
General Chemistry: Atoms First
2nd Edition
ISBN: 9780321809261
Author: John E. McMurry, Robert C. Fay
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.1, Problem 7.2CP
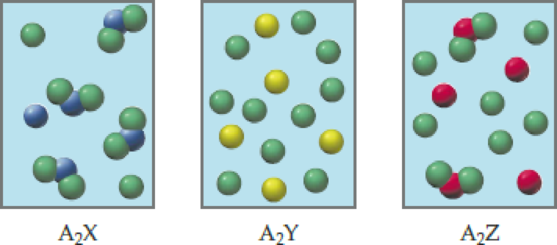
Three different substances, A2X, A2Y, and A2Z, are dissolved in water, with the following results. (Water molecules are omitted for clarity.) Which of the substances is the strongest electrolyte, and which is the weakest? Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Results
Search Results
Best Free Coursehero Unloc xb Success Confirmation of Q x
O Google Pas
alekscgi/x/lsl.exe/1o_u-IgNslkr 7j8P3jH-IQs_pBanHhvlTCeeBZbufu BYTI0Hz7m7D3ZcHYUt80XL-5alyVpwDXM TEZayFYCavJ17dZtpxbFD0Qggd1J
O States of Matter
Using a phase diagram to find a phase transition temperature or pressure
Gabr
3/5
he pressure above a pure sample of solid Substance X at 101. °C is lowered. At what pressure will the sample sublime? Use the phase diagram of X below to
nd your answer.
pressure (atm)
24-
12
solid
liquid
gas
200
400
temperature (K)
600
ote: your answer must be within 0.15 atm of the exact answer to be graded correct.
atm
Thanation
Check
© 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center I
Q Search
L³
ملة
301.7
348.9
193.7
308.6
339.5
160.6
337.7
464.7
223.5
370.5
326.6
327.5
336.1
317.9
203.8
329.8
221.9
331.7
211.7
309.6
223.4
353.7
334.6
305.6
340.0
304.3
244.7
QUESTION: Using this group of data on regular tomato sauce concentration readings answer the following questions:
1. 95% Cl Confidence Interval (mmol/L)
2. [Na+] (mg/100 mL)
3. 95% Na+ Confidence Interval (mg/100 mL)
Search Results
Search Results
Best Free Coursehero Unlo x b Success Confirmation of Q
aleks.com/alekscgi/x/sl.exe/10_u-lgNslkr7j8P3jH-IQs_pBan HhvlTCeeBZbufu BYTIOHz7m7D3ZcHYUt80XL-5alyVpwDXM TEZayFYCav
States of Matter
Using a phase diagram to find a phase transition temperature or pressure
Use the phase diagram of Substance X below to find the temperature at which X turns to a gas, if the pressure above the solid is 3.7 atm.
pressure (atm)
0.
32-
16
solid
liquid
gas
200
temperature (K)
Note: your answer must be within 20 °C of the exact answer to be graded correct.
Дос
X
Chapter 7 Solutions
General Chemistry: Atoms First
Ch. 7.1 - What is the molar concentration of Br ions in a...Ch. 7.1 - Three different substances, A2X, A2Y, and A2Z, are...Ch. 7.2 - Prob. 7.3PCh. 7.3 - Write net ionic equations for the following...Ch. 7.4 - Predict whether each of the following compounds is...Ch. 7.4 - Predict whether a precipitation reaction will...Ch. 7.4 - How might you use a precipitation reaction to...Ch. 7.4 - An aqueous solution containing an anion,...Ch. 7.5 - Prob. 7.9PCh. 7.5 - Give likely chemical formulas corresponding to the...
Ch. 7.5 - Write a balanced ionic equation and net ionic...Ch. 7.5 - The following pictures represent aqueous solutions...Ch. 7.6 - Assign an oxidation number to each atom in the...Ch. 7.7 - Prob. 7.14PCh. 7.7 - In each of the following reactions, tell which...Ch. 7.8 - Predict whether the following reactions will...Ch. 7.8 - Element B will reduce the cation of element A (A+)...Ch. 7.8 - Use the following reactions to arrange the...Ch. 7.9 - Write unbalanced half-reactions for the following...Ch. 7.9 - Balance the following net ionic equation by the...Ch. 7.9 - Balance the following equation by the...Ch. 7.10 - What is the molar concentration of Fe2+ ion in an...Ch. 7.10 - In the common lead storage battery used in...Ch. 7.10 - Many dozens of different solvents are used in...Ch. 7 - Prob. 7.25CPCh. 7 - Assume that an aqueous solution of a cation,...Ch. 7 - Assume that an aqueous solution of OH, represented...Ch. 7 - Prob. 7.28CPCh. 7 - Prob. 7.29CPCh. 7 - Prob. 7.30CPCh. 7 - Prob. 7.31CPCh. 7 - Prob. 7.32SPCh. 7 - Prob. 7.33SPCh. 7 - Write net ionic equations for the reactions listed...Ch. 7 - Prob. 7.35SPCh. 7 - Prob. 7.36SPCh. 7 - Prob. 7.37SPCh. 7 - Prob. 7.38SPCh. 7 - Prob. 7.39SPCh. 7 - Classify each of the following substances as...Ch. 7 - Prob. 7.41SPCh. 7 - What is the total molar concentration of ions in...Ch. 7 - What is the total molar concentration of ions in...Ch. 7 - Which of the following substances are likely to be...Ch. 7 - Which of the following substances are likely to be...Ch. 7 - Predict whether a precipitation reaction will...Ch. 7 - Predict whether a precipitation reaction will...Ch. 7 - Prob. 7.48SPCh. 7 - Prob. 7.49SPCh. 7 - Prob. 7.50SPCh. 7 - How would you prepare the following substances by...Ch. 7 - What is the mass and the identity of the...Ch. 7 - What is the mass and the identity of the...Ch. 7 - Prob. 7.54SPCh. 7 - Prob. 7.55SPCh. 7 - Prob. 7.56SPCh. 7 - Assume that you have an aqueous solution of an...Ch. 7 - Prob. 7.58SPCh. 7 - Prob. 7.59SPCh. 7 - Prob. 7.60SPCh. 7 - Write balanced ionic equations for the following...Ch. 7 - Write balanced net ionic equations for the...Ch. 7 - Prob. 7.63SPCh. 7 - If the following solutions are mixed, is the...Ch. 7 - Prob. 7.65SPCh. 7 - How many milliliters of 1.00 M KOH must be added...Ch. 7 - How many milliliters of 2.00 M HCl must be added...Ch. 7 - Where in the periodic table are the best reducing...Ch. 7 - Prob. 7.69SPCh. 7 - In each of the following instances, tell whether...Ch. 7 - Prob. 7.71SPCh. 7 - Prob. 7.72SPCh. 7 - Prob. 7.73SPCh. 7 - Prob. 7.74SPCh. 7 - Prob. 7.75SPCh. 7 - Prob. 7.76SPCh. 7 - Prob. 7.77SPCh. 7 - Prob. 7.78SPCh. 7 - Neither strontium (Sr) nor antimony (Sb) is shown...Ch. 7 - Prob. 7.80SPCh. 7 - Prob. 7.81SPCh. 7 - Prob. 7.82SPCh. 7 - Prob. 7.83SPCh. 7 - Prob. 7.84SPCh. 7 - Balance the half-reactions in Problem 7.83,...Ch. 7 - Prob. 7.86SPCh. 7 - Prob. 7.87SPCh. 7 - Balance the following half-reactions: (a) (acidic)...Ch. 7 - Prob. 7.89SPCh. 7 - Write balanced net ionic equations for the...Ch. 7 - Write balanced net ionic equations for the...Ch. 7 - Write balanced net ionic equations for the...Ch. 7 - Prob. 7.93SPCh. 7 - Prob. 7.94SPCh. 7 - Prob. 7.95SPCh. 7 - Prob. 7.96SPCh. 7 - Prob. 7.97SPCh. 7 - Prob. 7.98SPCh. 7 - Standardized solutions of KBrO3 are frequently...Ch. 7 - Prob. 7.100SPCh. 7 - Prob. 7.101SPCh. 7 - Prob. 7.102SPCh. 7 - Calcium levels in blood can be determined by...Ch. 7 - Balance the equations for the following reactions...Ch. 7 - Prob. 7.105CHPCh. 7 - Prob. 7.106CHPCh. 7 - Balance the equations for the following reactions...Ch. 7 - Prob. 7.108CHPCh. 7 - Prob. 7.109CHPCh. 7 - Prob. 7.110CHPCh. 7 - Prob. 7.111CHPCh. 7 - Prob. 7.112CHPCh. 7 - Succinic acid, an intermediate in the metabolism...Ch. 7 - How could you use a precipitation reaction to...Ch. 7 - How could you use a precipitation reaction to...Ch. 7 - Write a balanced net ionic equation for each of...Ch. 7 - A 100.0 mL solution containing aqueous HCl and HBr...Ch. 7 - Write balanced net ionic equations for the...Ch. 7 - Prob. 7.119CHPCh. 7 - Prob. 7.120CHPCh. 7 - When 75.0 mL of a 0.100 M lead(II) nitrate...Ch. 7 - Prob. 7.122CHPCh. 7 - Prob. 7.123CHPCh. 7 - Hydrogen peroxide can be either an oxidizing or...Ch. 7 - Prob. 7.125CHPCh. 7 - Iron content in ores can be determined by a redox...Ch. 7 - A mixture of FeCl2 and NaCl is dissolved in water,...Ch. 7 - Prob. 7.128MPCh. 7 - The following three solutions are mixed: 100.0 mL...Ch. 7 - Prob. 7.130MPCh. 7 - Prob. 7.131MPCh. 7 - Prob. 7.132MPCh. 7 - Prob. 7.133MPCh. 7 - Brass is an approximately 4:1 alloy of copper and...Ch. 7 - Prob. 7.135MPCh. 7 - Prob. 7.136MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the reaction below to answer the following questions: Acetoacetic ester can be prepared by the Claisen self-condensation reaction of ethyl acetate. 1. NaOEt, EtOH H&C OCH CH3 2 H30 H3C CH2 OCH2CH3 A. Write the complete stepwise mechanism for this reaction. Show all electron flow with arrows and draw all intermediate structures. B. Ethyl acetate can be prepared from ethanol as the only organic starting material. Show all reagents and structures for all intermediates in this preparation. C. Give the structures of the ester precursors for the following Claisen condensation product and formulate the reaction. OEtarrow_forwardUse the phase diagram of Substance X below to find the temperature at which X turns to a gas, if the pressure above the solid is 3.7 atm. pressure (atm) 32 16 solid liquid gas 0 0 200 temperature (K) Note: your answer must be within 20 °C of the exact answer to be graded correct. Шос ☑ كarrow_forwardStarting from bromoethane, how could you prepare the following compounds: a. Ethanol. b. Acetaldehyde f. Acetone. e. 2-Propanol i. Acetoacetic ester. d. 2-Bromoacetic acid. c. Acetic acid g. Acetamide. j. Ethylmalonate k. Gama ketoacid. h. Ethyl magnesium bromide.arrow_forward
- - The pressure above a pure sample of solid Substance X at 60. °C is raised. At what pressure will the sample melt? Use the phase diagram of X below to find your answer. pressure (atm) 02 0.4 solid Hliquid gas 0 0 200 400 600 temperature (K) Note: your answer must be within 0.025 atm of the exact answer to be graded correct. ☐ atmarrow_forward15. What is the order of decreasing reactivity towards nucleophilic acyl substitution for the carboxylic acid derivatives? (most reactive first) 0 O H3C COC CH3 H₂C C N(CH3)2 H3C C OCH3 A. a. I, 11, 111, b. I, III, IV, II C. II, IV, III, I ° (CH3)2CH C OCH3 IV d. II, I, III, IV B. R COCR 0 0 0 13= RC NH2 RC OR RC CI === IV a. I, III, II, IV b. II, III, I, IV C. III, II, I, IV d. IV, I, III, IIarrow_forwardDraw the formula of the product obtained by reacting D-Tallose with bromine water.arrow_forward
- Choose the best reagent(s) for carrying out the following conversions from the list below. Place the letter corresponding to the best choice in the blank to the left of the conversion. a. KMnO4, H3O+ b. Tollens' Reagent [oxidizing reagent] C. NaBH4, ethanol d. 1. BH3 2. H3O+ e. 1. CH3MgBr, ether 2. H3O+ f. CrO3, H2SO4, H₂O g. 1. Mg, ether 2. CO2 3. H3O+ h. 1. NaCN 2. H2SO4, H2, heat i. O3, then Zn and HOAC j. CH₂I A. B. C. CH CH=CHCH2COOH Br CEN CH COOH + HOOCCH COOH COOH 010 CH3arrow_forwardDraw the structures for each of the intermediates in the boxes provided for the synthesis below. OCH3 Fe HO HNO (CHOO pynding H₂504 LHNO2 NACH-I Fa H₂O HCL HNO 180arrow_forwardProvide structure(s) for the starting material(s), reagent(s) or the major organic product(s) of each of the following reactions or sequences of reactions. Show all relevant stereochemistry [three only] A. o 11 (CH3)CH — C— C ether (CH3)2CH-C-O-C-CH3 B. CH3 CHy CI Staf OH C. HC OCHS + H₂Oarrow_forward
- Consider the reaction sequence below to answer the following questions: EtO Compound X 1. NaOEt, EtOH OEt Br CO₂Et NaOEt, EtOH Compound Z CO₂Et Compound Y A. Compound X, diethyl propanedioate, is more commonly known as a. ethyl acetoacetate b. acetoacetic ester C. oxalic ester d. malonic ester 3. Write the complete stepwise mechanism for the conversion of Compound X into Compound Y. Show all electron flow with arrows and draw all intermediate structures.arrow_forwardClassify each of the following nitrogen atoms in the following compounds as primary, secondary, tertiary, or quaternary [three only] CH3 HO-CHCHNHCH3 A. B. C. H&C CH3 D. HO phedrine CH2CHCH3 amphetamine NH₂ mepiquat chloride faxofenadine OH H&C CH CO₂Harrow_forwardDraw the structure of the aldol self-condensation product for each of the following compounds. If a compound does not undergo aldol self-condensation, explain why it does not. A. B. CHICHCH₂OH CH3CHCH2CH CH3 CH3 C. CH 30 H3C-C-C-H CH3 questionsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Precipitation Reactions: Crash Course Chemistry #9; Author: Crash Course;https://www.youtube.com/watch?v=IIu16dy3ThI;License: Standard YouTube License, CC-BY