUPAC name of an aliphatic compound can easily be developed by following the steps stated below in the given order.
- Identifying the principal functional group.
- Selecting the main chain.
- Selecting the root name for the principal chain.
- Addition of the suffix for the double/triple bond in the main carbon chain to the name of the chain.
- Addition of the suffix used to indicate the principal functional group to the name of the chain.
- Naming the substituent groups.
- Adding the names of the substituent groups to the name of the chain.
- Numbering the carbon chain.
- Writing the numbers that are used to indicate the positions of the main functional group and the substituent groups in front of these groups.
| Number of carbon atoms | Root name | Name of the corresponding |
| 1 | meth | Methane |
| 2 | eth | Ethane |
| 3 | prop | Propane |
| 4 | but | Butane |
| 5 | pent | Pentane |
| 6 | hex | Hexane |
Table 2: The root names used for the compounds according to the number of carbon atoms and the names of the corresponding hydrocarbons.
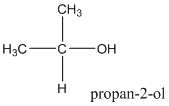
According to this explanation-OH is the functional group of an alcohol. This functional group is present in the main carbon chain of the example compound and hence resulting in absence of any substituent groups. Since there are only three carbon atoms in the main chain one should use the root name −prop in naming the compound. Also the −OH functional group is connected to the second carbon of the main chain and hence one must also indicate the position of −OH by using 2- as well.
(c)
Interpretation:
In this subpart one must identify the functional group that is present in each molecule and thereby indicate the family of organic compounds to which the compound specifically belongs to.
Concept Introduction:
The International Union of Pure and Applied Chemistry-IUPAC has developed a method for systematically naming organic compounds. The name given to a compound according to the IUPAC nomenclature consists of several parts the critical or the first part is to identify the main functional group present in a molecule in order to name it correctly. It allows the identification of the class of the compound easily possible without any ambiguity. The table indicating the main functional groups present in organic molecules can be summarized in the table below.
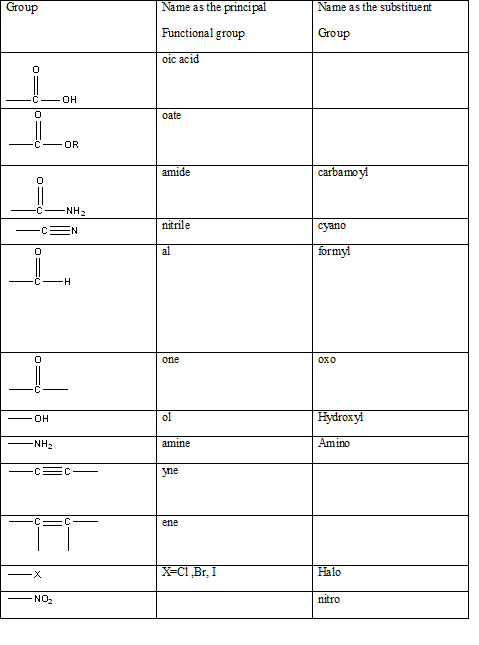
Table 1: The series of functional groups arranged in the decreasing order of the priority.
Answer to Problem 57QAP
Explanation of Solution
- The name given to a compound according to the IUPAC nomenclature consists of several parts.
- The suffix that is used to indicate the main functional group of the structure.
- Name of the chain that is used to identify the main carbon chain of the compound.
- The prefixes that are used to indicate the substituent groups.
- The numbers that are used to indicate the places at which the substituent groups, additional groups and the main functional groups are attached to the chain. The IUPAC name of an aliphatic compound can easily be developed by following the steps stated below in the given order.
- The main functional group should be identified first.
| Number of carbon atoms | Root name | Name of the corresponding |
| 1 | meth | Methane |
| 2 | eth | Ethane |
| 3 | prop | Propane |
| 4 | but | Butane |
| 5 | pent | Pentane |
| 6 | hex | Hexane |
Table 2: The root names used for the compounds according to the number of carbon atoms and the names of the corresponding hydrocarbons.
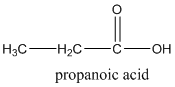
According to this explanation-COOH is the functional group of a
(d)
Interpretation:
In this subpart one must identify the functional group that is present in each molecule and thereby indicate the family of organic compounds to which the compound specifically belongs to.
Concept Introduction:
The International Union of Pure and Applied Chemistry-IUPAC has developed a method for systematically naming organic compounds. The name given to a compound according to the IUPAC nomenclature consists of several parts the critical or the first part is to identify the main functional group present in a molecule in order to name it correctly. It allows the identification of the class of the compound easily possible without any ambiguity. The table indicating the main functional groups present in organic molecules can be summarized in the table below.
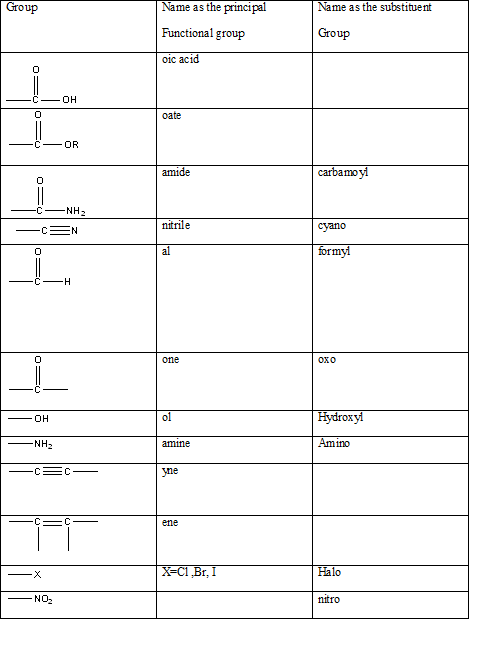
Table 1: The series of functional groups arranged in the decreasing order of the priority.
Answer to Problem 57QAP
Explanation of Solution
- The name given to a compound according to the IUPAC nomenclature consists of several parts.
- The suffix that is used to indicate the main functional group of the structure.
- Name of the chain that is used to identify the main carbon chain of the compound.
- The prefixes that are used to indicate the substituent groups.
- The numbers that are used to indicate the places at which the substituent groups, additional groups and the main functional groups are attached to the chain.
The IUPAC name of an aliphatic compound can easily be developed by following the steps stated below in the given order.
- The main functional group should be identified first.
- The root name for the principal chain should be selected.
- In the name of the chain, the suffix for the double or triple bond in the parent chain should be added.
- The substituent groups should be named.
- The name of the substituent groups should be added in the name of chain.
- The carbon atoms in the chain should be numbered.
- The minimum possible number is given to the main functional group present as a substituent in the chain.
| Number of carbon atoms | Root name | Name of the corresponding |
| 1 | meth | Methane |
| 2 | eth | Ethane |
| 3 | prop | Propane |
| 4 | but | Butane |
| 5 | pent | Pentane |
| 6 | hex | Hexane |
Table 2: The root names used for the compounds according to the number of carbon atoms and the names of the corresponding hydrocarbons.
According to this explanation- CHO is the functional group of a carboxylic acid. This functional group is present in the main carbon chain of the example compound and hence resulting in absence of any substituent groups. Since there are only two carbon atoms in the main chain one should use the root name −eth in naming the compound. Also, the −CHO functional group is named as −al in the IUPAC name.
(e)
Interpretation:
In this subpart one must identify the functional group that is present in each molecule and thereby indicate the family of organic compounds to which the compound specifically belongs to.
Concept Introduction:
The International Union of Pure and Applied Chemistry-IUPAC has developed a method for systematically naming organic compounds. The name given to a compound according to the IUPAC nomenclature consists of several parts the critical or the first part is to identify the main functional group present in a molecule in order to name it correctly. It allows the identification of the class of the compound easily possible without any ambiguity. The table indicating the main functional groups present in organic molecules can be summarized in the table below.
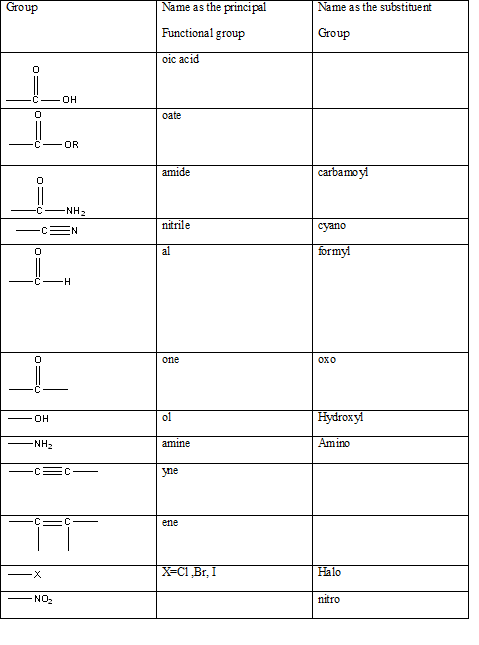
Table 1: The series of functional groups arranged in the decreasing order of the priority.
Answer to Problem 57QAP
Explanation of Solution
- The name given to a compound according to the IUPAC nomenclature consists of several parts.
- The suffix that is used to indicate the main functional group of the structure.
- Name of the chain that is used to identify the main carbon chain of the compound.
- The prefixes that are used to indicate the substituent groups.
- The numbers that are used to indicate the places at which the substituent groups, additional groups and the main functional groups are attached to the chain.
The IUPAC name of an aliphatic compound can easily be developed by following the steps stated below in the given order.
- The main functional group should be identified first.
