
Concept explainers
Write Lewis symbols for the following atoms. (a) Kr; (b) Ge; (c) N; (d) Ga; (e) As; (f) Rb.
(a)
Interpretation:
Interpret Lewis symbol of Kr.
Concept introduction:
Lewis symbol is the representation of the atoms in elemental form found in the periodic table. The element is represented with the universal symbol along with the valence electron present in its valence shell.
The valence electron present in the atom is decided by total number of electrons present in outermost shell.
Answer to Problem 1E
Lewis symbol of Kr is:
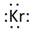
Explanation of Solution
In periodic table, Krypton is present in 18 group and in 4 period. The symbol of Krypton is Kr carrying 8 valence electrons in its valence shell.
Thus, the Lewis symbol of Kr is:
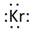
(b)
Interpretation:
Interpret the Lewis symbol of Ge.
Concept introduction:
Lewis symbol is the representation of the atoms in elemental form found in the periodic table. The element is represented with the universal symbol along with the valence electron present in its valence shell.
The valence electron present in the atom is decided by total number of electrons present in outermost shell.
Answer to Problem 1E
Lewis symbol of Ge is:

Explanation of Solution
In periodic table, Germanium is present in 14 group and in 4 period. The symbol of Germanium is Ge carrying 4 valence electrons in its valence shell.
Thus, the Lewis symbol of Ge is:

(c)
Interpretation:
Interpret the Lewis symbol of N.
Concept introduction:
Lewis symbol is the representation of the atoms in elemental form found in the periodic table. The element is represented with the universal symbol along with the valence electron present in its valence shell.
The valence electron present in the atom is decided by total number of electrons present in outermost shell.
Answer to Problem 1E
Lewis symbol of N is:
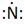
Explanation of Solution
In periodic table, Nitrogen is present in 15 group and in 2 period. The symbol of Nitrogen is N carrying 5 valence electrons in its valence shell.
Thus, the Lewis symbol of N is:
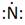
(d)
Interpretation:
Interpret the Lewis symbol of Ga.
Concept introduction:
Lewis symbol is the representation of the atoms in elemental form found in the periodic table. The element is represented with the universal symbol along with the valence electron present in its valence shell.
The valence electron present in the atom is decided by total number of electrons present in outermost shell.
Answer to Problem 1E
Lewis symbol of Ga is:
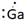
Explanation of Solution
In periodic table, Gallium is present in 13 group and in 4 period. The symbol of Gallium is Ga carrying 3 valence electrons in its valence shell.
Thus, Lewis symbol of Ga is:
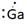
(e)
Interpretation:
Interpret the Lewis symbol of As.
Concept introduction:
Lewis symbol is the representation of the atoms in elemental form found in the periodic table. The element is represented with the universal symbol along with the valence electron present in its valence shell.
The valence electron present in the atom is decided by total number of electrons present in outermost shell.
Answer to Problem 1E
Lewis symbol of As is:
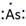
Explanation of Solution
In periodic table, Arsenic is present in 15 group and in 4 period. The symbol of Arsenic is As carrying 5 valence electrons in its valence shell.
Thus, the Lewis symbol of As is:
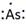
(f)
Interpretation:
Interpret the Lewis symbol of Rb.
Concept introduction:
Lewis symbol is the representation of the atoms in elemental form found in the periodic table. The element is represented with the universal symbol along with the valence electron present in its valence shell.
The valence electron present in the atom is decided by total number of electrons present in outermost shell.
Answer to Problem 1E
Lewis symbol of Rb is:
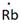
Explanation of Solution
In periodic table, Rubidium is present in 1 group and in 5th period. The symbol of rubidium is Rb carrying 1 valence electron in its valence shell.
Thus, the Lewis symbol of Rb is:
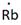
Want to see more full solutions like this?
Chapter 10 Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Additional Science Textbook Solutions
Biology: Life on Earth with Physiology (11th Edition)
Chemistry: The Central Science (14th Edition)
Introductory Chemistry (6th Edition)
Campbell Biology (11th Edition)
Microbiology: An Introduction
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
- Experiment 1 Data Table 1: Conservation of Mass - Initial Mass Data Table 1 Data Table 2 Data Table 3 Data Table 4 Panel 1 Photo 1 Data Table 5 Reaction Mass of test tube and 5.0% HC₂H₂O2 (g) # (A) (B) Mass of NaHCO, (g) Mass of balloon and NaHCO, (g) (C) 0.10 1 0829 14.38g 0.20 2 0.929 14.29g 0.35 1.00g 3 14.25g 0.50 1.14g 14.29 Experiment 1 Data Table 2: Moles of HC2H3O2 Reaction Volume of Mass of Moles of HC₂H₂O₂ 5.0% Vinegar (g) (ML) 5.0 0.25 0042 mol 2 5.0 0.25 0042 mol 3 5.0 0.25 0042 mol 5.0 0.25 0042 mol Experiment 1 Data Table 3: Moles of NaHCO3 Reaction Mass of NaHCO (g) 10g 20g 35g 50g Experiment 1 Data Table 4: Theoretical Yield of CO₂ Reaction # 1 2 3 Experiment 1 Total mass before reaction (g) (D=A+C) 15.29 15.21g 15.25g 15.349 Exercise 1 Data Table 1 Data Table 2 Data Table 3 Data Table 4 Panel 1 Photo 1 Data Table 5 Exercise 1- Data Table 1 Data Table 2 DataTable 3 Data Table 4 Panel 1 Photo 1 Data Table 5 Exercise 1- Moles of NaHCO 0012 mol 0025 mol 0044 mol 0062 mol…arrow_forwardThe chemical reaction you investigated is a two-step reaction. What type of reaction occurs in each step? How did you determine your answer?arrow_forwardWhat is the relationship between the limiting reactant and theoretical yield of CO2?arrow_forward
- From your calculations, which reaction experiment had closest to stoichiometric quantities? How many moles of NaHCO3 and HC2H3O2 were present in this reaction?arrow_forward18. Arrange the following carbocations in order of decreasing stability. 1 2 A 3124 B 4213 C 2431 D 1234 E 2134 SPL 3 4arrow_forwardAcetic acid is added to DI water at an initial concentration of 10 -6 M (Ka=1.8x10-5) A. Using the "ICE" Method, what would the pH be at equilibrium? State assumptions and show your work. B. Using the simultaneous equations method, what would the pH be at equilibrium? Show your workarrow_forward
- 1. Show that the change in entropy for a fixed amount of ideal gas held at a constant temperature undergoing a volume change is given by the simple equation AS = NkB In Hint: Start with the equation M dS = du + (Œ) dv - Ž (#) an, dU du+av-dN; j=1 Why doesn't the equation for the entropy of an ideal gas depend on the strength of the intermolecular forces for the gas?arrow_forward2. Make an ice cube at 1 bar pressure by freezing an amount of liquid water that is 2 cm x 2 cm x 2 cm in volume. The density of liquid water at 0 °C is 1.000 g cm³ and the density of ice at 0 °C is 0.915 g cm³. Note that this difference in density is the reason your water pipes burst if they freeze and why you shouldn't forget to take your bottle of pop out of the freezer if you put it in there to try and cool it down faster. A. What is the work of expansion upon freezing? B. Is work done on the system or by the system?arrow_forwardI have a excitation/emission spectra of a quinine standard solution here, and I'm having trouble interpreting it. the red line is emission the blue line is excitation. i'm having trouble interpreting properly. just want to know if there is any evidence of raman or rayleigh peaks in the spectra.arrow_forward
- Give the major product of the following reaction. excess 1. OH, H₂O 1.OH H CH3CH2CH21 H 2. A.-H₂O Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars. The single bond is active by default.arrow_forward2. Use Hess's law to calculate the AH (in kJ) for: rxn CIF(g) + F2(g) → CIF 3 (1) using the following information: 2CIF(g) + O2(g) → Cl₂O(g) + OF 2(g) AH = 167.5 kJ ΔΗ 2F2 (g) + O2(g) → 2 OF 2(g) 2C1F3 (1) + 202(g) → Cl₂O(g) + 3 OF 2(g) о = = -43.5 kJ AH = 394.1kJarrow_forwardci Draw the major product(s) of the following reactions: (3 pts) CH3 HNO3/H2SO4 HNO3/ H2SO4 OCH3 (1 pts)arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





