
CHEMISTRY-MASTERINGCHEMISTRY W/ETEXT
8th Edition
ISBN: 9780135204634
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 6, Problem 6.25CP
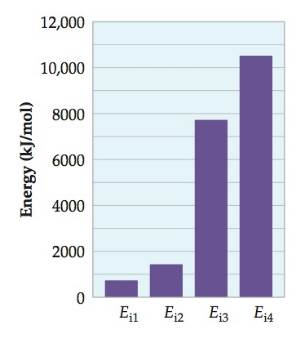
This figure represents the successive ionization energy of an atom in the third period of the periodic table. Which atom is this most likely to be?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
QUESTION: Fill in the answers in the empty green boxes regarding 'Question 5: Calculating standard error of regression'
*The images of the data showing 'coefficients for the standard curve' have been provided
Using the Nernst equation to calculate nonstandard cell voltage
Try Again
Your answer is wrong. In addition to checking your math, check that you used the right data and DID NOT round any intermediate calculations.
A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction:
2+
2+
Sn²+ Ba(s)
(aq) + Ba (s) Sn (s) + Ba²+ (aq)
→>>
Suppose the cell is prepared with 6.10 M Sn
2+
2+
in one half-cell and 6.62 M Ba
in the other.
Calculate the cell voltage under these conditions. Round your answer to 3 significant digits.
1.71 V
☐ x10
☑
5
0/5
?
00.
18
Ar
Question: Find both the b (gradient) and a (y-intercept) value from the list of data below:
(x1 -x̄)
370.5
(y1 - ȳ)
5.240
(x2 - x̄)
142.5
(y2 - ȳ)
2.004
(x3 - x̄)
28.5
(y3 - ȳ)
0.390
(x4 - x̄)
-85.5
(y4 - ȳ)
-1.231
(x5 - x̄)
-199.5
(y5 - ȳ)
-2.829
(x6 - x̄)
-256.5
(y6 - ȳ)
-3.575
Chapter 6 Solutions
CHEMISTRY-MASTERINGCHEMISTRY W/ETEXT
Ch. 6 - Prob. 6.1PCh. 6 - APPLY 6.2 Which of the following sets of ions are...Ch. 6 - Which atom or ion has the largest radius:...Ch. 6 - Conceptual APPLY 6.4 Which of the following...Ch. 6 - Use the periodic table to order the elements from...Ch. 6 - Given the orbital filling diagrams for the valence...Ch. 6 - Which has the largest third ionization energy: Be,...Ch. 6 - Conceptual APPLY 6.8 The figure on the right...Ch. 6 - Order the following elements from least to most...Ch. 6 - Conceptual APPLY 6.10 Which of the indicated three...
Ch. 6 - What electron configuration does the strontium...Ch. 6 - Prob. 6.12ACh. 6 - Prob. 6.13PCh. 6 - APPLY 6.14 Calculate the energy of electrostatic...Ch. 6 - Which substance has the largest lattice energy:...Ch. 6 - One of the following pictures represents NaCl and...Ch. 6 - Prob. 6.17PCh. 6 - What structural features do ionic liquids havethat...Ch. 6 - PROBLEM 6.18 Compare the following two ionic...Ch. 6 - PROBLEM 6.19 An ionic liquid consisting of a bulky...Ch. 6 - Where on the periodic table would you find the...Ch. 6 - Which of the following spheres is likely to...Ch. 6 - Circle the approximate part or parts of the...Ch. 6 - Prob. 6.24CPCh. 6 - This figure represents the successive ionization...Ch. 6 - In the following drawings, red spheres represent...Ch. 6 - Which of the following drawings is more likely to...Ch. 6 - Prob. 6.28CPCh. 6 - Which of the following alkali metal halides has...Ch. 6 - Which of the following alkali metal halides has...Ch. 6 - Three binary compounds are represented on the...Ch. 6 - Prob. 6.32CPCh. 6 - Prob. 6.33CPCh. 6 - What is the difference between a covalent bond and...Ch. 6 - Prob. 6.35SPCh. 6 - What is the difference between a molecule and an...Ch. 6 - Prob. 6.37SPCh. 6 - How many protons and electrons are in each of the...Ch. 6 - What is the identity of the element X in the...Ch. 6 - Prob. 6.40SPCh. 6 - Prob. 6.41SPCh. 6 - Prob. 6.42SPCh. 6 - Prob. 6.43SPCh. 6 - What doubly positive ion has the following...Ch. 6 - Prob. 6.45SPCh. 6 - Prob. 6.46SPCh. 6 - Which element in the transition-metal series Sc...Ch. 6 - Prob. 6.48SPCh. 6 - Prob. 6.49SPCh. 6 - Order the following ions from smallest to largest:...Ch. 6 - Order the following ions from smallest to largest:...Ch. 6 - Which ion has a larger atomic radius, Cu+ or Cu2+...Ch. 6 - Which ion hasa larger atomic radius, Fe2+ or Fe3+...Ch. 6 - The following ions all have the same number of...Ch. 6 - Which of the ions Se2,F,O2 and Rb+ has the largest...Ch. 6 - Which group of elements in the periodic table has...Ch. 6 - Prob. 6.57SPCh. 6 - Which element in each of the following sets has...Ch. 6 - Order the elements in each set from the smallest...Ch. 6 - (a) Which has the smaller second ionization...Ch. 6 - (a) Which has the smaller fourth ionization...Ch. 6 - Three atoms have the following electron...Ch. 6 - Three atoms have the following electron...Ch. 6 - The first four ionization energies in kJ/mol of a...Ch. 6 - The first four ionization energies in kJ/mol of a...Ch. 6 - Prob. 6.66SPCh. 6 - Prob. 6.67SPCh. 6 - Prob. 6.68SPCh. 6 - Prob. 6.69SPCh. 6 - Why is energy usually released when an electron is...Ch. 6 - Why does ionization energy increase regularly...Ch. 6 - No element has a negative second electron...Ch. 6 - Why does phosphorus have a less negative electron...Ch. 6 - Prob. 6.74SPCh. 6 - What noble-gas configurations and charge are the...Ch. 6 - Each of the following pairs of elements will react...Ch. 6 - Each of the following pairs of elements will react...Ch. 6 - Element X reacts with element Y to give a product...Ch. 6 - Element X reacts with element Y to give a product...Ch. 6 - Calculate the energy change in kilojoules per mole...Ch. 6 - Prob. 6.81SPCh. 6 - Find the lattice energy of LiBr(s) in Table 6.3,...Ch. 6 - Look up the lattice energies in Table 6.3, and...Ch. 6 - Born-4-Iaber cycles, such as those shown in...Ch. 6 - Calculate a lattice energy for CaH2(s) in...Ch. 6 - Calculate the overall energy change in kilojoules...Ch. 6 - The estimated lattice energy for CsF2(s) is +2347...Ch. 6 - Calculate the overall energy change in kilojoules...Ch. 6 - Use the data in Problem 6.88 to calculate an...Ch. 6 - Use the data and the result in Problem 6.84 to...Ch. 6 - Prob. 6.91SPCh. 6 - Calculate overall energy changes in kilojoules per...Ch. 6 - Prob. 6.93SPCh. 6 - We saw in Section 6.7 that the reaction of solid...Ch. 6 - Draw a Born—Haber cycle for the reaction of sodium...Ch. 6 - Use the following information plus the data given...Ch. 6 - Prob. 6.97SPCh. 6 - Prob. 6.98SPCh. 6 - Order the following compounds according to their...Ch. 6 - Prob. 6.100MPCh. 6 - Heating elemental cesium and platinum together for...Ch. 6 - Given the following information, construct a...Ch. 6 - Consider the electronic structure of the element...Ch. 6 - Prob. 6.104MPCh. 6 - The ionization energy of an atom can be measured...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Calculating standard reaction free energy from standard reduction... Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG° for the following redox reaction. Be sure your answer has the correct number of significant digits. 3Cu+ (aq) + Cro²¯ (aq) +4H₂O (1) → 3Cu²+ (aq) +Cr(OH)3 (s)+5OH˜¯ (aq) 0 kJ ☐ x10 00. 18 Ararrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 241.7 mL of a 0.4900M solution of methylamine (CH3NH2) with a 0.7800M solution of HNO3. The pK of methylamine is 3.36. Calculate the pH of the base solution after the chemist has added 17.7 mL of the HNO3 solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO3 solution added. Round your answer to 2 decimal places. pH = ☑ ? 18 Ararrow_forwardThe following is two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 223.4 148.7 353.7 278.2 334.6 268.7 305.6 234.4 340.0 262.7 304.3 283.2 244.7 143.6 QUESTION: For both groups of data calculate the answers attached in the image.arrow_forward
- The following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0mmol/L 262.7mmol/L QUESTION: For both groups (Regular & Salt Reduced tomato sauce) of data provide answers to the following calculations below: 1. Standard Deviation (Sx) 2. T Values (t0.05,4) 3. 95% Confidence Interval (mmol/L) 4. [Na+] (mg/100 mL) 5. 95% Confidence Interval (mg/100 mL)arrow_forwardIf we have leucine (2-amino-4-methylpentanoic acid), alanine (2-aminopropanoic acid) and phenylalanine (2-amino-3-phenylpropanoic acid), indicate the tripeptides that can be formed (use the abbreviated symbols Leu., Ala and Phe).arrow_forwardBriefly state why trifluoroacetic acid is more acidic than acetic acid.arrow_forward
- Explain why acid chlorides are more reactive than amides in reactions with nucleophiles.arrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 101.7 mL of a 0.3500M solution of piperidine (C5H10NH) with a 0.05700M solution of HClO4. The pK of piperidine is 2.89. Calculate the pH of the base solution after the chemist has added 682.9 mL of the HClO solution to it. 4 Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HClO solution added. 4 Round your answer to 2 decimal places. pH = .11 00. 18 Ararrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0 262.7 QUESTION: For both groups of data provide answers to the calculations attached in the imagearrow_forward
- 7. Concentration and uncertainty in the estimate of concentration (class data) Class mean for sample (Regular) |[Cl-] (mmol/L) class mean Sn za/2 95% Confidence Interval (mmol/L) [Na+] (mg/100 mL) 95% Confidence Interval (mg/100 mL)arrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 223.4 148.7 353.7 278.2 334.6 268.7 305.6 234.4 340.0 262.7 304.3 283.2 244.7 143.6 QUESTION: For both groups of data calculate the answers attached in the image.arrow_forwardGive reason(s) for six from the followings [using equations if possible] a. Addition of sodium carbonate to sulfanilic acid in the Methyl Orange preparation. b. What happened if the diazotization reaction gets warmed up by mistake. c. Addition of sodium nitrite in acidified solution in MO preparation through the diazotization d. Using sodium dithionite dihydrate in the second step for Luminol preparation. e. In nitroaniline preparation, addition of the acid mixture (nitric acid and sulfuric acid) to the product of step I. f. What is the main reason of the acylation step in nitroaniline preparation g. Heating under reflux. h. Fusion of an organic compound with sodium. HAND WRITTEN PLEASEarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning  Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Quantum Numbers, Atomic Orbitals, and Electron Configurations; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Aoi4j8es4gQ;License: Standard YouTube License, CC-BY
QUANTUM MECHANICAL MODEL/Atomic Structure-21E; Author: H to O Chemistry;https://www.youtube.com/watch?v=mYHNUy5hPQE;License: Standard YouTube License, CC-BY