- The root name for the principal chain should be selected.
- In the name of the chain, the suffix for the double or triple bond in the parent chain should be added.
- The substituent groups should be named.
- The name of the substituent groups should be added in the name of chain.
- The carbon atoms in the chain should be numbered.
- The minimum possible number is given to the main functional group present as a substituent in the chain.
| Number of carbon atoms | Root name | Name of the corresponding |
| 1 | meth | Methane |
| 2 | eth | Ethane |
| 3 | prop | Propane |
| 4 | but | Butane |
| 5 | pent | Pentane |
| 6 | hex | Hexane |
Table 2: The root names used for the compounds according to the number of carbon atoms and the names of the corresponding hydrocarbons.
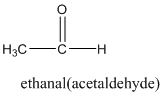
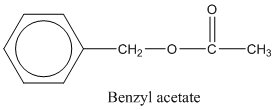
In this case the main functionality is −COOR group. In this particular example the −R group is the benzyl group which has a CH2 group attached to the benzene group.
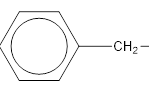
benzyl group.
(f)
Interpretation:
In this subpart one must identify the functional group that is present in each molecule and thereby indicate the family of organic compounds to which the compound specifically belongs to.
Concept Introduction:
The International Union of Pure and Applied Chemistry-IUPAC has developed a method for systematically naming organic compounds. The name given to a compound according to the IUPAC nomenclature consists of several parts the critical or the first part is to identify the main functional group present in a molecule in order to name it correctly. It allows the identification of the class of the compound easily possible without any ambiguity. The table indicating the main functional groups present in organic molecules can be summarized in the table below.
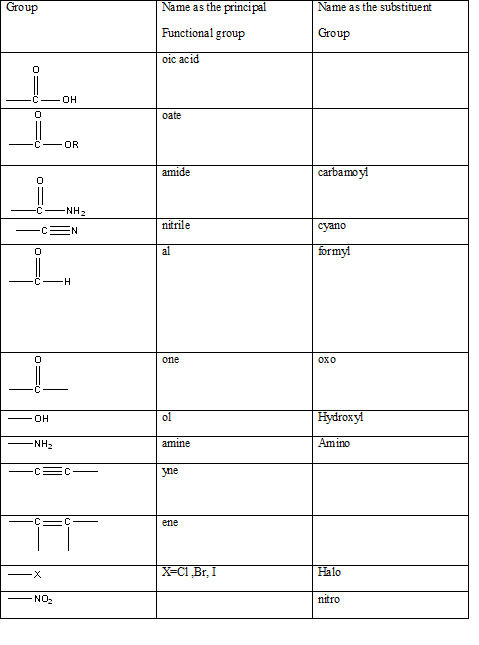
Table 1: The series of functional groups arranged in the decreasing order of the priority.
Answer to Problem 57QAP
Explanation of Solution
- The name given to a compound according to the IUPAC nomenclature consists of several parts.
- The suffix that is used to indicate the main functional group of the structure.
- Name of the chain that is used to identify the main carbon chain of the compound.
- The prefixes that are used to indicate the substituent groups.
- The numbers that are used to indicate the places at which the substituent groups, additional groups and the main functional groups are attached to the chain. The IUPAC name of an aliphatic compound can easily be developed by following the steps stated below in the given order.
- The main functional group should be identified first.
| Number of carbon atoms | Root name | Name of the corresponding |
| 1 | meth | Methane |
| 2 | eth | Ethane |
| 3 | prop | Propane |
| 4 | but | Butane |
| 5 | pent | Pentane |
| 6 | hex | Hexane |
Table 2: The root names used for the compounds according to the number of carbon atoms and the names of the corresponding hydrocarbons.
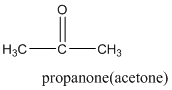
According to this explanation C=O is the functional group of a carboxylic acid. This functional group is present in the main carbon chain of the example compound and hence resulting in absence of any substituent groups. Since there are only three carbon atoms in the main chain one should use the root name −prop in naming the compound. Also, the −C=O functional group is named as −one in the IUPAC name.
(g)
Interpretation:
In this subpart one must identify the functional group that is present in each molecule and thereby indicate the family of organic compounds to which the compound specifically belongs to.
Concept Introduction:
The International Union of Pure and Applied Chemistry-IUPAC has developed a method for systematically naming organic compounds. The name given to a compound according to the IUPAC nomenclature consists of several parts the critical or the first part is to identify the main functional group present in a molecule in order to name it correctly. It allows the identification of the class of the compound easily possible without any ambiguity. The table indicating the main functional groups present in organic molecules can be summarized in the table below.
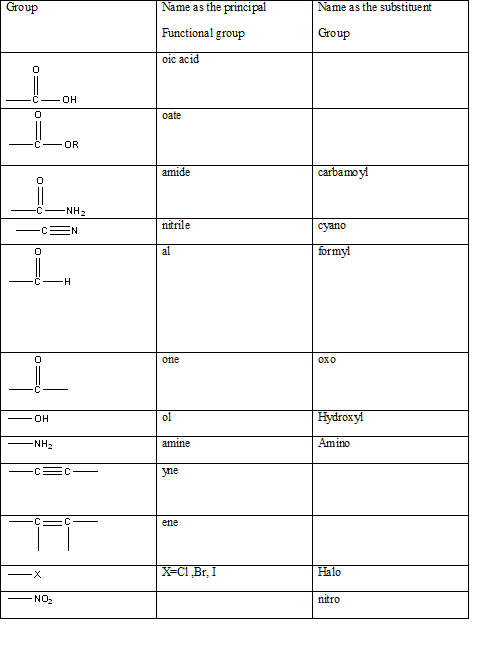
Table 1: The series of functional groups arranged in the decreasing order of the priority.
Answer to Problem 57QAP
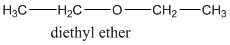
Explanation of Solution
- The name given to a compound according to the IUPAC nomenclature consists of several parts.
- The suffix that is used to indicate the main functional group of the structure.
- Name of the chain that is used to identify the main carbon chain of the compound.
- The prefixes that are used to indicate the substituent groups.
- The numbers that are used to indicate the places at which the substituent groups, additional groups and the main functional groups are attached to the chain. The IUPAC name of an aliphatic compound can easily be developed by following the steps stated below in the given order.
- The main functional group should be identified first.
| Number of carbon atoms | Root name | Name of the corresponding |
| 1 | meth | Methane |
| 2 | eth | Ethane |
| 3 | prop | Propane |
| 4 | but | Butane |
| 5 | pent | Pentane |
| 6 | hex | Hexane |
Table 2: The root names used for the compounds according to the number of carbon atoms and the names of the corresponding hydrocarbons.
A compound belonging to the class ether has the general form of R-O-R1 where R, R1 are the alkyl groups. R and R1 groups can be the same like in this example where R- group is the ethyl group containing two carbons in the alkyl chain.
Want to see more full solutions like this?
Chapter 20 Solutions
EBK INTRO.CHEMISTRY (NASTA EDITION)
- Is an acid-base reaction the only type of reaction that would cause leavening products to rise?arrow_forwardHelp me understand this! Thank you in advance.arrow_forward22.22 For each compound, indicate which group on the ring is more strongly activating and then draw a structural formula of the major product formed by nitration of the compound. Br CHO (a) CH3 (b) (c) CHO CH3 SO₂H (d) ☑ OCHS NO₂ (e) (f) CO₂H NHCOCH3 NHCOCH, (h) CHS 22.23 The following molecules each contain two aromatic rings. (b) 000-100- H3C (a) (c) Which ring in each undergoes electrophilic aromatic substitution more readily? Draw the major product formed on nitration.arrow_forward
- V Consider this step in a radical reaction: Br: ? What type of step is this? Check all that apply. Draw the products of the step on the right-hand side of the drawing area below. If more than one set of products is possible, draw any set. Also, draw the mechanism arrows on the left-hand side of the drawing area to show how this happens. ⚫ionization termination initialization neutralization none of the abc Explanation Check 80 Ο F3 F1 F2 2 F4 01 % do5 $ 94 #3 X 5 C MacBook Air 25 F5 F6 66 ©2025 ˇ F7 29 & 7 8arrow_forwardShow how to convert ethyl benzene to (a) 2,5-dichlorobenzoic acid and (b) 2,4-dichlorobenzoic acid.arrow_forwardno aiarrow_forward
- Polymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer formed in this reaction. Assume there are hydrogen atoms there are hydrogen atoms on the two ends of the trimer. Ignore inorganic byproducts.arrow_forwardDraw a tetramer if this alternating copolymer pleasearrow_forwardDraw the monomers required to synthesize this condensation polymer.arrow_forward
- Draw the monomers required to synthesize this condensation polymer.arrow_forward8:44 PM Sun Apr 13 Earn Freecash.com O Measurement and Matter =1 Setting up a unit conversion 110 Eddie says... ✰ www-awu.aleks.com A student sets up the following equation to convert a measurement. (The ? stands for a number the student is going to calculate.) Fill in the missing part of this equation. Note: your answer should be in the form of one or more fractions multiplied together. (- 4 J kJ -7.0 × 10 ☐ = ? mmol.°C mol °C x10 μ Explanation Check □·□ torox.io Grey Hill LLC. All Rightsarrow_forwardPolymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer formed in this reaction. Assume there are hydrogen atoms there are hydrogen atoms on the two ends of the trimer. Ignore inorganic byproducts please.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning  Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER





