
EBK ESSENTIAL ORGANIC CHEMISTRY
3rd Edition
ISBN: 9780100659469
Author: Bruice
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.14, Problem 24P
Which
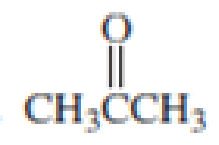
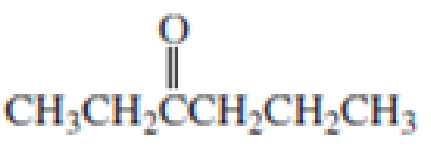
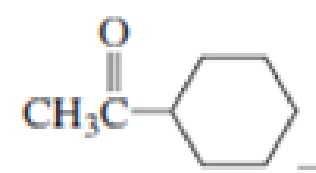
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
%Reflectance
95
90-
85
22 00 89
60
55
50
70
65
75
80
50-
45
40
WA
35
30-
25
20-
4000
3500
Date: Thu Feb 06 17:21:21 2025 (GMT-05:0(UnknownD
Scans: 8
Resolution: 2.000
3000
2500
Wavenumbers (cm-1)
100-
2981.77
1734.25
2000
1500
1000
1372.09
1108.01
2359.09
1469.82
1181.94
1145.20
1017.01
958.45
886.97
820.49
668.25
630.05
611.37
None
CH3
CH
H3C
CH3
H
OH
H3C-
-OCH2CH3
H3C
H
-OCH3
For each of the above compounds, do the following:
1. List the wave numbers of all the IR bands in the 1350-4000 cm-1 region.
For each one, state what bond or group it represents.
2. Label equivalent sets of protons with lower-case letters. Then, for each 1H NMR
signal, give the 8 value, the type of splitting (singlet, doublet etc.), and the number
protons it represents.
of
letter
δ value
splitting
# of protons
3. Redraw the compound and label equivalent sets of carbons with lower-case
letters.
Then for each set of carbons give the 5 value and # of carbons it represents.
letter
δ value
# of carbons
Chapter 6 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
Ch. 6.1 - Draw the mechanism for the reaction of cyclohexene...Ch. 6.2 - a. How many bond orbitals are available for...Ch. 6.2 - Prob. 3PCh. 6.2 - Prob. 4PCh. 6.3 - Prob. 5PCh. 6.3 - Prob. 6PCh. 6.3 - Prob. 7PCh. 6.5 - Prob. 9PCh. 6.5 - Prob. 10PCh. 6.5 - a. What is the major product of each of the...
Ch. 6.5 - Prob. 12PCh. 6.6 - What stereoisomers are obtained from each of the...Ch. 6.6 - Prob. 14PCh. 6.8 - Prob. 15PCh. 6.10 - Name the following:Ch. 6.10 - Draw the structure for each of the following: a....Ch. 6.10 - Draw the structures for and name the seven alkynes...Ch. 6.10 - Name the following:Ch. 6.10 - Name the following:Ch. 6.11 - What hybrid orbitals are used to form the...Ch. 6.13 - Prob. 22PCh. 6.14 - Prob. 23PCh. 6.14 - Which alkyne would be the best one to use for the...Ch. 6.14 - Prob. 25PCh. 6.14 - Prob. 26PCh. 6.15 - Describe the alkyne you would start with and the...Ch. 6.15 - What are products of the following reactions?Ch. 6 - Prob. 29PCh. 6 - Prob. 30PCh. 6 - Prob. 31PCh. 6 - Prob. 32PCh. 6 - What is each compounds systematic name?Ch. 6 - Prob. 34PCh. 6 - Prob. 35PCh. 6 - What reagents could be used to carry out the...Ch. 6 - Prob. 37PCh. 6 - Prob. 38PCh. 6 - Prob. 39PCh. 6 - Prob. 40PCh. 6 - Prob. 41PCh. 6 - Prob. 42PCh. 6 - Answer Problem 42 using 2-butyne as the starting...Ch. 6 - What is each compounds systematic name?Ch. 6 - Prob. 45PCh. 6 - Prob. 46PCh. 6 - Prob. 47PCh. 6 - Prob. 48PCh. 6 - Prob. 49PCh. 6 - Prob. 50PCh. 6 - Draw the keto tautomer for each of the following:Ch. 6 - Propose a mechanism for the following reaction...Ch. 6 - Prob. 53PCh. 6 - Prob. 54PCh. 6 - Prob. 55PCh. 6 - Propose a mechanism for the following reaction:Ch. 6 - Prob. 57PCh. 6 - Prob. 58PCh. 6 - Prob. 59PCh. 6 - Prob. 60PCh. 6 - Prob. 61PCh. 6 - Prob. 62P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonearrow_forwardCarbohydrates- Draw out the Hawthorne structure for a sugar from the list given in class. Make sure to write out all atoms except for carbons within the ring. Make sure that groups off the carbons in the ring are in the correct orientation above or below the plane. Make sure that bonds are in the correct orientation. Include the full name of the sugar. You can draw out your curve within the text box or upload a drawing below.arrow_forwardHow many milliliters of 97.5(±0.5) wt% H2SO4 with a density of 1.84(±0.01) g/mL will you need to prepare 2.000 L of 0.110 M H2SO4? If the uncertainty in delivering H2SO4 is ±0.01 mL, calculate the absolute uncertainty in the molarity (0.110 M). Assume there is negligible uncertainty in the formula mass of H2SO4 and in the final volume (2.000 L) and assume random error.arrow_forward
- Don't used hand raiting and don't used Ai solutionarrow_forward* How many milliliters of 97.5(±0.5) wt% H2SO4 with a density of 1.84(±0.01) g/mL will you need to prepare 2.000 L of 0.110 M H2SO4? * If the uncertainty in delivering H2SO4 is ±0.01 mL, calculate the absolute uncertainty in the molarity (0.110 M). Assume there is negligible uncertainty in the formula mass of NaOH and in the final volume (2.000 L) and assume random error.arrow_forwardYou are tasked with creating a calibration curve for the absorbance of cobalt solutions of various concentrations. You must prepare 5 standards with concentrations between 1.00 mg/L and 10.0 mg/L Co2+. You have a stock solution with a concentration of 40 mg/L Co2+ and all the standard lab glassware including transfer pipets and flasks. Explain how you would make your 5 standard solutions of various concentrations, including what glassware you would use to measure and prepare each solution.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY