
Introduction To General, Organic, And Biochemistry
12th Edition
ISBN: 9781337571357
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.4, Problem 16.7QC
Problem 17-7
Identify all hemiacetals and acetals in the following structures and tell whether each is formed from an
OH
(a) 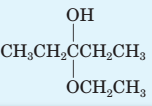
(b) CH3OCH2CH2OCH3
(c) 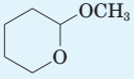
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Synthesize the following:
Did you report your data to the correct number of significant
figures?
Temperature of cold water (°C)
4.0
Temperature of hot water ("C)
87.0
Volume of cold water (mL)
94.0
Volume of hot water (mL)
78.0
Final temperature after mixing ("C)
41.0
Mass of cold water (g)
94.0
Mass of hot water (g)
78.0
Calorimeter constant (J/°C)
12.44
How to calculate the calorimeter constant
please draw the arrows
Chapter 16 Solutions
Introduction To General, Organic, And Biochemistry
Ch. 16.2 - Problem 17-1 Wrtie the IUPAC name for each...Ch. 16.2 - Prob. 16.2QCCh. 16.2 - Prob. 16.3QCCh. 16.4 - Prob. 16.4QCCh. 16.4 - Prob. 16.5QCCh. 16.4 - Problem 17-6 Show the reaction of benzaldehyde...Ch. 16.4 - Problem 17-7 Identify all hemiacetals and acetals...Ch. 16.5 - Prob. 16.8QCCh. 16 - 17-9 Answer true or false. (a) The one aldehyde...Ch. 16 - Prob. 2P
Ch. 16 - 17-11 What is the difference in structure between...Ch. 16 - 17-12 Is it possible for the carbon atom of a...Ch. 16 - 17-13 Which compounds contain carbonyl groups?Ch. 16 - 17-14 Following are structural formulas for two...Ch. 16 - 17-15 Draw structural formulas for the four...Ch. 16 - Prob. 8PCh. 16 - Prob. 9PCh. 16 - 17-18 Draw structural formulas for these ketones....Ch. 16 - 17-19 Write the JUPAC names for these compounds.Ch. 16 - Prob. 12PCh. 16 - 17-2 1 Explain why each name is incorrect. Write...Ch. 16 - Prob. 14PCh. 16 - Prob. 15PCh. 16 - 17-24 In each pair of compounds, select the one...Ch. 16 - Prob. 17PCh. 16 - 17-26 Account for the fact that acetone has a...Ch. 16 - 17-27 Pentane, 1-butanol, and butanal all have...Ch. 16 - 17-28 Show how acetaldehyde can form hydrogen...Ch. 16 - 17-29 Why can’t two molecules of acetone form a...Ch. 16 - 17-30 Answer true or false. (a) The reduction of...Ch. 16 - 17-3 1 Draw a structural formula for the principal...Ch. 16 - Prob. 24PCh. 16 - 17-33 What simple chemical test could you use to...Ch. 16 - 17-34 Explain why liquid aldehydes are often...Ch. 16 - 17-35 Suppose that you take a bottle of...Ch. 16 - 17-36 Explain why the reduction of an aldehyde...Ch. 16 - Prob. 29PCh. 16 - Prob. 30PCh. 16 - Prob. 31PCh. 16 - Prob. 32PCh. 16 - Prob. 33PCh. 16 - Prob. 34PCh. 16 - Prob. 35PCh. 16 - Prob. 36PCh. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - 17-47 What is the characteristic structural...Ch. 16 - Prob. 40PCh. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - Prob. 43PCh. 16 - Prob. 44PCh. 16 - Prob. 45PCh. 16 - 17-54 Following is the structure of...Ch. 16 - Prob. 47PCh. 16 - Prob. 48PCh. 16 - Prob. 49PCh. 16 - Prob. 50PCh. 16 - Prob. 51PCh. 16 - 17-60 1-Propanol can be prepared by the reduction...Ch. 16 - Prob. 53PCh. 16 - 17-62 Show how to bring about these conversions....Ch. 16 - Prob. 55PCh. 16 - Prob. 56PCh. 16 - Prob. 57PCh. 16 - Prob. 58PCh. 16 - 17-67 Draw structural formulas for these...Ch. 16 - Prob. 60PCh. 16 - 17-69 Propanal (bp 49°C) and 1-propanol (bp 97°C)...Ch. 16 - 17-70 What simple chemical test could you use to...Ch. 16 - Prob. 63PCh. 16 - Prob. 64PCh. 16 - Prob. 65PCh. 16 - 17-72 The following molecule is an enediol; each...Ch. 16 - 17-73 Alcohols can be prepared by the...Ch. 16 - 17-74 Glucose, C6H12O6, contains an aldehyde group...Ch. 16 - Prob. 69PCh. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - 17-78 Complete the following equation for these...Ch. 16 - 17-79 Write an equation for each conversion. (a)...Ch. 16 - Prob. 74P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Part 1. Draw monomer units of the following products and draw their reaction mechanism (with arrow pushing) Temporary cross-linked polymer Using: 4% polyvinyl alcohol+ methyl red + 4% sodium boratearrow_forwardcan you please answer both these questions and draw the neccesaryarrow_forwardcan you please give the answer for both these pictures. thankyouarrow_forward
- Part 1. Draw monomer units of the following products and draw their reaction mechanism (with arrow pushing) | Bakelite like polymer Using: Resorcinol + NaOH + Formalinarrow_forwardQuestion 19 0/2 pts 3 Details You have a mixture of sodium chloride (NaCl) and potassium chloride (KCl) dissolved in water and want to separate out the Cl- ions by precipitating them out using silver ions (Ag+). The chemical equation for the net ionic reaction of NaCl and KCl with silver nitrate, AgNO3, is shown below. Ag+(aq) + Cl(aq) → AgCl(s) The total mass of the NaCl/KCl mixture is 1.299 g. Adding 50.42 mL of 0.381 M solution precipitates out all of the Cl-. What are the masses of NaCl and KCl in the mixture? Atomic masses: g: Mass of NaCl g: Mass of KCL Ag = 107.868 g mol- 1 Cl = 35.453 g mol- 1 K = 39.098 g mol- N = 14.007 g mol−1 Na = 22.99 g mol−1 0 = 15.999 g mol 1 Question Help: ✓ Message instructor Submit Questionarrow_forwardPart 1. Draw monomer units of the following products and draw their reaction mechanism (with arrow pushing) Polyester fiber Using a) pthalic anhydride + anhydrous sodium acetate + ethylene glycol B)pthalic anhydride + anhydrous sodium acetate + glycerolarrow_forward
- Identify the missing starting materials/ reagents/ products in the following reactions. Show the stereochemistry clearly in the structures, if any. If there is a major product, draw the structures of the major product with stereochemistry clearly indicated where applicable. Show only the diastereomers (you do not have to draw the pairs of enantiomers). If you believe that multiple products are formed in approximately equal amounts (hence neither is the major product), draw the structures of the products, and show the detailed mechanism of these reactions to justify the formation of the multiple products. If you believe no product is formed, explain why briefly. (6 mark for each, except f and g, which are 10 mark each)arrow_forward3. What starting material would you use to synthesize 3-hydroxypentanoic acid using a NaBH4 reduction?arrow_forward1. Give stereochemical (Fischer projection) formulas for all (but no extras) the stereoisomers that could theoretically form during the reduction of a. the carbonyl group of 2-methyl-3--pentanone b. both carbonyl groups of 2,4-pentanedione (careful!) 2. Predict the products of the reduction of O=CCH2CH2CH2C=O with a. LiAlH4 b. NaBH4 CH3 OHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License