
Concept explainers
(a)
Interpretation:
To predict bond angles about each highlighted carbon.
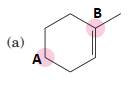
Concept Introduction:
VSEPR predicts the geometry and the bond angles for the molecules according to the theory, the structure is predicted by the lone pairs and bond pairs present in the bond formation.
Electron pair = Lone pair + Bond pair.
| Electron pair | Hybridization | Geometry | Bond angles. |
| 2 | sp | Linear | 180° |
| 3 | sp2 | Trigonal planar | 120° |
| 4 | sp3 | Tetrahedra or square planar | 109.5° or 90° |
| 5 | sp3 d | Trigonal bipyramidal | 120° and 90° |
| 6 | sp3 d2 | Octahedral | 90° |
(b)
Interpretation:
To predict bond angles about each highlighted carbon.
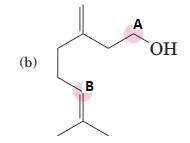
Concept Introduction:
VSEPR predicts the geometry and the bond angles for the molecules according to the theory, the structure is predicted by the lone pairs and bond pairs present in the bond formation.
Electron pair = Lone pair + Bond pair.
| Electron pair | Hybridization | Geometry | Bond angles. |
| 2 | sp | Linear | 180° |
| 3 | sp2 | Trigonal planar | 120° |
| 4 | sp3 | Tetrahedra or square planar | 109.5° or 90° |
| 5 | sp3 d | Trigonal bipyramidal | 120° and 90° |
| 6 | sp3 d2 | Octahedral | 90° |
(c)
Interpretation:
To predict bond angles about each highlighted carbon.
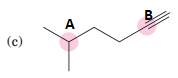
Concept Introduction:
VSEPR predicts the geometry and the bond angles for the molecules according to the theory, the structure is predicted by the lone pairs and bond pairs present in the bond formation.
Electron pair = Lone pair + Bond pair.
| Electron pair | Hybridization | Geometry | Bond angles. |
| 2 | sp | Linear | 180° |
| 3 | sp2 | Trigonal planar | 120° |
| 4 | sp3 | Tetrahedra or square planar | 109.5° or 90° |
| 5 | sp3 d | Trigonal bipyramidal | 120° and 90° |
| 6 | sp3 d2 | Octahedral | 90° |
(d)
Interpretation:
To predict bond angles about each highlighted carbon.
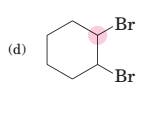
Concept Introduction:
VSEPR predicts the geometry and the bond angles for the molecules according to the theory, the structure is predicted by the lone pairs and bond pairs present in the bond formation.
Electron pair = Lone pair + Bond pair.
| Electron pair | Hybridization | Geometry | Bond angles. |
| 2 | sp | Linear | 180° |
| 3 | sp2 | Trigonal planar | 120° |
| 4 | sp3 | Tetrahedra or square planar | 109.5° or 90° |
| 5 | sp3 d | Trigonal bipyramidal | 120° and 90° |
| 6 | sp3 d2 | Octahedral | 90° |
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Introduction To General, Organic, And Biochemistry
- Draw the most stable cations formed in the mass spectrometer by a deavage of the following compound Draw the most stable cations formed in the mass spectrometer by a cleavage of the following compound онarrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting anand product sytucutrs, draw the curved electron-pusing arrows for the following reaction or mechanistic steps. Be sure to account for all bond-breaking and bind-making stepsarrow_forwardDraw the major elimination and substitution products formed in this reavtion. Use a dash or wedge bond to indicatr the stereochemistry of substituents on assymetric centers, wheere applicable. Ignore any inorganic byproducts.arrow_forward
- Draw the two possible products produced in this E2 elimination. Ignore any inorganic byproductsarrow_forwardDraw the major products of this SN1 reaction. Ignore any inorganic byproducts.arrow_forwardDraw the major elimination and substitution products formed in this reaction. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, wehre applicable. Ignore and inorganic byproducts.arrow_forward
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Drawing Arrows THE Problem 33 of 35 N. C:0 Na + Submit Drag To Pan +arrow_forwardDraw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore and inorganic byproducts.arrow_forwardDraw the major producrs of this SN1 reaction. Ignore any inorganic byproducts. Use a dash or wedge bond to indicate the sereochemistry of substituents on asymmetric centers where appllicable.arrow_forward
- 5) Oxaloacetic Acid is an important intermediate in the biosynthesis of citric acid. Synthesize oxaloacetic acid using a mixed Claisen Condensation reaction with two different esters and a sodium ethoxide base. Give your answer as a scheme Hint 1: Your final acid product is producing using a decarboxylation reaction. Hint 2: Look up the structure of oxalic acid. HO all OH oxaloacetic acidarrow_forward20. The Brusselator. This hypothetical system was first proposed by a group work- ing in Brussels [see Prigogine and Lefever (1968)] in connection with spatially nonuniform chemical patterns. Because certain steps involve trimolecular reac tions, it is not a model of any real chemical system but rather a prototype that has been studied extensively. The reaction steps are A-X. B+X-Y+D. 2X+ Y-3X, X-E. 305 It is assumed that concentrations of A, B, D, and E are kept artificially con stant so that only X and Y vary with time. (a) Show that if all rate constants are chosen appropriately, the equations de scribing a Brusselator are: dt A-(B+ 1)x + x²y, dy =Bx-x²y. diarrow_forwardProblem 3. Provide a mechanism for the following transformation: H₂SO A Me. Me Me Me Mearrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning


