
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 6.7, Problem 17P
What is the major product obtained from the reaction of HBr with each of the following?
a. 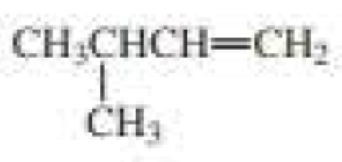
b. 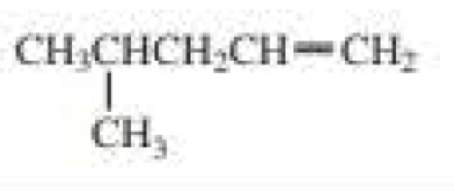
c. 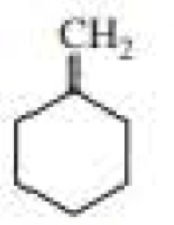
d. 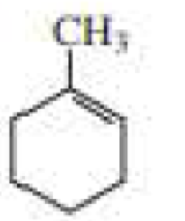
e. 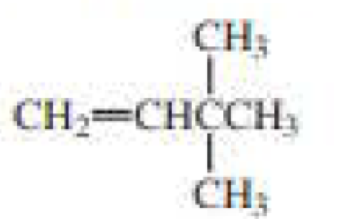
f. 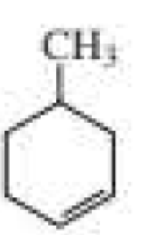
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
9. compore the Following two Venctions IN
termy Of Ronction Rate and explan in
detail the reasoning that led to your conclusion
+He p₁₂ 11-
ㅐ 15
.. +He
H #H
H
/
H
b. Compare
the Following too reactions 14
terms of reaction Rate and explain in detail
the reasoning that led to your conclusion
Н
d-C-
tłu
Na
+2446
е
-ll +2n
"H
a.
•Write all of the possible products
For the Following ronction
А
-----
H
-
H
H
+ H₂0 H+
Н
b. in Rite the complete reaction Mechaniszn
For the Formation of each product.
·C. Suggest what Reaction conditions could
Result in each product being the major
Product of the veaction:
a. Write the product For each of the
Following reactions
H
6-836-6
레
+H₂ N
A
H
A-C-C=C-C-CH + 2 Na +2 NH3 -
H H
b. Write the reaction Mechanism For.
reaction
each
Chapter 6 Solutions
Organic Chemistry
Ch. 6.1 - Draw the mechanism for the reaction of cyclohexene...Ch. 6.2 - a. How many bond orbitals are avilable for...Ch. 6.2 - Prob. 3PCh. 6.2 - Prob. 4PCh. 6.3 - Prob. 5PCh. 6.4 - Prob. 6PCh. 6.4 - What alkene should be used to synthesize each of...Ch. 6.4 - Prob. 8PCh. 6.5 - The pKa of a protonated alcohol is about 2.5, and...Ch. 6.5 - Prob. 10P
Ch. 6.5 - Prob. 11PCh. 6.6 - a. What is the major product or each or the...Ch. 6.6 - Prob. 14PCh. 6.6 - Prob. 15PCh. 6.7 - What is the major product obtained from the...Ch. 6.8 - Which is more highly regionselective: reaction of...Ch. 6.8 - Prob. 19PCh. 6.9 - What will be the product of the preceding reaction...Ch. 6.9 - Prob. 21PCh. 6.9 - Prob. 22PCh. 6.9 - Prob. 23PCh. 6.9 - What is the product of the addition of 1Cl to...Ch. 6.9 - What will be the major product obtained from the...Ch. 6.9 - Propose a mechanism for the following reaction:Ch. 6.10 - Draw structures for the following: a. 24...Ch. 6.10 - What alkene would you treat with a peroxyacid in...Ch. 6.11 - What products are formed when the following...Ch. 6.11 - Prob. 30PCh. 6.11 - Prob. 31PCh. 6.12 - Prob. 34PCh. 6.12 - Prob. 35PCh. 6.13 - Prob. 36PCh. 6.13 - Prob. 37PCh. 6.13 - Prob. 38PCh. 6.14 - What characteristics must the reactant of a...Ch. 6.15 - Prob. 40PCh. 6.15 - What stereoisomers are obtained from each of the...Ch. 6.15 - Prob. 45PCh. 6.15 - Prob. 46PCh. 6.15 - Prob. 47PCh. 6.15 - Prob. 49PCh. 6.15 - Prob. 50PCh. 6.15 - Prob. 51PCh. 6.15 - Prob. 52PCh. 6.15 - Prob. 53PCh. 6.16 - Prob. 54PCh. 6.17 - Prob. 55PCh. 6.18 - Explain why 3-methykyclohexene should not be used...Ch. 6.18 - Prob. 58PCh. 6 - Prob. 59PCh. 6 - Prob. 60PCh. 6 - What is the major product of the reaction of...Ch. 6 - Give two names for each of the following:Ch. 6 - Prob. 63PCh. 6 - Prob. 64PCh. 6 - Prob. 65PCh. 6 - What are the products of the following reactions?...Ch. 6 - When 3-methyl-1-butene reacts with HBr, two alkyl...Ch. 6 - Prob. 68PCh. 6 - Draw curved arrows to show the flow of electrons...Ch. 6 - What reagents are needed to carry out the...Ch. 6 - Prob. 71PCh. 6 - Prob. 72PCh. 6 - Prob. 73PCh. 6 - Prob. 74PCh. 6 - a. Draw the product or products that will be...Ch. 6 - The second-order rate constant (in units of M1s1)...Ch. 6 - Which compound has the greater dipole moment?Ch. 6 - Prob. 78PCh. 6 - Prob. 79PCh. 6 - Prob. 80PCh. 6 - Prob. 81PCh. 6 - Prob. 82PCh. 6 - Prob. 83PCh. 6 - Prob. 84PCh. 6 - Prob. 85PCh. 6 - Prob. 86PCh. 6 - Prob. 87PCh. 6 - Prob. 88PCh. 6 - Prob. 89PCh. 6 - Prob. 90PCh. 6 - 91. a. How many alkenes could you treat with H2,...Ch. 6 - Draw the products of the following reactions. If...Ch. 6 - Prob. 93PCh. 6 - Prob. 94PCh. 6 - Two chemists at Dupont found that lCH2Znl is...Ch. 6 - Prob. 96PCh. 6 - Prob. 97PCh. 6 - What alkene gives the product shown after...Ch. 6 - Prob. 99PCh. 6 - Prob. 100PCh. 6 - Prob. 101PCh. 6 - Prob. 102PCh. 6 - Prob. 103PCh. 6 - Propose a mechanism for the following reaction:Ch. 6 - Prob. 106PCh. 6 - Prob. 107P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- help draw the moleculearrow_forwardHow to draw this claisen condensation reaction mechanisms/arrow_forwardWrite all of Me Possible Products For each Of the Following reactions. In each case identity all pains of enantiomers, all digsterzoners and all Meso compounds 9. 11-60 11-0-11 V-G Η Η H ~ C-11 +HB+ - 1 H b. पन्ना 171-0-11 H-C-H Н C-C=c-call +HBr Perendez ==arrow_forward
- How can i draw the mechanisms for this molecule?arrow_forwarda. Discuss and explain he difference IN Stability between the Chai and Boat Гольцу от судомехане b. For the Following Molecule draw both possible Clain conformations and explain which one is more stable and for what Reason. H. CH₂ CH₂ H "Harrow_forwarddraw out these molecules pleasearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
SAR of Anticancer(Antineoplastic) Drug/ Alkylating agents/ Nitrogen Mustard; Author: Pharmacy Lectures;https://www.youtube.com/watch?v=zrzyK3LhUXs;License: Standard YouTube License, CC-BY