
Organic Chemistry
11th Edition
ISBN: 9781118133576
Author: T. W. Graham Solomons, Craig Fryhle
Publisher: Wiley, John & Sons, Incorporated
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 30P
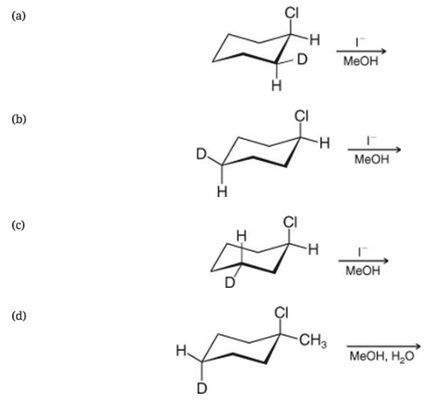
Write conformational structures for the substitution products of the following deuterium-labeled compounds:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
QUESTION: Fill out the answers to the empty green boxes attached in the image.
*Ensure you all incorporate all 27 values (per column)*
You need to make a buffer by dissolving benzoic acid and sodium benzoate in
water. What is the mass of benzoic acid that you would weigh out, in mg, to
create 50 mL of a buffer at pH = 4.7 that will change pH no more than 0.10 units
with the addition of 0.001 moles of acid or base?
Enter just the answer without the units (mg) - just the number will do!
Draw the formula for 3-isopropylcyclopentane-1-carbonyl chloride.
Chapter 6 Solutions
Organic Chemistry
Ch. 6 - Prob. 1PPCh. 6 - Prob. 2PPCh. 6 - Prob. 3PPCh. 6 - Prob. 4PPCh. 6 - Prob. 5PPCh. 6 - PRACTICE PROBLEM 6.6
reactions that involve...Ch. 6 - PRACTICE PROBLEM 6.7 Rank the following...Ch. 6 - Prob. 8PPCh. 6 - Prob. 9PPCh. 6 - Prob. 10PP
Ch. 6 - Prob. 11PPCh. 6 - Prob. 12PPCh. 6 - Prob. 13PPCh. 6 - Prob. 14PPCh. 6 - Prob. 15PPCh. 6 - Prob. 16PPCh. 6 - Prob. 17PPCh. 6 - Prob. 18PPCh. 6 - Prob. 19PPCh. 6 - Prob. 20PCh. 6 - Prob. 21PCh. 6 - Which SN1 reaction of each pair would you expect...Ch. 6 - Prob. 23PCh. 6 - Prob. 24PCh. 6 - Listed below are several hypothetical nucleophilic...Ch. 6 - Prob. 28PCh. 6 - Write conformational structures for the...Ch. 6 - 6.28 Consider the reaction of with .
(a) Would...Ch. 6 - Prob. 33PCh. 6 - Prob. 34PCh. 6 - Prob. 35PCh. 6 - Prob. 36PCh. 6 - 1-Bromobicyclo[2, 2,1] heptane is extremely...Ch. 6 - When ethyl bromide reacts with potassium cyanide...Ch. 6 - Prob. 41PCh. 6 - Prob. 42PCh. 6 - When the alkyl bromides (listed here) were...Ch. 6 - Prob. 44PCh. 6 - Prob. 45PCh. 6 - Prob. 46PCh. 6 - Prob. 47PCh. 6 - 6.42 The reaction of chloroethane with water in...Ch. 6 - Prob. 49PCh. 6 - Prob. 50PCh. 6 - Prob. 51PCh. 6 - Prob. 52PCh. 6 - 1-Bromo[2.2.1] bicycloheptane is unreactive toward...Ch. 6 - Open the computer molecular model tided...Ch. 6 - Prob. 56PCh. 6 - Consider the solvolysis reaction of (1S,...Ch. 6 - 2. Consider the following sequence of reactions,...Ch. 6 - Prob. 26PCh. 6 - Your task is to prepare isopropyl methyl ether by...Ch. 6 - Prob. 29PCh. 6 - 6.53 cis-4-Bromocyclohexanol racemic C6H10O...Ch. 6 - Prob. 31PCh. 6 - Explain the following observations: When...Ch. 6 - Prob. 38PCh. 6 - Prob. 1QCh. 6 - Prob. 2QCh. 6 - 6.3 A kinetic study yielded the following reaction...Ch. 6 - Prob. 4QCh. 6 - 6.5 Supply the missing reactants, reagents,...Ch. 6 - Which SN2 reaction will occur most rapidly....Ch. 6 - 6.7 Provide three-dimensional structures for the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
The bioremediation process shown in the photograph is used to remove benzene and other hydrocarbons from soil c...
Microbiology: An Introduction
A de-superheater has a flow of ammonia of 1.5kg/s at 1000kPa,100C that is mixed with another flow of ammonia at...
Fundamentals Of Thermodynamics
The genes dumpy (dp), clot (cl), and apterous (ap) are linked on chromosome II of Drosophila. In a series of tw...
Concepts of Genetics (12th Edition)
What is the difference between cellular respiration and external respiration?
Human Physiology: An Integrated Approach (8th Edition)
13. How are connective tissues classified? List the various types.
Principles of Anatomy and Physiology
In the explosion of a hydrogen-filled balloon, 0.50 g of hydrogen reacts with 4.0 g of oxygen. How many grams o...
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- QUESTION: Fill out the answers to the empty green boxes attached in the image. *Ensure you all incorporate all 27 values (per column)*arrow_forwardComplete the following reactions- hand written pleasearrow_forwardGive the organic product: O A O B Ос ○ D -NH–CH3 + CH3 CH3 NEN C ? A CH3 CH3 NH- CH3 B CH3 CH3 N=N- C CH3 CH3 N=NNH CH3 D CH3 N=N CH3 NHCH3 LNH CHOarrow_forward
- Finish the reaction- hand written pleasearrow_forwardGive the organic products: (benzyne) Br ? CH3 + K* :NH, liq NH3 HINT: Two products are formed. Each is a substituted aniline; they are isomers of each other. NH2 II I H₂N. CH3 CH3 III Select one: ○ A. I and II ○ B. I and III O C. I and IV O D. II and III O E. III and IV H₂N CH3 IV CH₂-NH2arrow_forwardPredict the major products of this organic reaction: HBr (1 equiv) cold ? Some important notes: • Draw the major product, or products, of this reaction in the drawing area below. • You can draw the products in any arrangement you like. • Pay careful attention to the reaction conditions, and only include the major products. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. • Note that there is only 1 equivalent of HBr reactant, so you need not consider the case of multiple additions. Erase something Explanation Check 2025 McGraw Hill LLC. All Rights Reserved. Terarrow_forward
- Q14. Fill this chart: (please refer to ppt notes/browser to answer these questions) What alcohol is also called wood alcohol? What is the common name of ethanol? Draw the structure of phenol and thiophene? Are bigger chain alcohol like heptanol and octanol are soluble or insoluble in water and explain it ? Are ethers soluble or insoluble in water? What suffix and prefix are used for alcohol while naming alcohol and ether? What the process called when we add water to any alkene to make alcohol? Q16. Draw the diagram of following aromatic compound (practice from previous module) Aniline Phenol Benzoic acid Methyl benzoate Q17. a. Write the oxidation reactions for the 2 propanol. b. Write the oxidation reaction of the ethanol.arrow_forwardQuestion 11 of 18 (1 point) Question Attempt: 3 of How many signals do you expect in the 'H NMR spectrum for this molecule? Br Br Write the answer below. Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H atoms that would contribute to the same signal as the H already highlighted red. Note for advanced students: In this question, any multiplet is counted as one signal. Number of signals in the 'H NMR spectrum. 1 For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. No additional Hs to color in top molecule Check For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box…arrow_forwardOrganic Chemistry Esterification reactions 1. Write the steps to prepare ester. 2. Write complete reaction of ethanol and acetic acid to make ester. 3. What does ester smell like? What are the uses of ester. 4. What the role of sulfuric acid in the esterification reactionarrow_forward
- 11. Complete the following esterification reaction with names of all the reactants and products under. Hint: Remove the water and end up with ester R-C-OH + ROH R-C-OR + H₂O A carboxylic acid An alcohol An ester Water BYJU'S H-C-C O-H Нин C-C-C-H HAAA H O-C-C-C-H AAA Ethanoic acid Propanol Water Propyl ethanoate By com CH3COOH + CH3CH2CH2CH₂CH₂OH → Practice for alcohols aldehydes and ketones: 12. Draw the structures from the following names mixed of alcohol/aldehyde and ketone: a. 4-methyl cyclohexanone b. 3-methyl-2-pentenal c. 2,3-dimethylcyclohexanone d. 1,3propanediol or Propane 1,3 diol 13. Write systematic names for the following compounds identify functional group: a. b. (CH3)2CH-C OH c) CH(CH₂)-- OH -,-,arrow_forwardmay you please show all steps! i am having a hard time understanding and applying in this format, thank you!arrow_forward10. Complete the substitution reaction of 2 pentanol with these reagents. Reagents & Reaction Conditions use practice sheet. Please write only major products, minor product like water, other gases are not required. Hint: In substitution of alcohol, we generally substitute OH group with Halogens like cl, Br, F using some reagent containing halogens. Ensure to add halogens to the same carbon number where you are removing OH from Examples Alcohols can be converted to Alkyl Halides with HX acids HBr H₂O HCI + H₂O HI + H₂O CH,CH₂OH + SOCI₂ CH,CH₂OH + PCI₁₂ A BBYJU'S CH CHCI + SO₂+ HCI CH₂CH CIP(OH), + HCI CH,CH₂OH + PCI CHCHCI + POCI + HCI CH,CH₂OH + PBr, CH,CH,Br + P(OH), + HBr 1. Reaction with HBr with 2 Pentanol 2.Reaction with HI with 2 pentanol © Byjus.com 3.Reaction with HCI+ZnCl,, with 2 pentanol (Zncl2 is catalyst no role) 4.Reaction with SOCI,, with 2 Pentanol 5.Reaction with PBr; or PCl, with 2 pentanolarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY