
Chemistry: Principles and Practice
3rd Edition
ISBN: 9780534420123
Author: Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 13, Problem 13.23QE
Nitrogen monoxide reacts with chlorine to form nitrosyl chloride.
The figure shows the increase in nitrosyl chloride concentration under appropriate experimental conditions. The concentration of nitrosyl chloride actually starts at zero, although this fact may be difficult to see in the figure.
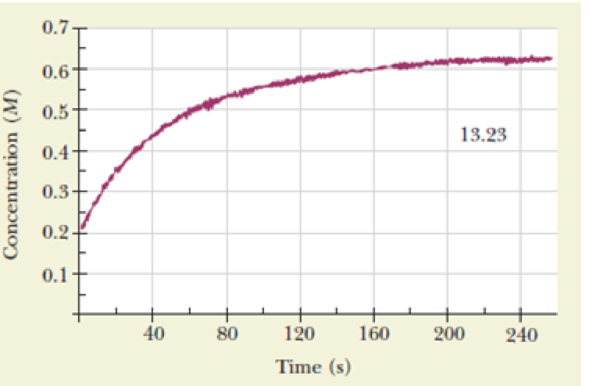
- (a) Write an expression for the
rate of reaction in terms of a changing concentration. - (b) Calculate the average rate of reaction between 40 and 120 seconds.
- (c) Calculate the instantaneous rate of reaction after 80 seconds.
- (d) Calculate the instantaneous rate of consumption of chlorine 60 seconds after the start of the reaction.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Draw the most likely mechanism for the following:
Calculate the number of moles of HI that are at equilibrium with 4.9 mol of H2 and 4.9 mol of I2 in a 5.00-L flask at 448 °C. (those are the equilibrium values)
Ε.
HO
HO
Chapter 13 Solutions
Chemistry: Principles and Practice
Ch. 13 - Prob. 13.1QECh. 13 - Prob. 13.2QECh. 13 - What is the difference between the integrated and...Ch. 13 - Prob. 13.4QECh. 13 - Explain why half-lives are not normally used to...Ch. 13 - Derive an expression for the half-life of a...Ch. 13 - Prob. 13.7QECh. 13 - Prob. 13.8QECh. 13 - Prob. 13.9QECh. 13 - Prob. 13.10QE
Ch. 13 - Prob. 13.11QECh. 13 - Prob. 13.12QECh. 13 - Prob. 13.13QECh. 13 - Prob. 13.14QECh. 13 - Prob. 13.15QECh. 13 - Prob. 13.16QECh. 13 - Prob. 13.17QECh. 13 - Prob. 13.18QECh. 13 - Prob. 13.19QECh. 13 - Prob. 13.20QECh. 13 - Prob. 13.21QECh. 13 - Prob. 13.22QECh. 13 - Nitrogen monoxide reacts with chlorine to form...Ch. 13 - Prob. 13.24QECh. 13 - Prob. 13.25QECh. 13 - Prob. 13.26QECh. 13 - Prob. 13.27QECh. 13 - Prob. 13.28QECh. 13 - Prob. 13.29QECh. 13 - Prob. 13.30QECh. 13 - Prob. 13.31QECh. 13 - Prob. 13.32QECh. 13 - Prob. 13.33QECh. 13 - Write a rate law for NO3(g) + O2(g) NO2(g) +...Ch. 13 - Prob. 13.35QECh. 13 - Prob. 13.36QECh. 13 - Prob. 13.37QECh. 13 - Rate data were obtained at 25 C for the following...Ch. 13 - Prob. 13.39QECh. 13 - Prob. 13.40QECh. 13 - Prob. 13.41QECh. 13 - Prob. 13.42QECh. 13 - Prob. 13.43QECh. 13 - Prob. 13.44QECh. 13 - Prob. 13.45QECh. 13 - Prob. 13.46QECh. 13 - Prob. 13.47QECh. 13 - Prob. 13.48QECh. 13 - When formic acid is heated, it decomposes to...Ch. 13 - Prob. 13.50QECh. 13 - The half-life of tritium, 3H, is 12.26 years....Ch. 13 - Prob. 13.52QECh. 13 - Prob. 13.53QECh. 13 - Prob. 13.54QECh. 13 - Prob. 13.55QECh. 13 - Prob. 13.56QECh. 13 - The decomposition of ozone is a second-order...Ch. 13 - Prob. 13.58QECh. 13 - Prob. 13.59QECh. 13 - Prob. 13.60QECh. 13 - A reaction rate doubles when the temperature...Ch. 13 - Prob. 13.62QECh. 13 - Prob. 13.63QECh. 13 - Prob. 13.64QECh. 13 - Prob. 13.65QECh. 13 - The activation energy for the decomposition of...Ch. 13 - Prob. 13.67QECh. 13 - Prob. 13.68QECh. 13 - Prob. 13.69QECh. 13 - Prob. 13.70QECh. 13 - Prob. 13.71QECh. 13 - Prob. 13.72QECh. 13 - Prob. 13.73QECh. 13 - Prob. 13.74QECh. 13 - Prob. 13.75QECh. 13 - Prob. 13.76QECh. 13 - Prob. 13.77QECh. 13 - Prob. 13.78QECh. 13 - Prob. 13.79QECh. 13 - Prob. 13.80QECh. 13 - The gas-phase reaction of nitrogen monoxide with...Ch. 13 - Prob. 13.82QECh. 13 - Prob. 13.83QECh. 13 - A catalyst reduces the activation energy of a...Ch. 13 - Prob. 13.85QECh. 13 - Prob. 13.86QECh. 13 - Prob. 13.87QECh. 13 - Prob. 13.88QECh. 13 - Prob. 13.89QECh. 13 - Prob. 13.90QECh. 13 - Prob. 13.91QECh. 13 - Prob. 13.92QECh. 13 - Prob. 13.93QECh. 13 - Prob. 13.94QECh. 13 - Prob. 13.95QECh. 13 - Prob. 13.96QECh. 13 - Prob. 13.98QECh. 13 - Prob. 13.99QE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- a. OH H₂N-O -Ph H+ acyclic productarrow_forwardeks.com/aleksogi/x/sl.exe/1o_u-IgNslkr7j8P3jH-IQs_pBanHhvTCeeBZbufuBYTI0Hz7m7D3ZS17Hd6m-HIl6n52njJN-TXdQA2X9yID-1SWQJTgnjARg30 111 States of Matter Understanding conceptual components of the enthalpy of solution 0/5 Ge A small amount of acetonitrile (CH, CN) is dissolved in a large amount of water. Imagine separating this process into the four stages sketched below. (These sketches show only a portion of the substances, so you can see the density and distribution of atoms and molecules in them.) CH,CN H₂O B 88 C Use these sketches to answer the questions in the table below. The enthalpy of solution AH is negative soln when CH3CN dissolves in water. Use this information to list the stages in order of increasing enthalpy. Would heat be absorbed or released if the system moved from Stage C to D? What force would oppose or favor the system moving from Stage C to D? Check all that apply. 1 absorbed O released neither absorbed nor released. none O ionic bonding force covalent bonding force…arrow_forwardIn a system with an anodic overpotential, the variation of ŋ as a function of the current density: 1. at low fields is linear 2. at higher fields, it follows Tafel's law Find the range of current densities for which the overpotential has the same value as when calculated for cases 1 and 2 (maximum relative difference of 5% with respect to the behavior for higher fields). To which overpotential range does this correspond? Data: 10 = 1.5 mA cm², T = 300°C, ẞ = 0.64, R = 8.314 J K 1 mol¹ and F = 96485 C mol-1.arrow_forward
- Indicate 10.6 with only one significant figure.arrow_forwardIf I have 10 data points for variables x and y, when I represent y versus x I obtain a line with the equation y = mx + b. Is the slope m equal to dy/dx?arrow_forwardThe data for the potential difference of a battery and its temperature are given in the table. Calculate the entropy change in J mol-1 K-1 (indicate the formulas used).Data: F = 96485 C mol-1arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning  Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Kinetics: Initial Rates and Integrated Rate Laws; Author: Professor Dave Explains;https://www.youtube.com/watch?v=wYqQCojggyM;License: Standard YouTube License, CC-BY