
Concept explainers
(a)
Interpretation:
The name of the following molecule should be determined.
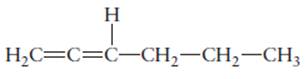
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons.
Rules of naming
- First choose the longest continuous chain of carbon atoms having double bond known as parent chain.
- The numbering of parent chain should be done in a way that the double bond and substituents get the lowest number.
- The root name of the carbon chain is same as in case of
alkanes , but “−ane” ending is replaced by “−ene” - The appropriate name should be given to every alkyl group and denote its position on the parent chain with the number.
- The alkyl groups are written in alphabetical order.
(b)
Interpretation:
The name of the following molecule should be determined.
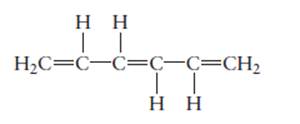
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Unsaturated hydrocarbon having double bond is known as alkene having general molecular formula
Rules of naming alkenes are:
- First choose the longest continuous chain of carbon atoms having double bond known as parent chain.
- The numbering of parent chain should be done in a way that the double bond and substituents get the lowest number.
- The root name of the carbon chain is same as in case of alkanes, but “−ane” ending is replaced by “−ene”
- The appropriate name should be given to every alkyl group and denote its position on the parent chain with the number.
- The alkyl groups are written in alphabetical order.
(c)
Interpretation:
The name of the following molecule should be determined.
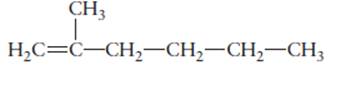
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Unsaturated hydrocarbon having double bond is known as alkene having general molecular formula
Rules of naming alkenes are:
- First choose the longest continuous chain of carbon atoms having double bond known as parent chain.
- The numbering of parent chain should be done in a way that the double bond and substituents get the lowest number.
- The root name of the carbon chain is same as in case of alkanes, but “−ane” ending is replaced by “−ene”
- The appropriate name should be given to every alkyl group and denote its position on the parent chain with the number.
- The alkyl groups are written in alphabetical order.
(d)
Interpretation:
The name of the following molecule should be determined.
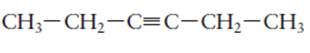
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Unsaturated hydrocarbon having double bond is known as alkene having general molecular formula
Rules of naming alkynes are:
1. First choose the longest continuous chain of carbon atoms having triple bond known as parent chain.
2. The numbering of parent chain should be done in a way that the triple bond and substituents get the lowest number.
3. The root name of the carbon chain is same as in case of alkanes, but “−ane” ending is replaced by “−yne”
4. The appropriate name should be given to every alkyl group and denote its position on the parent chain with the number.
5. The alkyl groups are written in alphabetical order.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Principles of Modern Chemistry
- One liter of chlorine gas at 1 atm and 298 K reacts completely with 1.00 L of nitrogen gas and 2.00 L of oxygen gas at the same temperature and pressure. A single gaseous product is formed, which fills a 2.00 L flask at 1.00 atm and 298 K. Use this information to determine the following characteristics of the product:(a) its empirical formula;(b) its molecular formula;(c) the most favorable Lewis formula based on formal charge arguments (the central atom is N);(d) the shape of the molecule.arrow_forwardHow does the square root mean square velocity of gas molecules vary with temperature? Illustrate this relationship by plotting the square root mean square velocity of N2 molecules as a function of temperature from T=100 K to T=300 K.arrow_forwardDraw product B, indicating what type of reaction occurs. F3C CF3 NH2 Me O .N. + B OMearrow_forward
- Benzimidazole E. State its formula. sState the differences in the formula with other benzimidazoles.arrow_forwardDraw product A, indicating what type of reaction occurs. F3C CN CF3 K2CO3, DMSO, H₂O2 Aarrow_forward19) Which metal is most commonly used in galvanization to protect steel structures from oxidation? Lead a. b. Tin C. Nickel d. Zinc 20) The following molecule is an example of a: R₁ R2- -N-R3 a. Secondary amine b. Secondary amide c. Tertiary amine d. Tertiary amidearrow_forward
- pls helparrow_forwardpls helparrow_forward35) Complete the following equation by drawing the line the structure of the products that are formed. Please note that in some cases more than one product is possible. You must draw all possible products to recive full marks! a. ethanol + 2-propanol + H2SO4 → b. OH conc. H2SO4 CH2 H3C CH + K2Cr2O7 C. d. H3C A pressure CH3 + H2 CH Pt catalystarrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





