
Interpretation: The explanation for no formation of indicated alcohols from
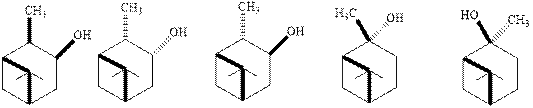
Concept introduction: Hydroboration-oxidation involves a sequence of two reactions. The hydroboration stage involves the treatment of alkene with diborane that generates alkyl borane. In second stage, hydrogen peroxide in alkaline medium is used to oxidize the alkyl borane produced in step 1 that leads to the synthesis of alcohols from
Anti-Markovnikov’s Rule serves as the basis of hydroboration. It states that the negative part of reagent must go to the carbon that has less alkyl substituents or more
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- What is the name of the following compound? SiMe3arrow_forwardK Draw the starting structure that would lead to the major product shown under the provided conditions. Drawing 1. NaNH2 2. PhCH2Br 4 57°F Sunny Q Searcharrow_forward7 Draw the starting alkyl bromide that would produce this alkyne under these conditions. F Drawing 1. NaNH2, A 2. H3O+ £ 4 Temps to rise Tomorrow Q Search H2arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

