
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 46P
For each of the following, write a mechanism that accounts for the reaction shown.
(a)
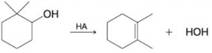
(b)
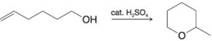
(c)
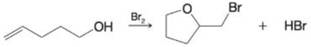
(d)

Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used hand raiting and don't used Ai solution
Q3: Arrange each group of compounds from fastest SN2 reaction rate to slowest SN2
reaction rate.
CI
Cl
H3C-Cl
CI
a)
A
B
C
D
Br
Br
b)
A
B
C
Br
H3C-Br
D
Q4: Rank the relative nucleophilicity of halide ions in water solution and DMF solution,
respectively.
F CI
Br |
Q5: Determine which of the substrates will and will not react with NaSCH3 in an SN2 reaction to
have a reasonable yield of product.
NH2
Br
Br
Br
.OH
Br
Chapter 11 Solutions
Organic Chemistry
Ch. 11 - Prob. 1PPCh. 11 - Practice Problem 11.2 Give bond-line formulas and...Ch. 11 - Prob. 3PPCh. 11 - Prob. 4PPCh. 11 - Prob. 5PPCh. 11 - Prob. 6PPCh. 11 - Prob. 7PPCh. 11 - Practice Problem 11.8 Show how you would prepare...Ch. 11 - Prob. 9PPCh. 11 - Prob. 10PP
Ch. 11 - Practice Problem 11.11
An exception to what is...Ch. 11 - Prob. 12PPCh. 11 - Prob. 13PPCh. 11 - Prob. 14PPCh. 11 - Prob. 15PPCh. 11 - Prob. 16PPCh. 11 - Prob. 17PPCh. 11 - Prob. 18PPCh. 11 - Practice Problem 11.19
Propose structures for each...Ch. 11 - Prob. 20PPCh. 11 - Prob. 21PPCh. 11 - Prob. 22PPCh. 11 - Prob. 23PPCh. 11 - Prob. 24PPCh. 11 - Give an IUPAC substitutive name for each of the...Ch. 11 - Prob. 26PCh. 11 - Prob. 27PCh. 11 - Prob. 28PCh. 11 - Prob. 29PCh. 11 - 11.30. Show how you might prepare 2-bromobutane...Ch. 11 - Prob. 31PCh. 11 - Prob. 32PCh. 11 - Prob. 33PCh. 11 - Considering A-L to represent the major products...Ch. 11 - Write structures for the products that would be...Ch. 11 - Prob. 36PCh. 11 - Provide the reagents necessary for the following...Ch. 11 - 11.38. Predict the major product from each of the...Ch. 11 - Predict the products from each of the following...Ch. 11 - Provide the reagents necessary to accomplish the...Ch. 11 - 11.41. Provide reagents that would accomplish the...Ch. 11 - Prob. 42PCh. 11 - 11.43. A synthesis of the -receptor blocker called...Ch. 11 - Prob. 44PCh. 11 - Prob. 45PCh. 11 - 11.46. For each of the following, write a...Ch. 11 - 11.47. Vicinal halo alcohols (halohydrins) can be...Ch. 11 - Prob. 48PCh. 11 - Prob. 49PCh. 11 - Prob. 50PCh. 11 - Prob. 51PCh. 11 - Prob. 52PCh. 11 - Outlined below is a synthesis of the gypsy moth...Ch. 11 - Prob. 54PCh. 11 - Prob. 55PCh. 11 - Prob. 56PCh. 11 - 11.57. When the 3-bromo-2-butanol with the...Ch. 11 - 11.58. Reaction of an alcohol with thionyl...Ch. 11 - Prob. 59PCh. 11 - Prob. 60PCh. 11 - Prob. 1LGPCh. 11 - Prob. 2LGPCh. 11 - Synthesize the compound shown below from...
Additional Science Textbook Solutions
Find more solutions based on key concepts
8. Studies of DNA support which of the following?
a. Members of the group called australopiths were the first t...
Campbell Biology: Concepts & Connections (9th Edition)
4. How do gross anatomy and microscopic anatomy differ?
Human Anatomy & Physiology (2nd Edition)
2.81 In which of the fo1losing pairs do both numbers contain the same number of significant figures? (2.2)
a....
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
What effect will the concentrated energy from wave erosion eventually have on the shape of the coastline?
Applications and Investigations in Earth Science (9th Edition)
For the reaction shown, find the limiting reactant for each of the initial quantities of reactants. 4Al(s)+3O2(...
Introductory Chemistry (6th Edition)
The smell categories need to be identified for five substances which don’t fall under fishy, minty or sweet cat...
Living By Chemistry: First Edition Textbook
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Classify each molecule as optically active or inactive. Determine the configuration at each H соон Chirality center OH 애 He OH H3C Ноос H H COOH A K B.arrow_forwardQ1: Rank the relative nucleophilicity of the following species in ethanol. CH3O¯, CH3OH, CH3COO, CH3COOH, CH3S Q2: Group these solvents into either protic solvents or aprotic solvents. Acetonitrile (CH3CN), H₂O, Acetic acid (CH3COOH), Acetone (CH3COCH3), CH3CH2OH, DMSO (CH3SOCH3), DMF (HCON(CH3)2), CH3OHarrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward
- 10. The main product of the following reaction is [1.1:4',1"-terphenyl]-2'-yl(1h-pyrazol-4- yl)methanone Ph N-H Pharrow_forwardDraw the Fischer projection for a D-aldo-pentose. (aldehyde pentose). How many total stereoisomers are there? Name the sugar you drew. Draw the Fischer projection for a L-keto-hexose. (ketone pentose). How many total stereoisomers are there? Draw the enantiomer.arrow_forwardDraw a structure using wedges and dashes for the following compound: H- Et OH HO- H H- Me OHarrow_forward
- Which of the following molecules are NOT typical carbohydrates? For the molecules that are carbohydrates, label them as an aldose or ketose. HO Он ОН ОН Он ОН но ΤΗ HO ОН HO eve Он он ОН ОН ОН If polyethylene has an average molecular weight of 25,000 g/mol, how many repeat units are present?arrow_forwardDraw the a-anomer cyclized pyranose Haworth projection of the below hexose. Circle the anomeric carbons. Number the carbons on the Fischer and Haworth projections. Assign R and S for each chiral center. HO CHO -H HO -H H- -OH H -OH CH₂OH Draw the ẞ-anomer cyclized furanose Haworth projection for the below hexose. Circle the anomeric carbons. Number the carbons on the Fischer and Haworth projections. HO CHO -H H -OH HO -H H -OH CH₂OHarrow_forwardName the below disaccharide. Circle any hemiacetals. Identify the numbering of glycosidic linkage, and identify it as a or ẞ. OH HO HO OH HO HO HO OHarrow_forward
- What are the monomers used to make the following polymers? F. а. b. с. d. Вецер хочому なarrow_forward1. Propose a reasonable mechanism for the following transformation. I'm looking for curved mechanistic arrows and appropriate formal charges on intermediates. OMe MeO OMe Me2N NMe2 OTBS OH xylenes OMe 'OTBSarrow_forwardWhat is the polymer made from the following monomers? What type of polymerization is used for each? а. ОН H2N но b. ن -NH2 d. H₂N NH2 довarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License