
Organic Chemistry-Package(Custom)
4th Edition
ISBN: 9781259141089
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 18.54P
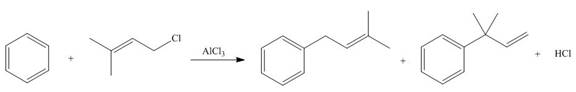
Draw a stepwise mechanism for the following reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When talking about the acidity of carboxylic acids, is it the same thing to say higher or stronger acidity?
Using the following two half-reactions, determine the pH range in which $NO_2^-\ (aq)$ cannot be found as the predominant chemical species in water.* $NO_3^-(aq)+10H^+(aq)+8e^-\rightarrow NH_4^+(aq)+3H_2O(l),\ pE^{\circ}=14.88$* $NO_2^-(aq)+8H^+(aq)+6e^-\rightarrow NH_4^+(aq)+2H_2O(l),\ pE^{\circ}=15.08$
Indicate characteristics of oxodec acid.
Chapter 18 Solutions
Organic Chemistry-Package(Custom)
Ch. 18 - Prob. 18.1PCh. 18 - Prob. 18.2PCh. 18 - Prob. 18.3PCh. 18 - Prob. 18.4PCh. 18 - Prob. 18.5PCh. 18 - What acid chloride would be needed to prepare each...Ch. 18 - Prob. 18.7PCh. 18 - Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.9PCh. 18 - Prob. 18.10P
Ch. 18 - Prob. 18.11PCh. 18 - Prob. 18.12PCh. 18 - Problem 18.14 Draw all resonance structures for...Ch. 18 - Classify each substituent as electron donating or...Ch. 18 - Prob. 18.15PCh. 18 - Label each compound as more or less reactive than...Ch. 18 - Rank the following compounds in order of...Ch. 18 - Problem 18.18 Rank the following compounds in...Ch. 18 - Prob. 18.19PCh. 18 - Problem 18.20 Draw the products of each...Ch. 18 - Prob. 18.21PCh. 18 - Prob. 18.22PCh. 18 - Devise a synthesis of each compound from the...Ch. 18 - Problem 18.24 Draw the products of each...Ch. 18 - Problem 18.25 Draw a stepwise mechanism for the...Ch. 18 - Problem 18.26 Draw the products of each...Ch. 18 - Prob. 18.27PCh. 18 - Prob. 18.28PCh. 18 - Problem 18.29 How could you use ethylbenzene to...Ch. 18 - Prob. 18.30PCh. 18 - Problem 18.31 What steps are needed to convert...Ch. 18 - Problem 18.32 Synthesize each compound from...Ch. 18 - Synthesize each compound from benzene.Ch. 18 - Prob. 18.34PCh. 18 - 18.35 What is the major product formed by an...Ch. 18 - Draw the products formed when phenol (C6H5OH) is...Ch. 18 - Prob. 18.37PCh. 18 - Draw the products of each reaction. a. e. i. b. f....Ch. 18 - What products are formed when benzene is treated...Ch. 18 - Draw the products of each reaction. a.d. b.e. c.f.Ch. 18 - You have learned two ways to make an alkyl...Ch. 18 - Prob. 18.42PCh. 18 - Prob. 18.43PCh. 18 - 18.45 Explain why each of the following reactions...Ch. 18 - Prob. 18.45PCh. 18 - 18.47 For each of the following substituted...Ch. 18 - Which benzene ring in each compound is more...Ch. 18 - 18.49 For each N-substituted benzene, predict...Ch. 18 - Explain each statement in detail using resonance...Ch. 18 - Prob. 18.50PCh. 18 - Explain why the meta product is formed in the...Ch. 18 - 18.53 Rank the aryl halides in each group in order...Ch. 18 - Prob. 18.53PCh. 18 - 18.54 Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.55PCh. 18 - 18.56 Draw a stepwise, detailed mechanism for the...Ch. 18 - Prob. 18.57PCh. 18 - 18.58 Draw a stepwise mechanism for the following...Ch. 18 - Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.60PCh. 18 - Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.62PCh. 18 - Prob. 18.63PCh. 18 - Synthesize each compound from benzene and any...Ch. 18 - Synthesize each compound from benzene and any...Ch. 18 - Synthesize each compound from toluene (C6H5CH3)...Ch. 18 - Prob. 18.67PCh. 18 - Use the reactions in this chapter along with those...Ch. 18 - Prob. 18.69PCh. 18 - Prob. 18.70PCh. 18 - Problem 18.69 Identify the structures of isomers A...Ch. 18 - Prob. 18.72PCh. 18 - Problem 18.71 Compound X (molecular formula ) was...Ch. 18 - Prob. 18.74PCh. 18 - The NMR spectrum of phenol () shows three...Ch. 18 - Explain the reactivity and orientation effects...Ch. 18 - Prob. 18.77PCh. 18 - Prob. 18.78PCh. 18 - Prob. 18.79PCh. 18 - Prob. 18.80P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the final product when hexanedioic acid reacts with 1º PCl5 and 2º NH3.arrow_forwardWhat is the final product when D-galactose reacts with hydroxylamine?arrow_forwardIndicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.arrow_forward
- The temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forwardQUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY